Select The True Statements About Endergonic Reactions.

Hey there, science curious pals! Ever feel like your brain is playing a little game of "spot the fact"? Well, today we're diving into something super cool, a little bit quirky, and totally fascinating: endergonic reactions! Don't let the fancy name scare you. Think of it like this: we're playing a game of "Select The True Statements About Endergonic Reactions." It's all about figuring out what’s what with these energetic little guys.
So, what are endergonic reactions anyway? Imagine trying to push a boulder uphill. Yeah, that's kind of the vibe. They're reactions that need a little nudge, a boost, a whole lot of energy input to get going. They don't just happen on their own, like a spontaneous sneeze. Nope, these guys require some serious elbow grease. Or, you know, chemical elbow grease.
Let's get this truth-seeking party started! Are you ready to separate the facts from the fiction? Grab your metaphorical magnifying glass and let’s explore some statements. See if you can be the ultimate endergonic reaction detective!
Statement 1: Endergonic reactions release energy.
Okay, deep breaths. What do you think? Do they dump energy out into the world like a confetti cannon? Or do they hoard it like a dragon with its gold? Think back to that uphill boulder. Does pushing it uphill give you energy? Absolutely not! It takes energy from you, right? So, if this statement claims they release energy, that sounds a little… off, wouldn't you say?
This is where the fun begins! It’s like a tiny brain teaser. We're looking for the true statements. If a statement sounds like it's describing the opposite of what we know about these energy-craving reactions, then that statement is probably… well, not true. It's a good thing we're just selecting the true ones, not trying to invent them!

Statement 2: Endergonic reactions require energy input.
Alright, let's chew on this one. "Require energy input." Does that sound familiar? Does it echo the idea of pushing that boulder uphill? When you’re pushing that boulder, you are definitely putting energy in, right? You're sweating, you're straining, you're putting in your all. So, if an endergonic reaction requires energy input, that sounds pretty darn plausible. It's like a hungry tummy that needs food to get going.
This is the kind of statement that makes you nod your head. "Yep, that makes sense!" It aligns with the core idea that these reactions aren't going to just chill and happen. They need a little spark, a little jolt, a little oomph to get moving. Think of it as paying for a service. Endergonic reactions are the ones that make you pay up front to get the job done.
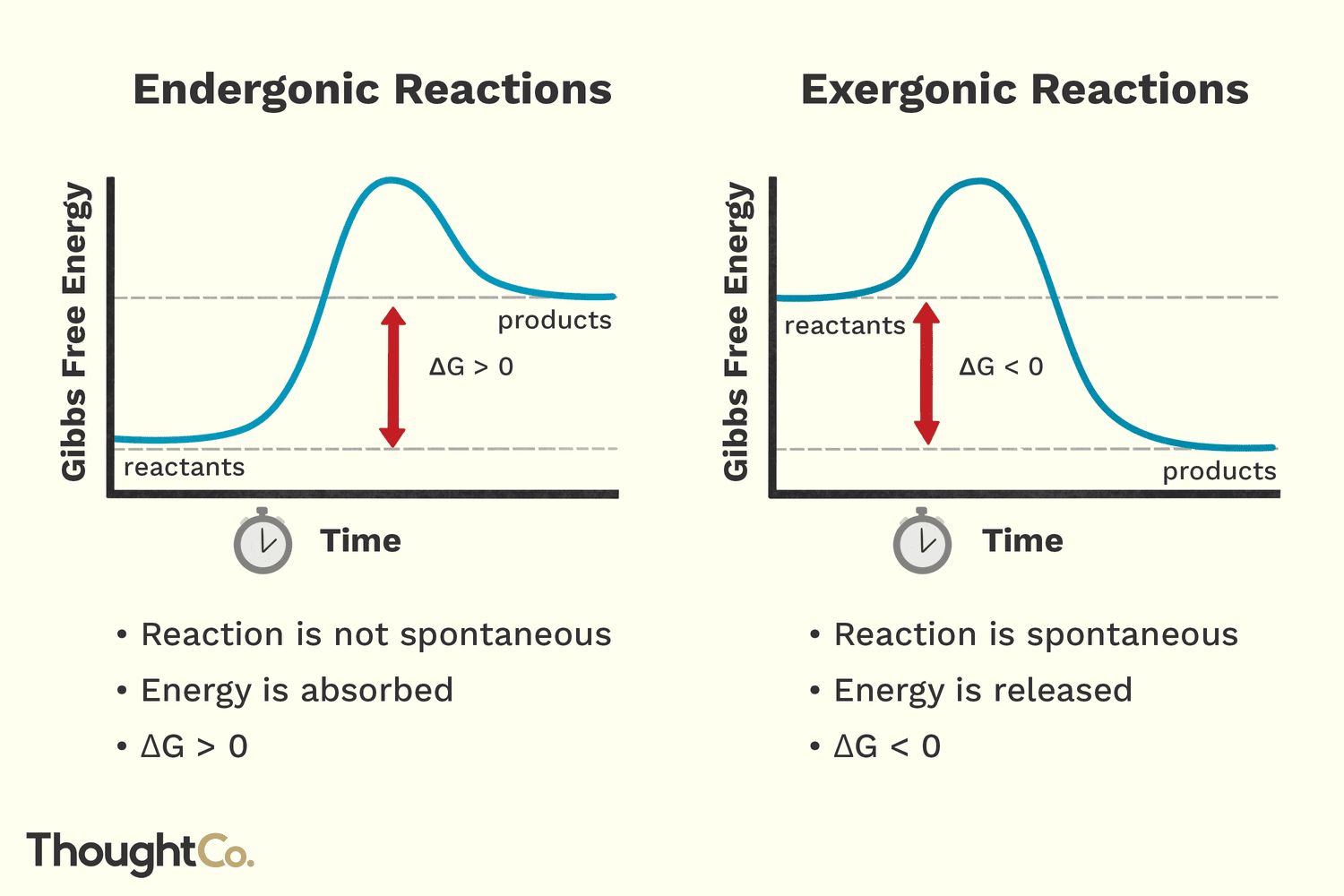
Statement 3: The change in Gibbs Free Energy (ΔG) for endergonic reactions is negative.
Whoa there, science nerds! Gibbs Free Energy. Sounds fancy, right? But let's break it down. This ΔG thing is basically a measure of whether a reaction will happen spontaneously. Think of it as the "will-it-or-won't-it" meter. If ΔG is negative, it means the reaction wants to happen, it's like it's ready to roll down a hill. Easy peasy.

Now, endergonic reactions are the ones we've been talking about that need that energy push. They don't want to happen spontaneously. They’re the ones resisting the downhill slide. So, if ΔG is negative for a spontaneous reaction, what would you guess it is for a non-spontaneous, energy-requiring reaction? It's likely the opposite, isn't it? So, if this statement says ΔG is negative for endergonic reactions, it’s probably trying to pull a fast one.
Statement 4: Endergonic reactions are non-spontaneous.
Let's circle back to our boulder friend. Is pushing a boulder uphill a spontaneous activity? Do you just wake up one morning and think, "You know what? I’m going to push this giant rock up a mountain today without any reason!" Probably not. It takes effort. It's not something that just happens. So, if endergonic reactions are like pushing that boulder, are they spontaneous? Or are they the opposite?
This is where the playful deduction comes in. If they require energy input, if they’re not just going to happen willy-nilly, then the word that fits them best is non-spontaneous. They’re the hesitant ones, the ones that need a little convincing. This statement feels like a strong contender for a true statement, wouldn't you agree? It’s a key characteristic, like a nickname that perfectly describes someone.
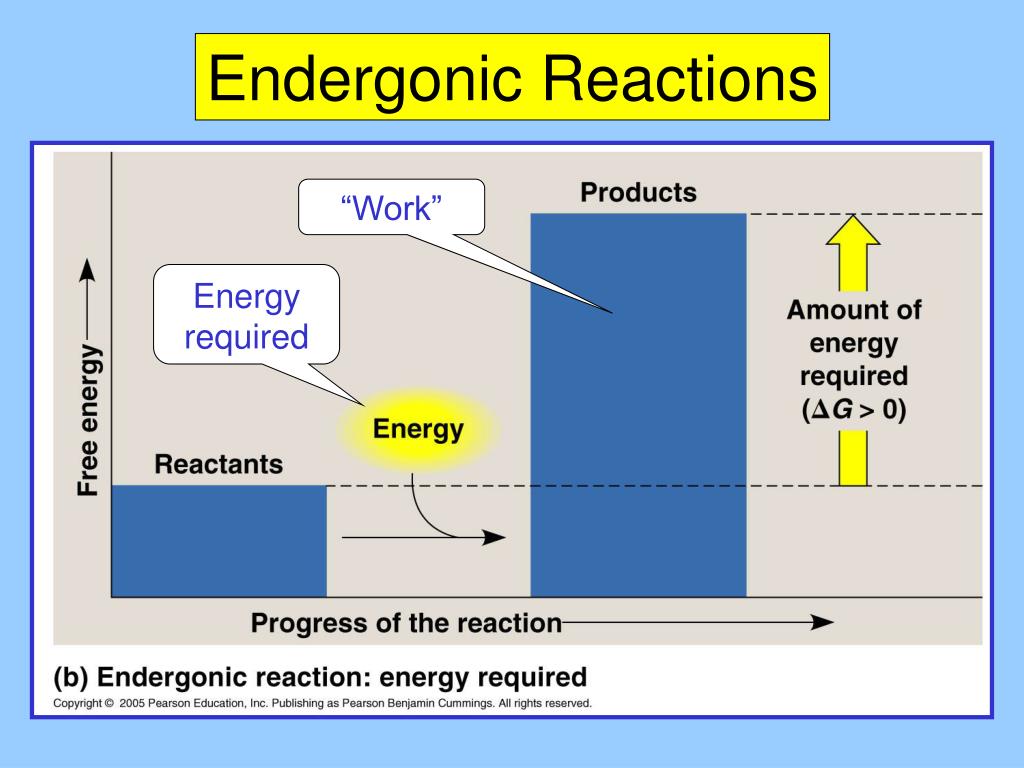
Statement 5: Endergonic reactions are coupled with exergonic reactions to proceed.
Okay, this is where things get really interesting, and honestly, a little bit like a superhero team-up! Sometimes, these energy-requiring endergonic reactions are like the sidekicks. They can't do it all on their own. But, if they team up with a reaction that does have a lot of energy to spare (that’s an exergonic reaction, the opposite kind!), then they can get the job done!
It’s like a powerful duo. The exergonic reaction provides the energy boost, the "oomph," and the endergonic reaction uses that borrowed energy to do its thing. They're coupled – linked together. One gives, and the other takes. This statement, talking about being coupled with exergonic reactions, sounds like a legitimate way these non-spontaneous reactions manage to happen in the real world. It’s a clever workaround!
Statement 6: The products of an endergonic reaction have less free energy than the reactants.
Let’s think about what "free energy" means in this context. It's kind of like the potential energy stored in the molecules. If a reaction is endergonic, remember, it takes energy in. So, if you're adding energy, where does that energy go? Does it just vanish? Or does it get stored somewhere?
Imagine you're building something complicated. You're putting in energy (effort, materials), and the final structure (the products) is going to be more complex, more "built up," and have more stored potential than the original pile of bricks and dust (the reactants). So, if endergonic reactions take in energy, it's likely that the products are going to end up with more stored energy, not less. Therefore, this statement claiming the products have less free energy sounds like it's telling us a fib.
So, what are the TRUE statements?
Let's tally 'em up! We're looking for the ones that truly describe our energy-hungry endergonic reactions. Based on our fun little detective work:
- Statement 2: Endergonic reactions require energy input. (Absolutely! They're the ones who need a handout.)
- Statement 4: Endergonic reactions are non-spontaneous. (Yep! They don't just happen by themselves.)
- Statement 5: Endergonic reactions are coupled with exergonic reactions to proceed. (This is a super common and important way they get their energy!)
Isn't that neat? We just played a game and learned some awesome stuff about how the chemical world works. These endergonic reactions might be a bit of a handful, but they're crucial for so many biological processes. Think about building muscles, or even just thinking! It all involves these energy-investing reactions. Keep that curiosity buzzing, and who knows what other scientific mysteries you'll solve!
