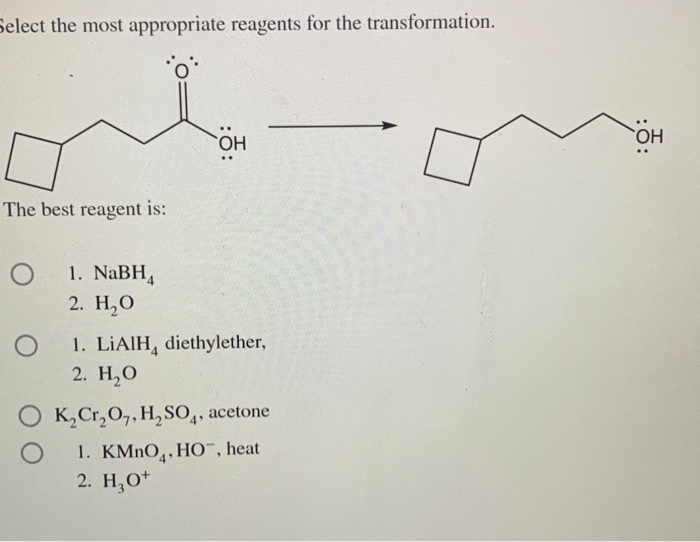
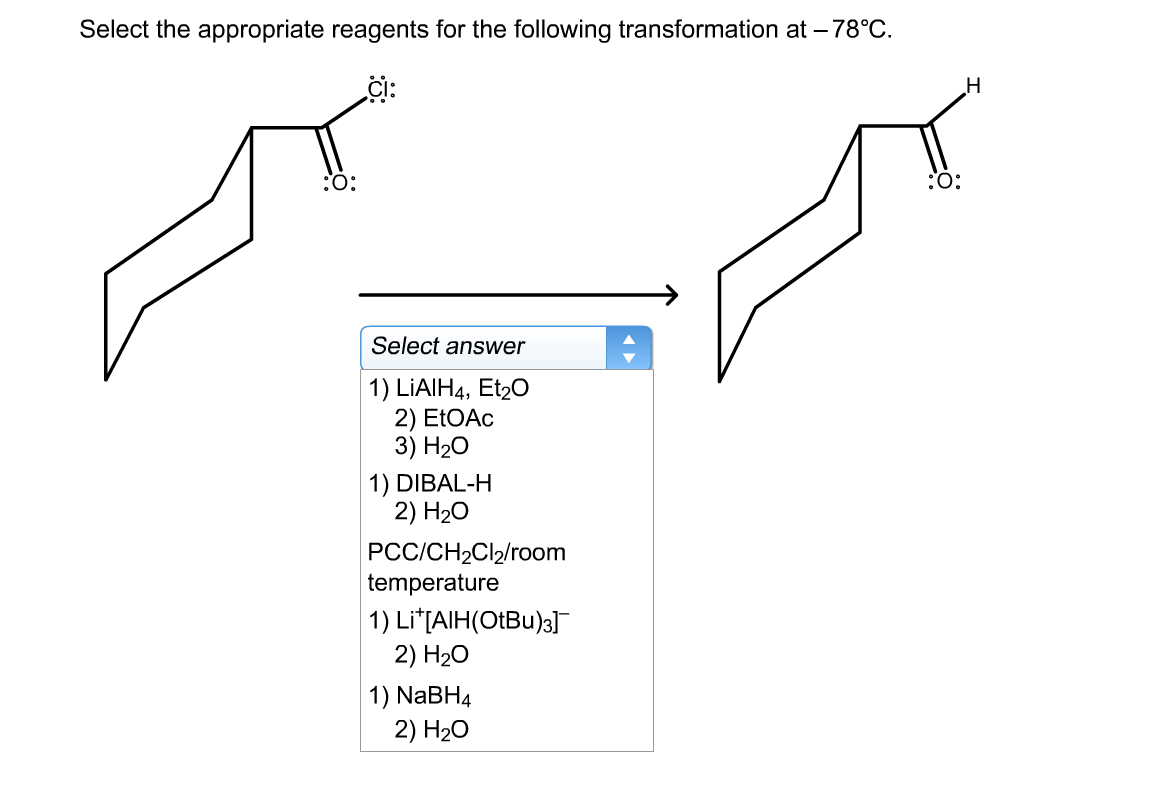
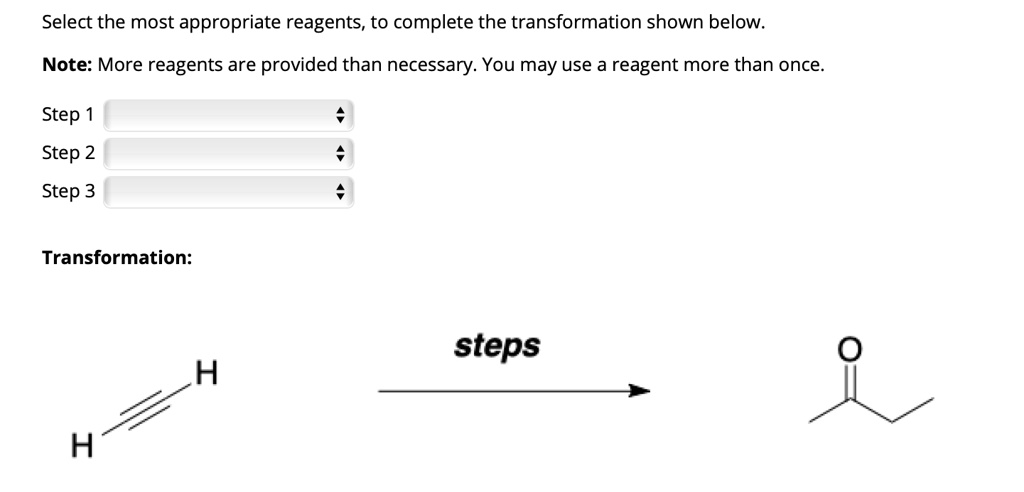
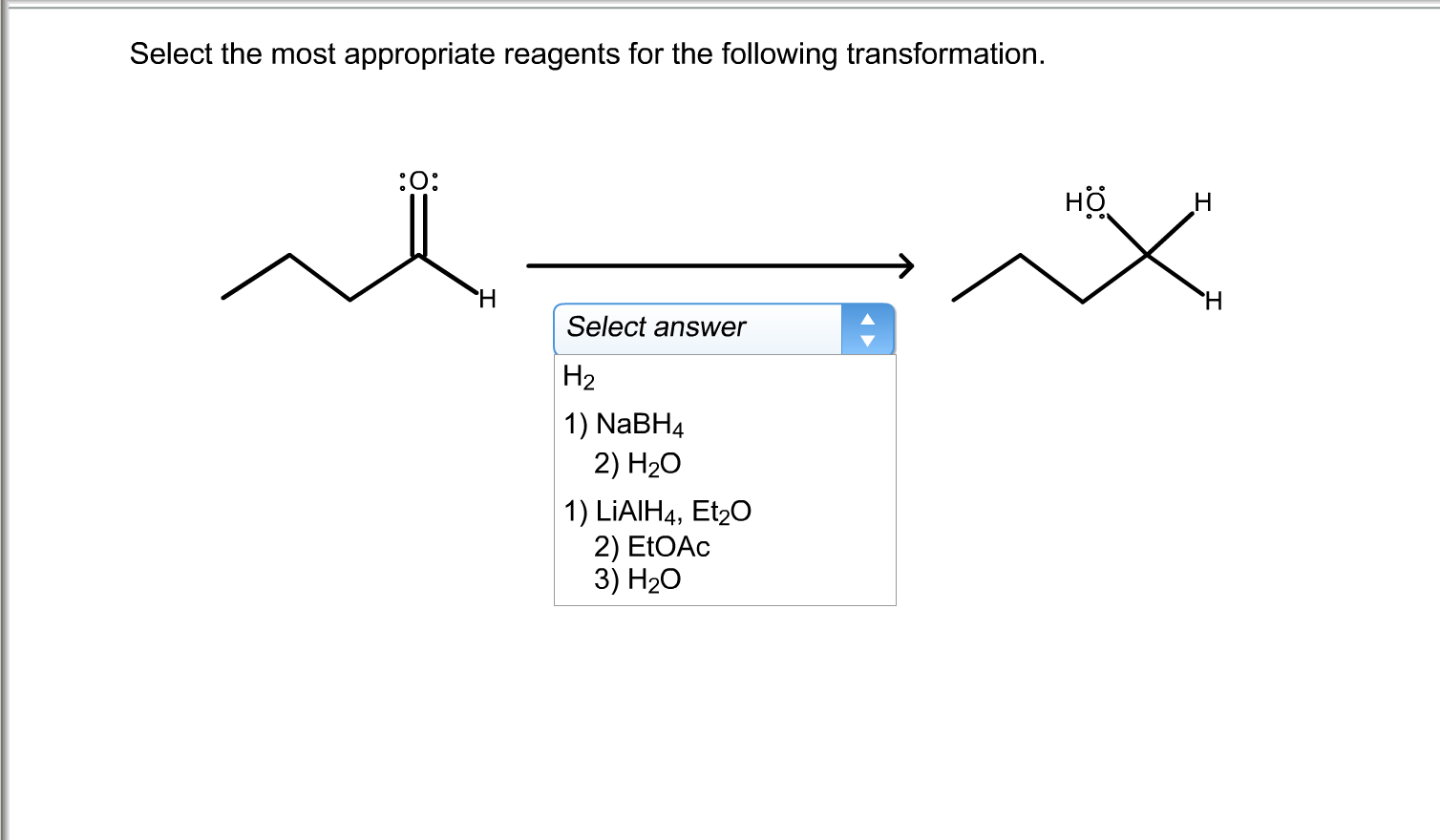
Select The Most Appropriate Reagents For The Transformation.
Alright, gather 'round, folks! Pull up a chair, grab your latte, and let's talk about something that sounds super exciting, I know. We're diving into the glamorous world of... choosing the right reagents. Now, before you picture me in a lab coat, goggles askew, wrestling with a bubbling beaker (which, let's be honest, happens more often than I'd like to admit), this is actually less about mad science and more about being a really, really good chef.
Think of it like this: you want to make a killer lasagna, right? You wouldn't grab a bag of cement and a tub of mayonnaise, would you? No! You need the right ingredients. Tomatoes that have actually seen the sun, pasta that’s just the right texture, and cheese that can pull a string for miles. Chemistry is kinda the same, but instead of pasta, we're messing with molecules, and instead of cheese pulls, we're aiming for molecular transformations.
So, what exactly is a "reagent"? In the grand, slightly dusty tome of chemistry, a reagent is basically your magic ingredient. It's the stuff you throw into the pot to make something happen. It's the catalyst, the starter, the spark plug. Without the right reagent, your molecule will just sit there, looking all shy and unreactive, like a teenager at a family reunion. We want our molecules to be party animals, folks!
The whole point of this fancy-pants "selection process" is to get your starting material (let's call it 'Bob') to transform into your desired product (let's call it 'Awesome Sauce'). And there are millions of Bob's out there. Some are simple, some are complex, and some are so unwieldy they probably need their own zip code. Similarly, there are more reagents than there are questionable life choices we've all made after 2 am. The trick is to find the perfect match.
Imagine you've got a molecule that's a bit like a grumpy old man. He’s got a stubborn bond that needs breaking, like his refusal to learn how to use a smartphone. You could try yelling at it (that's like using a really harsh acid, which might work, but it's going to cause a lot of collateral damage and frankly, it's just rude). Or, you could try a more subtle approach. Maybe a gentle persuasion? That's where the art of reagent selection comes in.
We're not just picking things out of a hat, you know. There are entire libraries dedicated to this stuff! It's like having a dating app for molecules, except instead of swiping left or right, you're looking at reactivity, selectivity, and yield. And trust me, a bad match here can lead to a chemical breakup that's more dramatic than a reality TV show finale. You end up with a mess of unwanted byproducts, a disappointed scientist, and a budget that's crying into its ramen.
The Art of Not Blowing Stuff Up (Mostly)
One of the biggest considerations when picking a reagent is safety. Nobody wants to be the chemist who accidentally invents a new shade of black (that's the color of everything after a fiery mishap). So, we ask ourselves: Is this reagent going to explode if I look at it funny? Does it require a hazmat suit and a bomb disposal unit just to open the bottle? If the answer is yes, we might want to explore other options, unless we're aiming for a career in Hollywood special effects.
Then there's selectivity. This is like trying to pickpocket a single coin from a very crowded, very chaotic pocket. You want your reagent to go after exactly what you want it to, and leave everything else alone. Imagine trying to remove a single Lego brick from a giant Lego castle without knocking the whole thing down. If your reagent is too aggressive, it might strip the paint off your molecule, or break off other important bits. We want precision, not a wrecking ball!
And let's not forget yield. This is basically how much of your 'Awesome Sauce' you actually get at the end. You could have a reagent that works perfectly and selectively, but if it only turns 1% of your 'Bob' into 'Awesome Sauce', well, that's just a sad day for everyone involved. We want our reagents to be efficient, to give us a good return on our molecular investment. It's like ordering a pizza and only getting one slice – highly unsatisfactory.
When Simple Isn't Enough
Sometimes, you’re faced with a molecule that’s a real diva. It's got all sorts of sensitive bits that can get messed up easily. Think of it like trying to iron a delicate silk shirt with a scorching hot iron – disaster waiting to happen. In these cases, you need mild reagents. These are the gentle souls of the reagent world, the ones that whisper sweet nothings to the molecule and coax it into changing its mind, rather than forcing it.
For instance, if you need to remove a hydrogen atom from a molecule (don't ask me why, sometimes the reasons are as bizarre as the molecules themselves), you wouldn't use a sledgehammer. You might use something like... well, let's just say a reagent that’s way less dramatic. It's like asking a ninja to pluck a single hair versus sending in a squadron of stormtroopers. The ninja, my friends, is your mild reagent.

On the flip side, some molecules are like Fort Knox. They're tough, they're stubborn, and they need something with a bit more oomph. This is where you might need stronger reagents. These are the drill-sergeants of the chemical world. They're not afraid to get their hands dirty and force a reaction to happen. But again, this comes with risks. Too much oomph, and you’ve just turned your valuable molecule into a pile of atomic dust. It's a fine line, a delicate dance between power and destruction.
And what about things that aren't quite reagents themselves, but play a crucial role? We're talking about catalysts! These are the backstage crew of our molecular theatre. They help the reaction happen, they speed things up, but they don't actually get used up themselves. They’re like that friend who always organizes the party – they make it happen, but they don’t drink all the punch. Enzymes in our bodies? Pure genius catalysts. Without them, we’d be moving slower than a sloth on tranquilizers.
So, the next time you see a chemical reaction happening, whether it's in a textbook or a dramatic movie scene (where they always make things explode way too easily), remember the unsung heroes: the reagents. They’re the quiet, or sometimes not-so-quiet, workhorses that make the magic happen. And choosing the right one is a blend of science, intuition, and a healthy dose of "please don't blow up my lab." Cheers to that!
