Select The True Statements About Protein Secondary Structure.

Okay, so let's talk about protein secondary structure. You know, those wiggly, folded-up bits inside your body. It’s a bit like trying to organize a pile of spaghetti, except way more important for, you know, life. We’re going to play a little game here. Some of these statements are true, some are… well, let’s just say they’re trying their best. Your mission, should you choose to accept it, is to pick out the ones that actually hold water. Think of it as a protein anatomy pop quiz, but with less pressure and more giggles.
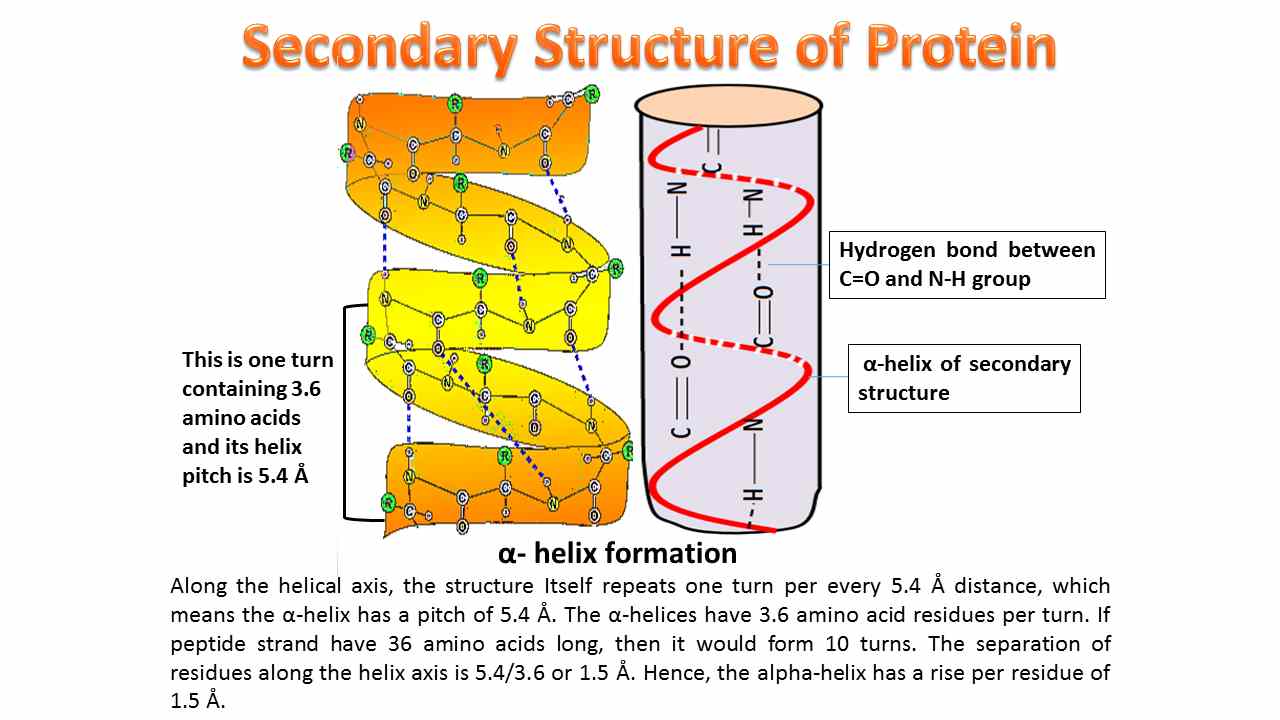
First up, we have the almighty alpha-helix. It’s probably the most famous one in the secondary structure club. Imagine a spring, or a curly fry. That’s pretty much the vibe. It’s a common way proteins like to twist themselves into shape.
Then there’s the beta-sheet. This one’s a bit more… organized. Think of corrugated cardboard, or maybe a neatly folded fan. It’s made of strands lying next to each other, all buddy-buddy.
Now, let's dive into the nitty-gritty of what makes these structures tick. It all comes down to those sneaky little hydrogen bonds. These are like tiny, invisible magnets holding the protein’s backbone together. Without them, our proteins would just… go their own way. And that would be a disaster, trust me.
The Alpha-Helix: More Than Just a Pretty Curl
So, the alpha-helix. It’s like the rockstar of secondary structures. It’s a coiled shape, and it’s super stable. This stability comes from those aforementioned hydrogen bonds.
Specifically, these bonds form between the carbonyl oxygen of one amino acid and the amide nitrogen of another amino acid. It’s a very specific dance they do, repeating every few amino acids. This precise bonding is what keeps the helix nice and tight.
Fun fact: Not all amino acids are equally thrilled about being in an alpha-helix. Some, like proline, are actually considered “helix breakers.” They’re a bit too rigid, or they throw a wrench in the whole hydrogen bonding mechanism. Poor proline, always the outcast.

The Beta-Sheet: The Organized Ensemble
Moving on to the beta-sheet. This structure isn’t just one strand doing its own thing. It’s made up of multiple beta-strands that lie side-by-side. These strands can be running in the same direction (parallel) or in opposite directions (antiparallel).
The magic here, again, is hydrogen bonding. But instead of happening within a single coil, these bonds form between the different beta-strands. It’s like a whole choir singing in harmony, holding each other up.
These sheets can be flat, or they can twist a bit. They can form large, flat surfaces or more complex, folded structures. It’s all about how those strands line up and bond.
The Unsung Heroes: Turns and Loops
While the alpha-helix and beta-sheet get all the glory, we can’t forget about the turns and loops. These are the connectors, the bit where the protein changes direction. They’re less structured, more flexible, but absolutely crucial.
Turns are usually short and abrupt. They allow the polypeptide chain to reverse direction quickly. Think of a sharp U-turn on a highway. They’re often stabilized by specific amino acids, like glycine and proline, who are surprisingly good at this sharp bending business.

Loops are longer and more varied. They can be quite flexible and often play a role in how proteins interact with other molecules. They’re the awkward dancers at the party, but sometimes they’re the ones who start all the interesting conversations.
Putting it All Together: The Grand Design
So, to recap the potentially true statements. We’re talking about structures that are held together by hydrogen bonds. These bonds are the glue that keeps our protein from unraveling into a meaningless blob of amino acids.
The alpha-helix is a coiled structure, like a spring. The beta-sheet is made of parallel or antiparallel strands, forming a more flattened or pleated structure. And we have the essential turns and loops that connect these more rigid elements.
It’s a beautiful, intricate system. Proteins aren’t just random strings; they have these specific, repeating shapes that are vital for their function. Whether they’re acting as enzymes, structural components, or signaling molecules, their secondary structure is the first step in getting there.

The Truth is Out There (Somewhere): Let’s See What You’ve Got!
Now, here’s where you come in. You’ve absorbed all this information, or at least some of it. Time to test your newfound knowledge. Which of the following statements rings true? Which ones are just… protein propaganda?
Is it true that all proteins are made up of only alpha-helices? (Spoiler alert: Probably not, unless you enjoy very predictable hair scrunchies.)
Is it true that beta-sheets are always perfectly flat and never have any twists? (Imagine trying to fold a piece of paper without any creases – a bit unrealistic, right?)
Is it true that hydrogen bonds are the primary force stabilizing both alpha-helices and beta-sheets? (This one feels like a strong contender, doesn't it? Like, really strong.)
Is it true that loops and turns are never involved in protein function and are simply filler? (If that were true, why would nature bother making them so specifically? Seems a bit wasteful.)
Is it true that proline is a common amino acid found in the core of alpha-helices? (Remember our conversation about helix breakers? This might be a trick question.)
Is it true that antiparallel beta-sheets have strands running in opposite directions and are often more stable than parallel ones? (The directionality thing seems important, doesn't it? And stability is always a plus.)
Is it true that the secondary structure refers to the overall 3D shape of a protein, including all its folded domains? (This sounds a bit… grander than just the local coiling and folding, doesn't it? Like confusing a single brick with the entire house.)
Is it true that the specific sequence of amino acids in a protein dictates which secondary structures it can form? (Think about it: the building blocks themselves must have a say in how they're arranged, right? Like LEGOs.)
So, there you have it. A whirlwind tour of protein secondary structures. Hopefully, you’re feeling a bit more enlightened, a bit more amused, and a bit more confident in spotting the actual truths amidst the protein hearsay. It's a wild world in there, but someone's got to keep it organized!
