S'more Chemistry An Introduction To Stoichiometry Answers

Get ready to have your mind blown, because we're about to embark on a deliciously scientific adventure! Forget boring textbooks and stuffy labs. We're diving headfirst into the sweet, gooey world of s'more chemistry and uncovering the secrets of stoichiometry. It's not as intimidating as it sounds, I promise!
Think about your last epic campfire. What was the undisputed star of the show? That’s right, the magnificent, mouth-watering s'more! That perfect combination of toasted marshmallow, melty chocolate, and crunchy graham cracker is more than just a tasty treat; it's a miniature chemical masterpiece.
And guess what? The secret to making perfect s'mores, every single time, lies in a concept called stoichiometry. Don't let the big word scare you! It's basically the science of how much of each ingredient you need to make a chemical reaction – or in our case, a perfect s'more – happen.
Imagine you're a s'more-making superhero. You have a secret formula: one graham cracker, one piece of chocolate, and one marshmallow make one glorious s'more. This is your balanced chemical equation for s'more bliss! Without these precise amounts, things can get… well, messy.
What happens if you have way too many marshmallows but only one tiny piece of chocolate? You end up with a sad, lonely marshmallow, a little bit melty, maybe, but definitely not a s'more. This is what chemists call a limiting reactant. It's the ingredient that runs out first and dictates how much product you can make.
In our s'more analogy, the chocolate is our limiting reactant. If you only have one square of chocolate, you can only make one s'more, no matter how many marshmallows and graham crackers you have stockpiled. It's the bottleneck of your s'more production line!
On the flip side, you might have a mountain of marshmallows. These are your excess reactants. They're the leftovers, the ingredients that you have more than enough of to make all the s'mores your chocolate allows. They're important, but they don't determine the maximum number of s'mores you can whip up.

So, how does this relate to the official "S'more Chemistry: An Introduction to Stoichiometry Answers"? Well, these "answers" are just the scientific way of figuring out those perfect ingredient ratios. They tell us that if we want to make, say, ten s'mores, we need exactly ten graham crackers, ten pieces of chocolate, and ten marshmallows. It's that simple!
Think of it like baking a cake. If a recipe calls for 2 cups of flour, 1 cup of sugar, and 3 eggs, you can't just throw in a whole bag of flour and expect a delicious outcome. You need those specific proportions for the cake to rise beautifully and taste amazing. Stoichiometry is just the same, but for chemical reactions and, you guessed it, s'mores!
Scientists use these stoichiometry "answers" to predict how much of a chemical product they can create from a certain amount of starting materials. This is crucial for everything from making medicines to creating new materials. And it all starts with understanding these fundamental ratios, just like we do with our s'mores.
Let's get a little more specific with our s'more equation. Imagine we’re super organized and have a perfectly balanced s’more kit. This kit contains the ideal ratio of everything. No more, no less. It's pure s'more perfection in every package!
Now, what if we decide to get a little fancy and add a whole extra graham cracker to each s'more? This is where things get interesting. We've now increased the amount of one ingredient, but the others are still in their original, balanced proportions. Our graham cracker is now an excess reactant.
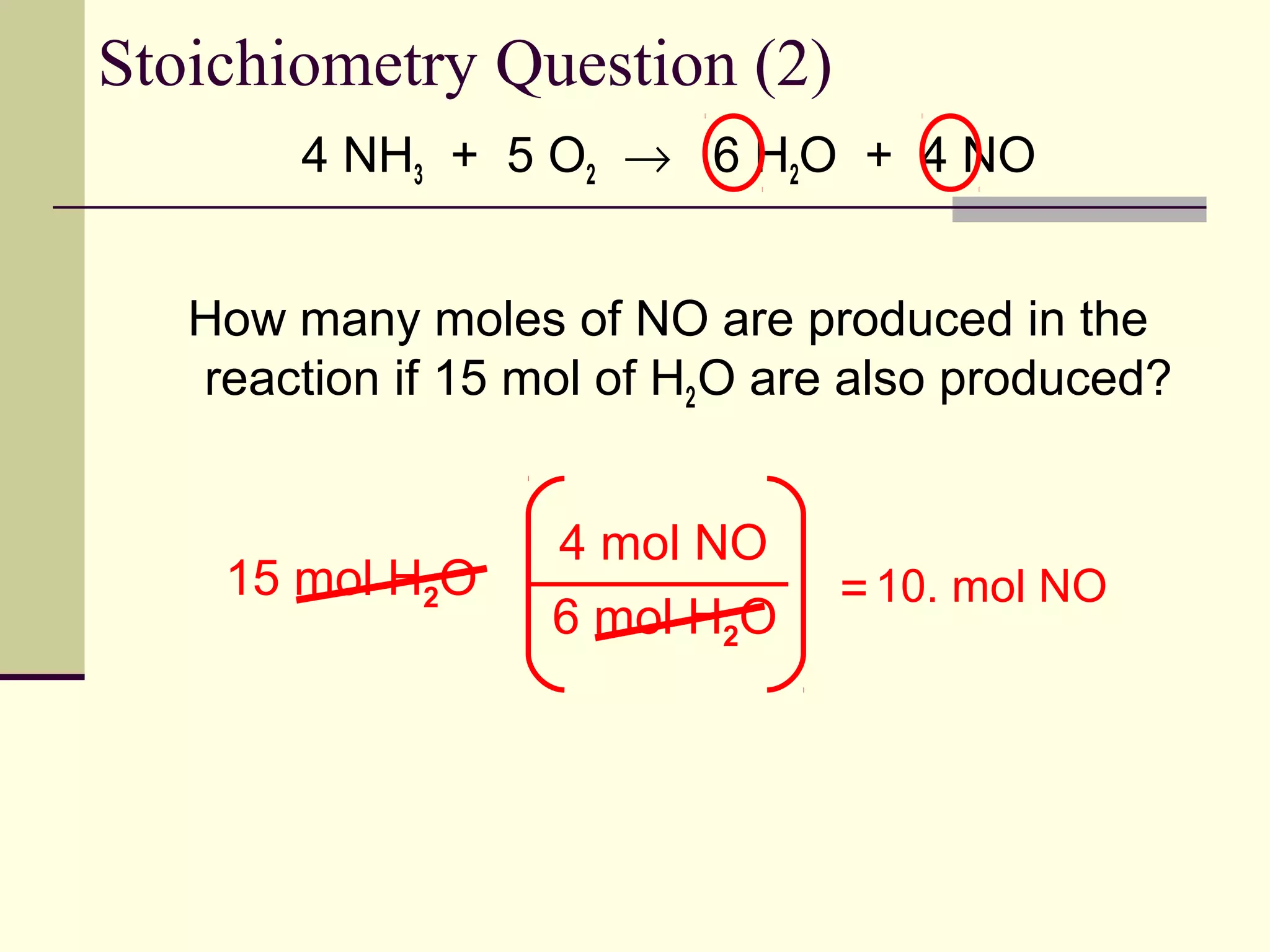
Even though we have extra graham crackers, we can still only make the same number of s'mores as before because our chocolate and marshmallows are still the limiting reactants. The number of chocolate squares dictates the s'more limit, not the abundance of graham crackers. It’s a tough lesson, but a tasty one!
The "S'more Chemistry: An Introduction to Stoichiometry Answers" essentially provide us with the perfect ratios. They're like the s'more-making commandments! Follow them, and you’ll achieve s'more nirvana. Ignore them, and you might end up with a pile of toasted marshmallows and a deep sense of regret.
It’s all about balance, my friends. Just like in life, having too much of one thing and not enough of another can throw things off. In the world of chemistry, and especially in the delicious realm of s'mores, stoichiometry is our guide to that perfect equilibrium.
Think about a chemistry lab where they're trying to synthesize a new compound. They meticulously measure out their reactants, armed with their stoichiometry answers. They know exactly how much of substance A to mix with substance B to get the maximum amount of their desired product, substance C.

This isn't just about making s'mores, though that's a fantastic starting point! This understanding of ratios is what powers industries. It’s how we ensure efficient production and minimize waste. Every time you see a product on a shelf, there's a good chance stoichiometry played a role in its creation.
So, next time you’re gathered around a crackling fire, with the sweet aroma of toasted marshmallows filling the air, take a moment to appreciate the science behind it all. You're not just making a snack; you're engaging in a delicious demonstration of stoichiometry!
You're a s'more scientist, a master of molecular combinations, a culinary chemist! Those "S'more Chemistry: An Introduction to Stoichiometry Answers" are your secret weapon. They unlock the door to perfectly proportioned, utterly delectable s'mores.
Don't be intimidated by the big words. Embrace the fun! Embrace the flavor! Embrace the chemistry! Because at the end of the day, who doesn't love a perfectly constructed s'more? It’s a testament to the beauty of balanced equations.
So, go forth and experiment responsibly! Grab your graham crackers, your chocolate, and your marshmallows. And remember, the "answers" to s'more stoichiometry aren't just numbers on a page; they're the keys to unlocking pure, unadulterated s'more joy. Your taste buds will thank you. The universe will thank you. And your campfire companions will definitely thank you.

The beauty of stoichiometry is that it applies to so many things beyond our campfire treats. It's a fundamental principle that helps us understand how everything around us interacts and transforms. From the air we breathe to the food we eat, the principles of balanced reactions are at play.
Consider a simple recipe for making water. You need two hydrogen atoms and one oxygen atom to make one water molecule. If you have a lot of hydrogen but only one oxygen, the oxygen is your limiting reactant. You can only make one water molecule. It’s a universal concept!
So, the next time someone asks you about stoichiometry, you can casually mention s'mores and watch their eyes light up. You’re not just another person who likes sweets; you’re a budding chemist with a serious understanding of proportional perfection. You're a legend!
The "S'more Chemistry: An Introduction to Stoichiometry Answers" are your friendly guide. They break down a complex scientific idea into a universally loved experience. They remind us that even the most ordinary things can have extraordinary scientific underpinnings. It’s a beautiful thing, really.
So, let's celebrate the perfect s'more. Let's celebrate the power of ratios. Let's celebrate the magic of chemistry! And let's all agree that stoichiometry, especially when it involves marshmallows and chocolate, is the coolest science ever. Keep those s'mores coming, and keep those chemical reactions balanced!
