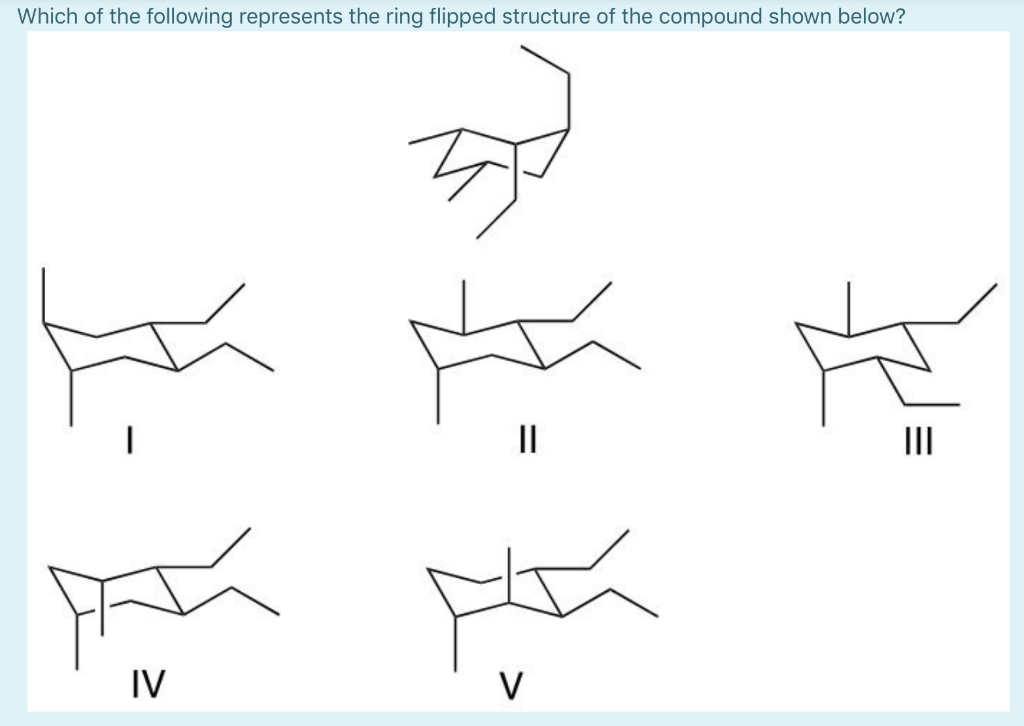
Select The Ring Flip For The Following Compound:
Okay, so you know how sometimes molecules can be, like, a bit dramatic? They’ve got these fancy shapes and they can even wiggle their way into different forms. It’s kind of like when you’re trying to get comfortable on the couch and you do that whole twisty-turny thing. Well, in the wild world of chemistry, this is called a ring flip. And today, we're diving into how to pick the right one for a super common molecule.
Imagine you've got a ring. A simple, six-membered ring, like cyclohexane. Pretty chill, right? But here's the kicker: it's not stuck in one flat shape. Nope. It can actually puff out and flatten a bit, then puff out the other way. It's like a little chemical limbo. This is its super-secret superpower: it can flip!
Think of it like this: one minute, your ring is chilling in what we call a “chair” conformation. It looks a bit like a beach chair, all comfy and reclined. Then, poof! It does a full 180 and becomes another chair conformation. Mind. Blown.
Now, why is this a big deal? Because where things attach to this ring matters. Are your little sticky-out bits (we call these substituents) pointing straight up or straight out? This is where the fun really begins. Let's say you have a cyclohexane ring with a little methyl group – that's just a tiny carbon chain, like a little accessory. We can draw it in two different chair forms, and the methyl group can be in two different positions: axial or equatorial.
Axial positions are like the upright poles of the chair. They're straight up and down. Equatorial positions are more like the arms of the chair, sticking out sideways. They're more on the "level." Get it? Like equator? Little geography joke for ya!
So, when our cyclohexane ring does its big flip, what happens to our methyl group? If it was in an axial position in the first chair, it becomes an axial position in the second chair. But here's the quirky bit: it flips to the other side of the ring. And if it was equatorial? You guessed it – it stays equatorial, just on the opposite side.

This is where the "select the ring flip" part comes in. We're not just talking about any ring flip. We’re talking about the preferred ring flip. The one that makes the molecule the happiest. And how do we know what makes a molecule happy? Well, it's all about energy. Molecules, just like us, want to be in the lowest energy state possible. They want to be comfy, not stressed out.
Imagine you're trying to carry a bunch of groceries. Would you rather hold them all really close to your body, or have them sticking way out, making you feel all unbalanced and wobbly? Most of us would go for the closer, more balanced option, right? It just feels better. That's the same idea with our ring flip.
When we have substituents on our cyclohexane ring, those axial positions can be a little…awkward. Think of them as being cramped. They often bump into other parts of the ring. This bumping causes what we call steric strain. It's like trying to squeeze into a tiny airplane seat with your knees hitting the seat in front of you. Not ideal.

Equatorial positions, on the other hand, are much more spacious. Your substituents have plenty of room to stretch out. Less bumping, less strain, less stress. Lower energy! So, molecules tend to prefer the conformation where their bulky substituents are in the equatorial positions.
Let’s take our methyl-substituted cyclohexane again. If the methyl group is in an axial position, it’s kind of crowded. It’s like having a big backpack on in a crowded subway car. But when the ring flips and the methyl group becomes equatorial, it’s like putting that backpack down on the floor. So much more chill.
Therefore, the preferred conformation is usually the one with the largest substituent in the equatorial position. This is the "happier," lower-energy arrangement. We can actually assign relative energies to these conformations. It’s like giving each possible shape a score, and the lowest score wins!

So, when you’re asked to "select the ring flip," what you're really doing is looking at your molecule and figuring out which chair conformation is the most stable. Which one has the least amount of that pesky steric strain. It’s like a molecular beauty contest, and stability is the winning criteria.
It's not just about picking a drawing. It's about understanding the forces at play. It's about how atoms interact and how they try to minimize their discomfort. It’s a little peek into the secret lives of molecules!
And here’s a super fun tidbit: the bigger the substituent, the more it hates being in an axial position. A tiny hydrogen atom doesn't mind too much. But a giant tert-butyl group? Oh boy, it really wants to be equatorial. It’s like trying to fit a giant inflatable flamingo into a small shoebox. It just won't work out well!

So, to pick the right ring flip, you just need to: 1. Draw your cyclohexane ring in one chair conformation. 2. Place your substituents in their axial and equatorial positions. 3. Imagine the ring flipping. Where do your substituents go? 4. Now, the magic question: which conformation has the bulkier substituent in the equatorial position? That's usually your winner! That's the one you want to highlight.
It’s like a detective puzzle, but with atoms. You’re looking for clues – the size of the groups, their positions – to figure out the most likely scenario. And the best part? This isn't just some abstract concept. This actually affects how molecules react. The shape of a molecule is super important for how it fits into other molecules and causes chemical reactions. So, this seemingly simple ring flip has big implications!
It's a fun way to think about how even the tiniest changes in shape can have a huge impact. It’s a little reminder that the world of molecules is always in motion, always adapting, and always seeking its most comfortable state. So next time you see a cyclohexane, give it a little mental ring flip! See if you can guess which conformation it’s chilling in. It’s a neat little trick that makes chemistry just a bit more… animate.
