Simple Distillation And Gas Chromatography Lab Report

Alright, gather 'round, my lab-coat-wannabes and science-curious cats! Today, we're diving headfirst into the glorious, sometimes slightly whiffy, world of a simple distillation and gas chromatography lab report. Now, before you picture me in a pristine white coat, meticulously swirling beakers like a mad scientist about to unleash a super-virus, let me set the record straight. My lab coat usually sports more questionable stains than a toddler's bib after a spaghetti incident, and my "mad scientist" moments are usually reserved for trying to figure out where I left my keys. But hey, we all have our talents, right?
So, what’s the big deal about this whole distillation and chromatography kerfuffle? Think of it like this: you've got a mysterious potion (or, you know, a mixture of liquids), and you want to figure out what's in it and how much of each thing you've got. It’s like being a detective, but instead of fingerprints, you’re looking for boiling points and retention times. And trust me, some of these chemical compounds have personalities as distinct as your Aunt Mildred’s award-winning fruitcake – notoriously difficult to separate!
Let's tackle simple distillation first. Imagine you have a murky brew of, let's say, rubbing alcohol and water. They're hanging out together, best buds, but you want to give them their own personal space. Simple distillation is basically the ultimate relationship counselor for liquids. You heat up your mixture, and the liquid with the lower boiling point – in our case, the alcohol – gets all excited, turns into a gas, and zips off into its own little adventure.
This gaseous adventurer then travels through a fancy-pants contraption called a condenser. Picture a long, glass tube with cold water snaking around it. It's like a really cold shower for our gas. Poor little guy, all excited from the heat, suddenly gets a blast of arctic air and, BAM! It condenses back into a liquid. Ta-da! You’ve separated the two! It's like kicking out a freeloading houseguest – they’re still in the building, but now they’re in their own separate room. And you, my friend, are left with a much purer sample. It’s not magic; it's just science with a splash of hot and cold. Revolutionary, I know.
Now, this is where things get a little more sophisticated, a bit like upgrading from a flip phone to a smartphone. Enter gas chromatography, or GC for those in the know (and now, you are!). If simple distillation is like a gentle nudge, GC is like a high-speed chase for molecules. Imagine you have a really complex mixture, like the aroma of a freshly baked croissant, or perhaps, a particularly pungent cheese. Trying to separate those with just heat? Good luck with that! You’d probably end up with a smoky mess and a very disappointed nose.
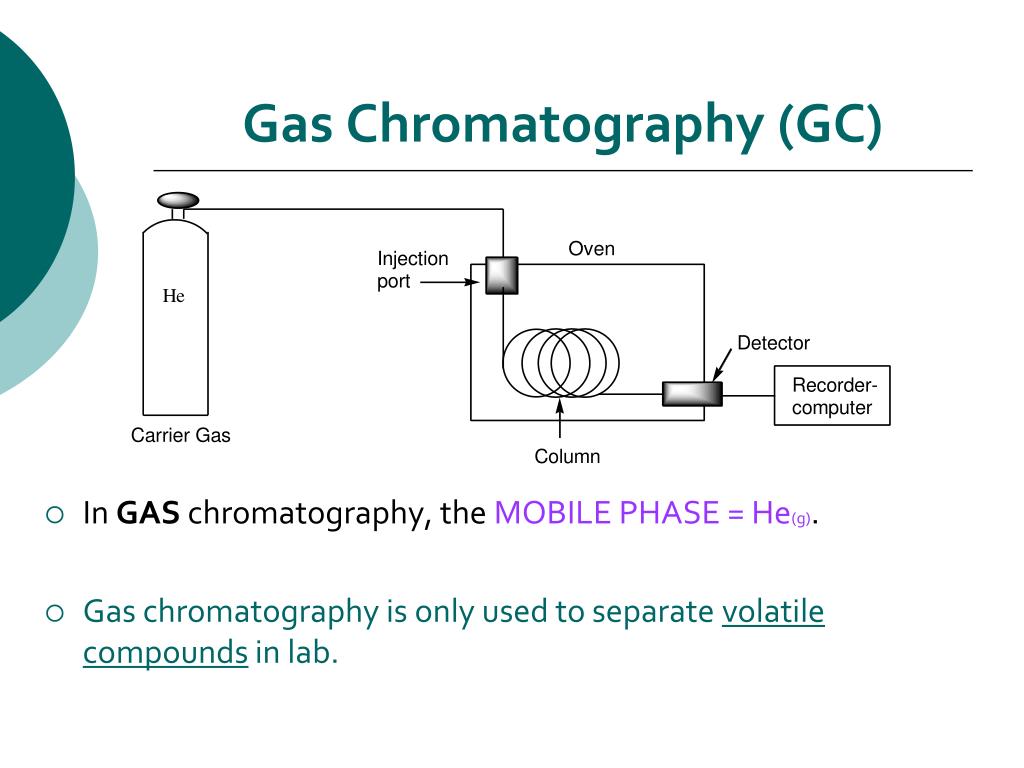
GC is like a sophisticated race track for your chemical compounds. You inject your (usually vaporized) mixture into a long, thin tube called a column. This column is packed with a special material, sort of like a chemical VIP lounge. Then, an inert gas, usually helium (because, let's face it, helium makes everything more fun), acts as the carrier, pushing your compounds through the column. And here’s the kicker: different compounds will interact with the material in the column differently.
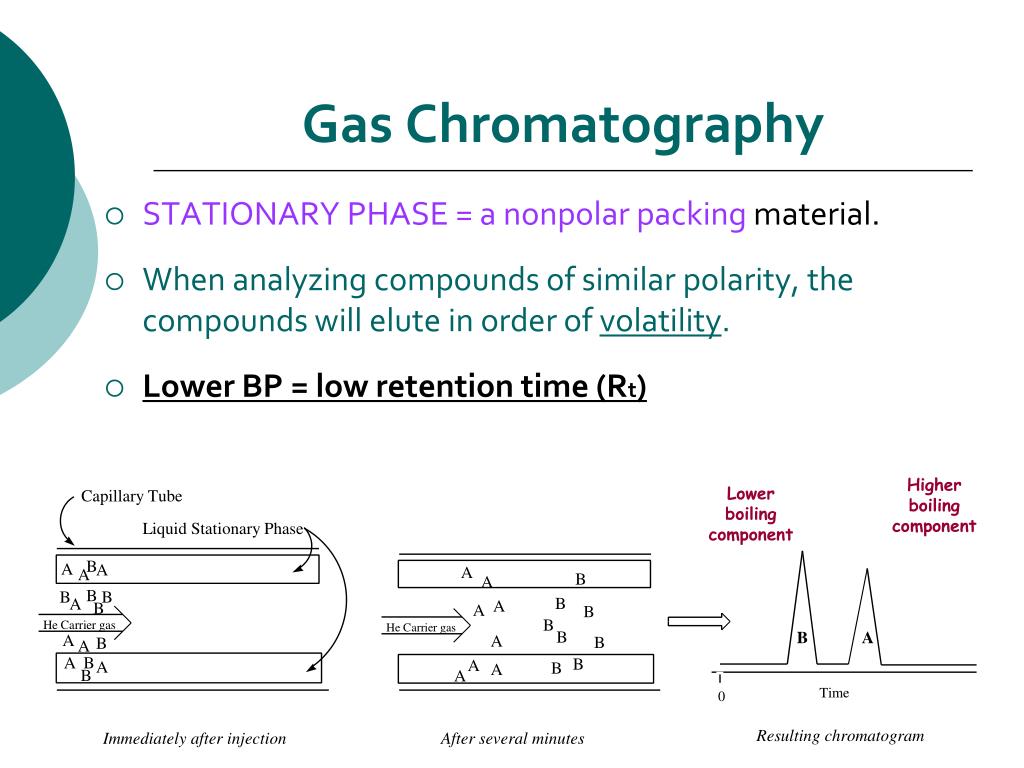
Some are like the super-popular kids, hanging out at the front of the pack, barely interacting with anyone. Others are like the wallflowers, taking their sweet time, mingling with the packing material, and getting a little delayed. The faster they zoom through, the less they like the party inside the column. The ones that take their sweet time? They’re the life of the party, or at least, the ones who get really chatty with the packing material.
And as each compound exits the column, it hits a detector. This detector is like the finish line judge, registering when each chemical "runner" crosses. It then spits out a graph, a glorious chromatogram, which looks like a series of humps. Each hump represents a different compound in your original mixture. The position of the hump tells you what it is (based on how long it took to get there – its retention time), and the size of the hump tells you how much of it there is. It’s like a molecular fingerprint and a popularity contest rolled into one!

So, in our hypothetical lab report, we might have started with a mixture that we thought was just alcohol and water. But lo and behold, thanks to the wonders of GC, we might discover a sneaky little third component, perhaps a trace of acetone, that we completely missed with our simple distillation. It's like finding an unexpected guest at your party – sometimes a delightful surprise, sometimes a slightly awkward encounter, but always an informative one.
The report itself? Well, that’s where we translate our scientific adventures into something vaguely resembling human language. We talk about our methods (the "how we did it," which usually involves a lot of fiddling and hoping for the best), our results (the "what we found," often represented by those pretty hump-graphs), and our conclusions (the "what it all means," which can sometimes feel like deciphering ancient hieroglyphs). We discuss potential sources of error, which is a polite way of saying, "Oops, we might have spilled a little bit, or the Bunsen burner decided to take a nap."
And then there’s the discussion section. This is where we get to sound super smart. We compare our findings to known values, ponder the implications of our discoveries, and maybe even theorize about the universe. Or, more likely, we explain why our distilled water had a faint whiff of something other than pure H2O. Perhaps a rogue yeast cell decided to audition for a role in our experiment? You never know!
The beauty of these techniques is their versatility. Simple distillation is great for separating liquids with significantly different boiling points, like getting the essential oils out of lavender or purifying that questionable homemade moonshine. GC, on the other hand, is your go-to for analyzing complex mixtures, from the ingredients in your perfume to the pollutants in the air. It’s like having a microscopic detective squad at your disposal.
So, next time you hear about a simple distillation or gas chromatography lab report, don't picture stuffy academics in sterile rooms. Picture a slightly chaotic but ultimately enlightening journey of separation and discovery. It's about understanding the building blocks of the world around us, one boiled, condensed, or chromatographically separated molecule at a time. And who knows? Maybe one day, we'll even use these techniques to figure out why my socks disappear in the laundry. That, my friends, would be a groundbreaking discovery indeed!
