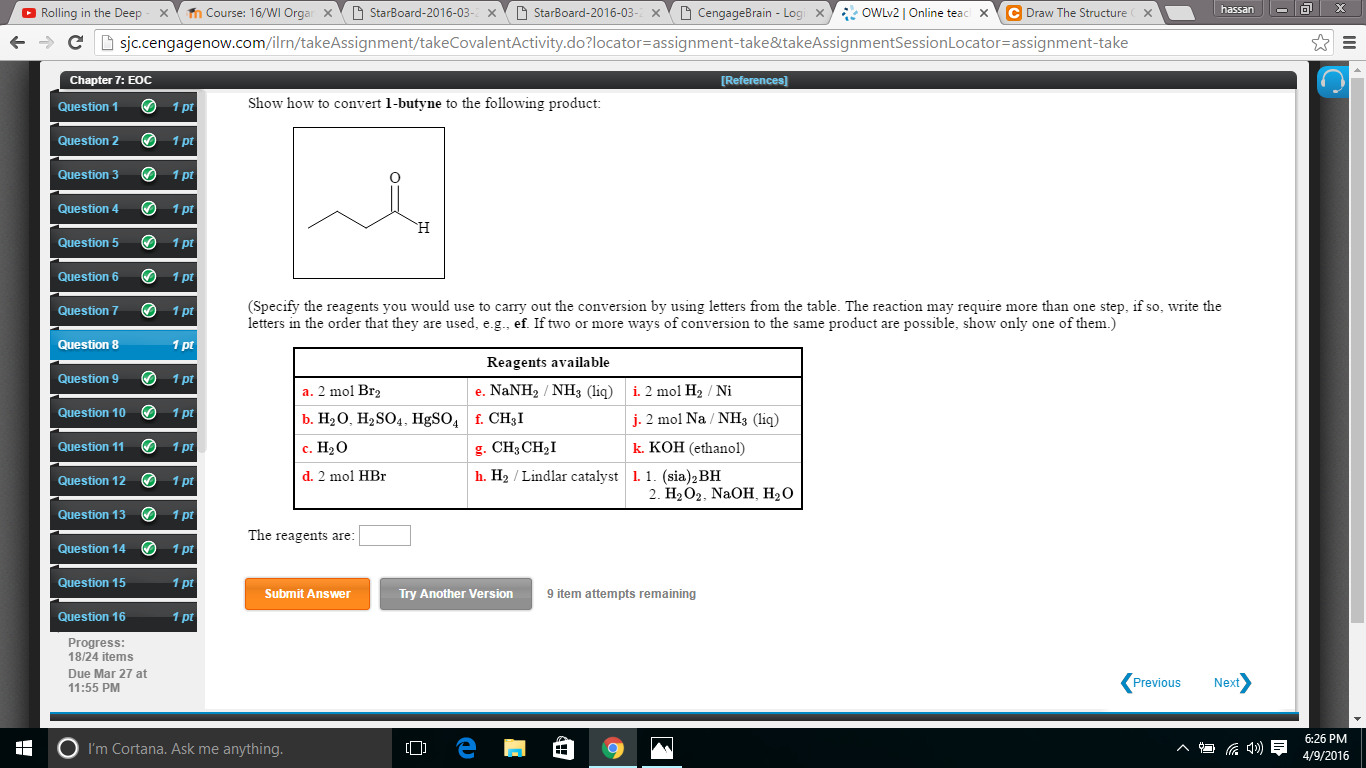
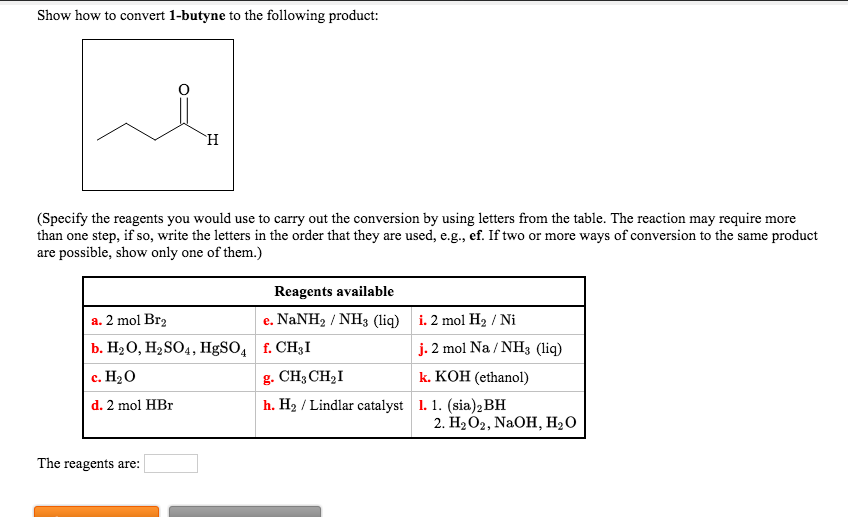
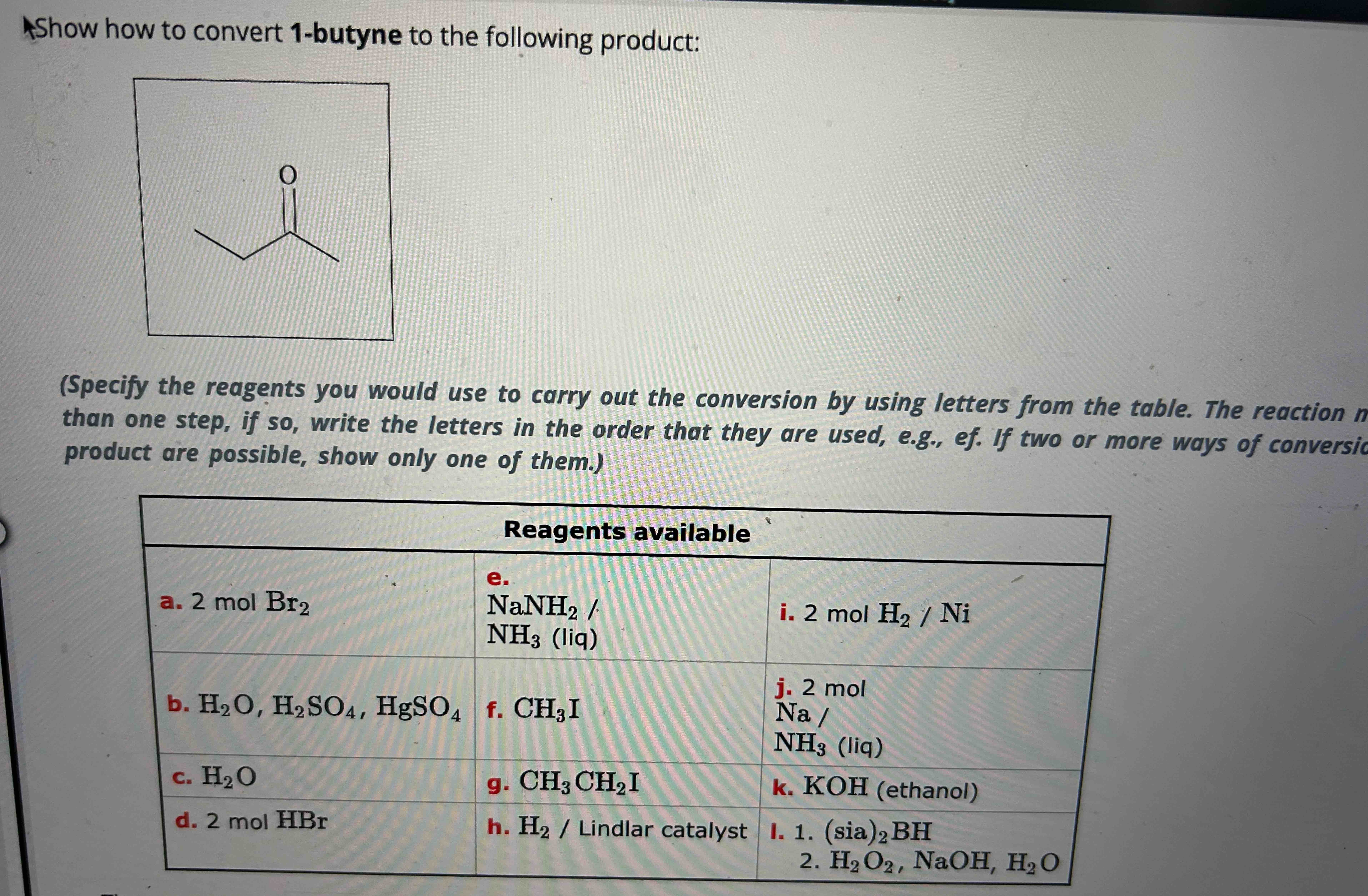
Show How To Convert 1 Butyne To The Following Product
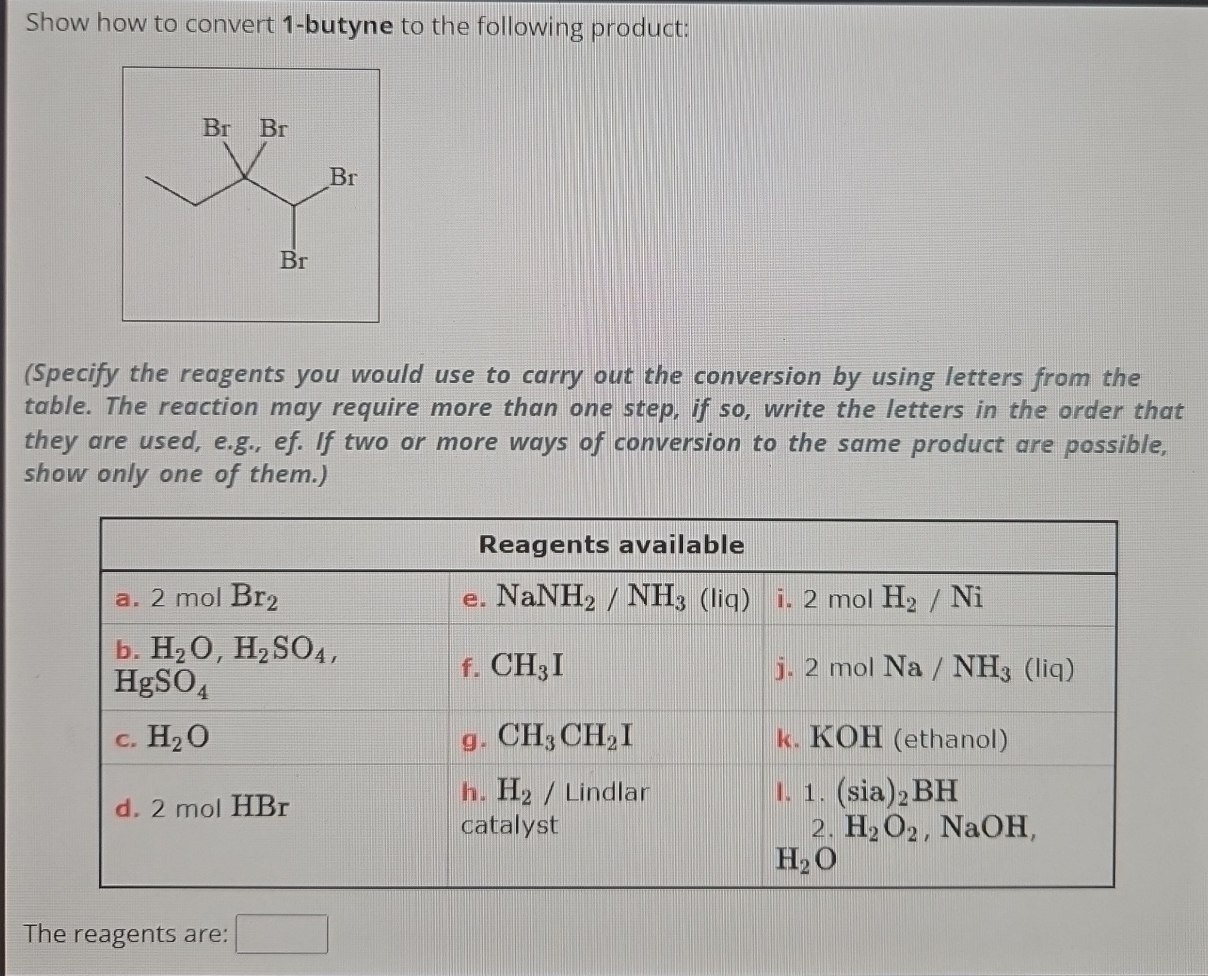
Alright, gather 'round, my caffeinated comrades! Let's talk about something that sounds like it belongs in a mad scientist's lab, but is actually pretty darn cool and, dare I say, delicious in a chemical sense. We're going to take a journey, a chemical escapade, from the humble beginnings of 1-butyne and transform it into something… well, something you might actually want to put in your body (after a LOT of rigorous testing, of course, we're not that mad). Think of it as a culinary makeover, but for molecules!
So, what is this mysterious 1-butyne? Imagine a very skinny, very excited molecule. It's got four carbon atoms all lined up, looking like a tiny, carbon-chain caterpillar. But here's the kicker: between the first and second carbon, there's a triple bond. Like, super glued together. They're practically holding hands with three little chemical hands. This triple bond is where all the magic, or in this case, the potential for exciting transformations, happens. It's the molecule's party trick, its "look at me!" moment. And just so we're clear, "butyne" tells us there are four carbons, and the "yne" suffix screams "triple bond!" The "1-" just means that triple bond is chilling right at the beginning of the chain. Easy peasy, right?
Now, our mission, should we choose to accept it (and we totally do, because where's the fun in saying no to chemistry?), is to turn this racy, triple-bonded creature into a specific product. Let's call it the "Fancy Fiesta Molecule." The instructions are a tad vague on what the Fancy Fiesta Molecule is, but that's part of the adventure! Think of it like getting a recipe that just says "make something delicious." We'll figure it out as we go, guided by the trusty laws of chemical transformation. For now, let's assume our Fancy Fiesta Molecule is something a bit more… relaxed than our hyperactive 1-butyne.
Our first step is to take that wild triple bond and make it a bit more manageable. Triple bonds are like really intense relationships. They’re strong, but they can be a bit… much. To tame this beast, we're going to introduce some hydrogen. Yep, good old H2. It's like bringing in a mediator to help two overexcited molecules calm down. We're going to add hydrogen across that triple bond, but we don't want to go too wild. If we just threw a bunch of hydrogen at it, we’d end up with a saturated, boring molecule. We're aiming for something in between.
This is where a special type of reaction comes in, a bit like using a very precise set of tiny chemical chopsticks. We're going to perform a catalytic hydrogenation. "Catalytic" just means we're using a helper molecule, a catalyst, to speed things up without actually getting consumed in the process. Think of it as the stage manager for our chemical play. The catalyst we're going to use is super cool: Lindlar's catalyst. This stuff is like a velvet rope at a VIP party. It's designed to stop the hydrogenation at a specific point – the cis-alkene stage. It's like saying, "Okay, you can have a nice, strong double bond, but no more!"
Lindlar's catalyst is usually made of palladium (a precious metal that looks a bit like silver and is used in fancy jewelry, so it's literally bling for your beaker!) that's been poisoned. "Poisoned" sounds bad, right? But in chemistry, it means we've intentionally made it less reactive. We've added something, like lead acetate or quinoline, to its surface. This makes it a bit picky, so it will only add one molecule of hydrogen across the triple bond, turning it into a double bond, and importantly, it forces the two new hydrogen atoms to attach to the same side of the double bond. This creates a cis-alkene.
Imagine our 1-butyne caterpillar again. The triple bond is where the exciting "wiggle" is. Lindlar's catalyst comes in, and with its help, we add two hydrogens. Instead of just sticking anywhere, they decide to be bros and bond to the same side of the new double bond. Voila! We've now transformed 1-butyne into (Z)-2-butene. "Z" here is the German word "zusammen," meaning "together," which is a fancy way of saying they're on the same side. It's like our caterpillar decided to grow a pair of cool, matching sunglasses.

So, we've gone from a triple threat to a double delight, specifically a cis-double bond. This is a pretty significant achievement! We've taken a highly reactive molecule and made it… well, still reactive, but in a more controlled, elegant way. Think of it like going from a chaotic rock concert to a smooth jazz performance. The energy is still there, but it's been refined.
Now, what if our Fancy Fiesta Molecule is something a little more… directional? What if we don't want the hydrogens on the same side? What if we want them on opposite sides, creating a trans-alkene? Well, Lindlar's catalyst is a bit of a stickler for its cis-rule. If you want to go for the trans-isomer, you need a different approach. This is where things get even more interesting, and frankly, a little bit like a chemical daredevil act.
To make a trans-alkene from an alkyne, you need a way to add two hydrogens, but make them come in from opposite sides. This is achieved using a method called dissolving metal reduction. Don't let the name scare you; it's not as dramatic as it sounds, although it does involve some rather energetic chemicals. We're going to use sodium metal (which is notoriously reactive and likes to fizz and pop when it meets water, so we're definitely not playing with this in the bathtub) dissolved in liquid ammonia. It's like giving our alkyne a chemical spa treatment with a very intense, very cold bath.
Here’s the mind-blowing part: liquid ammonia is super cold, around -33 degrees Celsius. So, you’ve got your 1-butyne, you’ve got your super-reactive sodium, and you’ve got your incredibly frigid liquid ammonia. The sodium metal dissolves in the ammonia, creating these cool, blue-colored solvated electrons. These electrons are the real stars of the show. They are super keen to react. They attack the triple bond of our 1-butyne.
The process is pretty neat. An electron attacks one carbon of the triple bond, turning it into a single bond and forming a carbanion. Then, the carbanion picks up a hydrogen atom from the ammonia. This happens again, but the electrons and protons are added in a way that favors the trans configuration. It's like the molecules are dancing, and the music (or the electrons and protons) encourages them to take opposing stances. The result? A trans-alkene! Specifically, if we start with 1-butyne and do this, we'd get (E)-2-butene. The "E" comes from the German "entgegen," meaning "opposite." So, our caterpillar has decided to sport a cool, asymmetrical pair of sunglasses.
So, to recap our chemical adventure: We started with 1-butyne, a molecule with a very enthusiastic triple bond. We learned that to tame this beast and get closer to our (still imaginary) Fancy Fiesta Molecule, we can add hydrogen. If we want a relaxed, same-side double bond (a cis-alkene), we use Lindlar's catalyst. It's a gentle but firm approach, like a skilled minder. But if we want our Fancy Fiesta Molecule to have a more rebellious, opposite-side double bond (a trans-alkene), we bring out the big guns: sodium metal in liquid ammonia. This is a more dramatic, high-energy reaction that guarantees the trans configuration.
Both of these reactions take our 1-butyne and turn it into a 2-butene isomer. The key difference lies in the stereochemistry – how the atoms are arranged in 3D space. It's like choosing between a perfectly symmetrical handshake and a cool, angled high-five. Both are forms of connection, but they look and feel different. And that, my friends, is the beauty of organic chemistry. With a few well-chosen reagents and a bit of chemical finesse, we can coax molecules into doing our bidding, transforming them from one form to another, all while adding a sprinkle of humor and a dash of surprising facts along the way. Cheers to the molecular makeovers!
