Select The Most Acidic Hydrogen In The Molecule Below

Imagine a molecule as a tiny, bustling community. Each atom is like a resident, and the hydrogens are the little ones, always a bit more energetic and eager to join in the fun.
Sometimes, in this molecular neighborhood, one particular hydrogen atom feels a bit more outgoing than the others. It's like the kid who’s always the first to shout "Race you to the corner!"
This "outgoing" hydrogen is what we call the most acidic. It’s the one most ready to pack its bags and go on an adventure, usually with a little help from a friend.
Let's peek at our special molecule and see if we can spot this adventurous hydrogen. It’s a bit like a game of "Where's Waldo?" but with atoms!
Think of the molecule as a little house. Some parts of the house are cozy and stable, while others are a bit more exposed, like a window that’s always slightly ajar.
The hydrogen atoms are like little hands reaching out from the house. We're looking for the hand that's most likely to get a good grip and pull away.
In our molecule, there's a special arrangement of atoms that makes one hydrogen feel a bit more "special." It's like having a favorite toy that you just can't wait to play with.
This special feeling comes from the neighbors around it. Some neighbors are really good at sharing their energy, and that makes our hydrogen feel super charged!
One side of our molecule has a really kind and generous neighbor. This neighbor is called oxygen, and it's known for being a bit of a magnet for attention.
When hydrogen is near a super-attractive neighbor like oxygen, it starts to feel a little bit lighter, a little bit more ready to be picked up and taken somewhere new.
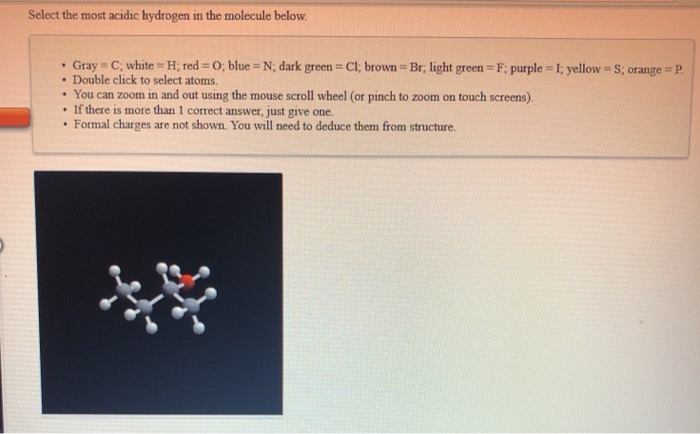
Think of it like a strong magnet pulling on a little paperclip. The closer the paperclip is, the stronger the pull!
Our molecule has a few different hydrogen atoms, each with their own little homes within the molecular structure. Some are tucked away, safe and sound.
But one hydrogen, in particular, is hanging out in a spot where it gets a lot of positive vibes. It's like being on the sunny side of the street!
This hydrogen feels a bit more exposed, a bit more ready for action. It's the one that's most likely to respond when a friendly chemical "hand" comes along.
We can tell which hydrogen is the most acidic by looking at what's surrounding it. It's like judging a book by its cover, but in a very scientific way.
If a hydrogen is next to a very electronegative atom, that's like having a super-strong friend who’s really good at encouraging you to try new things.
Electronegativity is just a fancy word for how much an atom likes to hog electrons. Oxygen is a big electron hog!

So, when our hydrogen is near a big electron hog like oxygen, it starts to feel a little bit depleted. It’s like giving away your favorite toy – you feel a little bit less attached.
This feeling of being a little less attached is what makes the hydrogen acidic. It's ready to say "See ya!" and move on.
In our specific molecule, there are a couple of places where hydrogen atoms live. We need to compare their living situations.
One group of hydrogens might be hanging out with a carbon atom. Carbon is a pretty chill neighbor, not too pushy.
But another hydrogen is hanging out with a very eager neighbor. This neighbor is none other than our beloved oxygen!
This oxygen neighbor is so good at attracting electrons that it pulls them away from the hydrogen. It's like a cosmic vacuum cleaner!
When the hydrogen loses some of its electron friends to oxygen, it starts to feel a bit positively charged. Think of it as feeling a bit lighter and more eager to find new friends.
This positively charged hydrogen is the one that’s most ready to be "donated" or taken away in a chemical reaction.

It’s the hydrogen that’s most likely to get picked up by a passing base, like a friendly taxi service for molecules.
So, when we look at our molecule, we're scanning for the hydrogen that has the most appealing gig. Who's got the best view?
We're looking for the hydrogen that's living next door to the molecule's biggest attention-seeker.
And in this case, the star of the show is the hydrogen that's cozied up to the oxygen atom.
This hydrogen has a special connection with its oxygen neighbor. They’re like a dynamic duo, but in a chemical way.
The oxygen is so good at pulling electrons that it makes this particular hydrogen feel… well, a bit vulnerable.
And that vulnerability makes it the most eager to leave the molecular party. It's the first one to grab its coat when it hears a good opportunity.
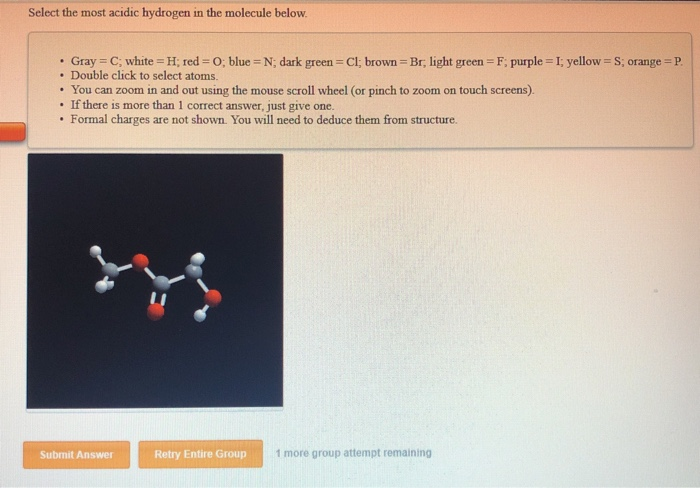
So, the most acidic hydrogen is the one that’s closest to the electron-hungry oxygen.
It’s like the hydrogen is saying, "Hey, this oxygen is pulling all the good vibes! I think I'll try my luck over there!"
This is why understanding these tiny molecular interactions is so cool. It’s all about who’s pulling whom, and who’s ready for an adventure!
Even in the smallest things, there are always those who are a little more ready to explore. And that's what makes chemistry so wonderfully dynamic.
It's a constant dance of attraction and detachment, all happening at a speed we can only imagine.
The acidic hydrogen is just a testament to the fact that some atoms are simply better at making friends – or at least, at being pulled towards new ones!
It's a heartwarming thought, isn't it? That even in the silent world of molecules, there's a sense of readiness for change and new experiences.
And that's the story of our adventurous, most acidic hydrogen!
