Silicon-oxygen Bonds Are Stronger Than Magnesium-oxygen Bonds Because:
Hey there, curious minds! Ever wonder why some things in the world just… stick together really, really well? Like, rock-solidly? Today, we’re diving into a little bit of material magic, specifically why those sneaky silicon-oxygen bonds are total powerhouses, punching way above their weight class compared to magnesium-oxygen bonds. Sounds science-y, I know, but stick with me. It’s actually super cool and explains a lot about the stuff we see every day!
So, let’s break it down. Imagine you have two people holding hands. Sometimes it's a gentle, casual grip, and other times it's a really firm, determined embrace. Chemical bonds are kind of like those hand-holds, but between atoms. They’re what hold everything together, from the air you breathe to the phone you’re probably reading this on.
The Usual Suspects: Silicon, Oxygen, and Magnesium
We’re talking about silicon (Si), oxygen (O), and magnesium (Mg). These are pretty common elements. Oxygen is like the universal connector, always eager to pair up. Silicon is famously found in sand and glass (think silicon dioxide, aka SiO₂ – that’s quartz!). Magnesium is that lightweight metal you find in things like… well, that special chalk rock climbers use to keep their hands dry, and also in alloys for airplanes because it’s light and strong. Makes sense, right?
Now, when atoms bond, they’re usually trying to get to a happy, stable state. Often, this involves sharing or transferring electrons, which are tiny, negatively charged particles. Think of electrons as the "glue" that holds atoms together.
Why is Silicon-Oxygen So Tough?
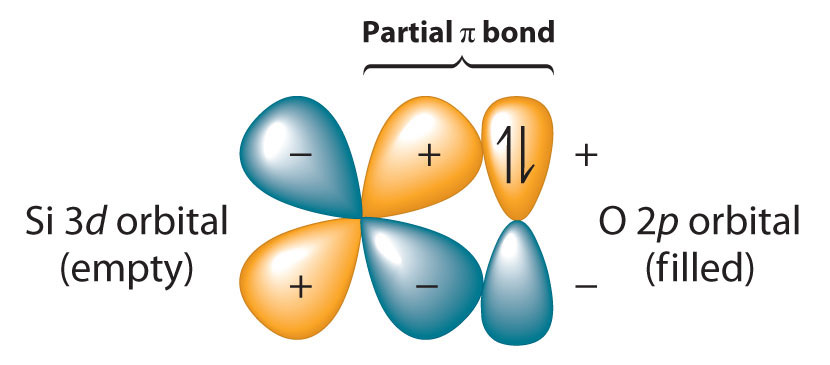
Okay, so here’s where the plot thickens. The silicon-oxygen bond is just… stronger. It’s like comparing a friendly handshake to a full-on bear hug. Why this difference? It all boils down to a few key factors, and they’re pretty neat when you think about them.
One of the big players is something called electronegativity. Don't let the fancy name scare you! It just means how much an atom wants to pull electrons towards itself. Oxygen is a real electron-hog. It’s super electronegative. Silicon is also pretty electronegative, but not quite as much as oxygen. Magnesium, on the other hand, is a bit of a pushover when it comes to electrons; it’s not very electronegative at all.
When silicon and oxygen bond, oxygen is really pulling on those shared electrons. This creates a kind of "polarized" bond, where the electrons spend more time around the oxygen atom, leaving the silicon atom a little bit electron-deprived. This strong pull and uneven sharing creates a powerful attraction, making the bond very stable and difficult to break. It's like two people playing tug-of-war, and one is pulling with all their might!
Magnesium and oxygen? Magnesium is more than happy to give its electrons to oxygen. This isn't a shared bond in the same way; it's more like an ionic bond, where electrons are essentially transferred. While this is also a strong attraction, the way silicon and oxygen share (or, more accurately, unevenly share) electrons creates a bond with a bit more… oomph.
The Role of Size and Shape
It’s not just about electron-grabbing, though. The size of the atoms also plays a role. Silicon atoms are a bit smaller than magnesium atoms. Think of it like this: imagine trying to wrap your arms around someone. If they’re a little smaller, you can get a tighter, more intimate hug. In the case of silicon and oxygen, their relative sizes allow for a very close and efficient overlap of their electron clouds. This close proximity means the attraction between the positively charged nucleus of one atom and the negatively charged electrons of the other is more intense.
Magnesium is larger. When it bonds with oxygen, the atoms are a bit further apart, and the electron "overlap" isn’t quite as snug. This means the attractive forces, while still significant, aren’t as intensely focused as they are in a silicon-oxygen bond.
A Fun Analogy Time!
Let’s try a fun comparison. Imagine you’re trying to build with LEGOs.
Silicon and Oxygen: This is like having two LEGO bricks that have really strong magnets built into them. When you snap them together, they click with immense force. You have to put in a good amount of effort to pull them apart. These are your tough ceramics and glasses!
Magnesium and Oxygen: This is more like two LEGO bricks with a very sturdy interlocking mechanism. They hold together really well, but the force is more about the physical shape fitting together perfectly, rather than an invisible magnetic pull. This is more like the ionic compounds you find with magnesium. Still strong, but the nature of the connection is a little different, and in the case of Si-O, the "magnetic" strength is often greater.
Another way to think about it: Imagine you're trying to break a sticky note. It's easy to peel off, right? That's kind of like some weaker bonds. Now imagine trying to break a piece of super glue that has set. That's more like the silicon-oxygen bond – it takes a serious effort and energy to break it!
Why Does This Matter?
So, why should we care about the strength of these bonds? Well, it has HUGE implications for the materials we use!

The incredible strength of silicon-oxygen bonds is why silica-based materials, like glass and many ceramics, are so durable and resistant to heat. Think about your coffee mug, the windshield of your car, or even the circuits in your computer (which rely heavily on silicon!). These are all made possible because those Si-O bonds are incredibly tough to break.
Materials like magnesium oxide (MgO), while still quite strong, have slightly different properties. They might be more brittle or have different melting points. Understanding these bond differences allows scientists and engineers to design new materials with specific properties. Want something that can withstand extreme heat? You’re probably looking at materials rich in silicon and oxygen. Need something lightweight and strong for an aircraft? Magnesium alloys might be your friend.
It's this subtle dance of electrons, atomic sizes, and the fundamental forces of nature that gives us the incredible variety of materials we have at our disposal. The next time you see a piece of glass or a sturdy ceramic, you can appreciate the unseen power of those strong silicon-oxygen bonds holding it all together!
Isn't it fascinating how much power is locked into the tiny interactions between atoms? It’s a constant reminder that even the seemingly mundane stuff around us is a marvel of chemical engineering!
