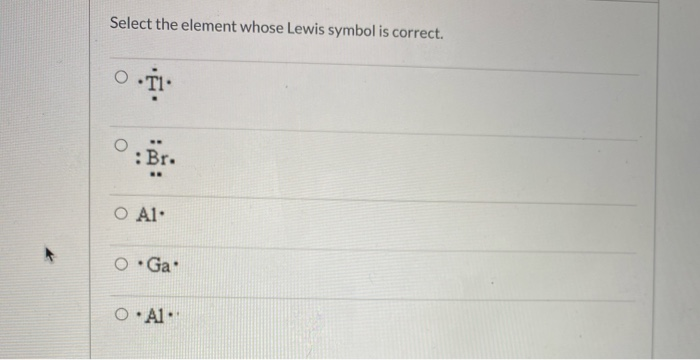
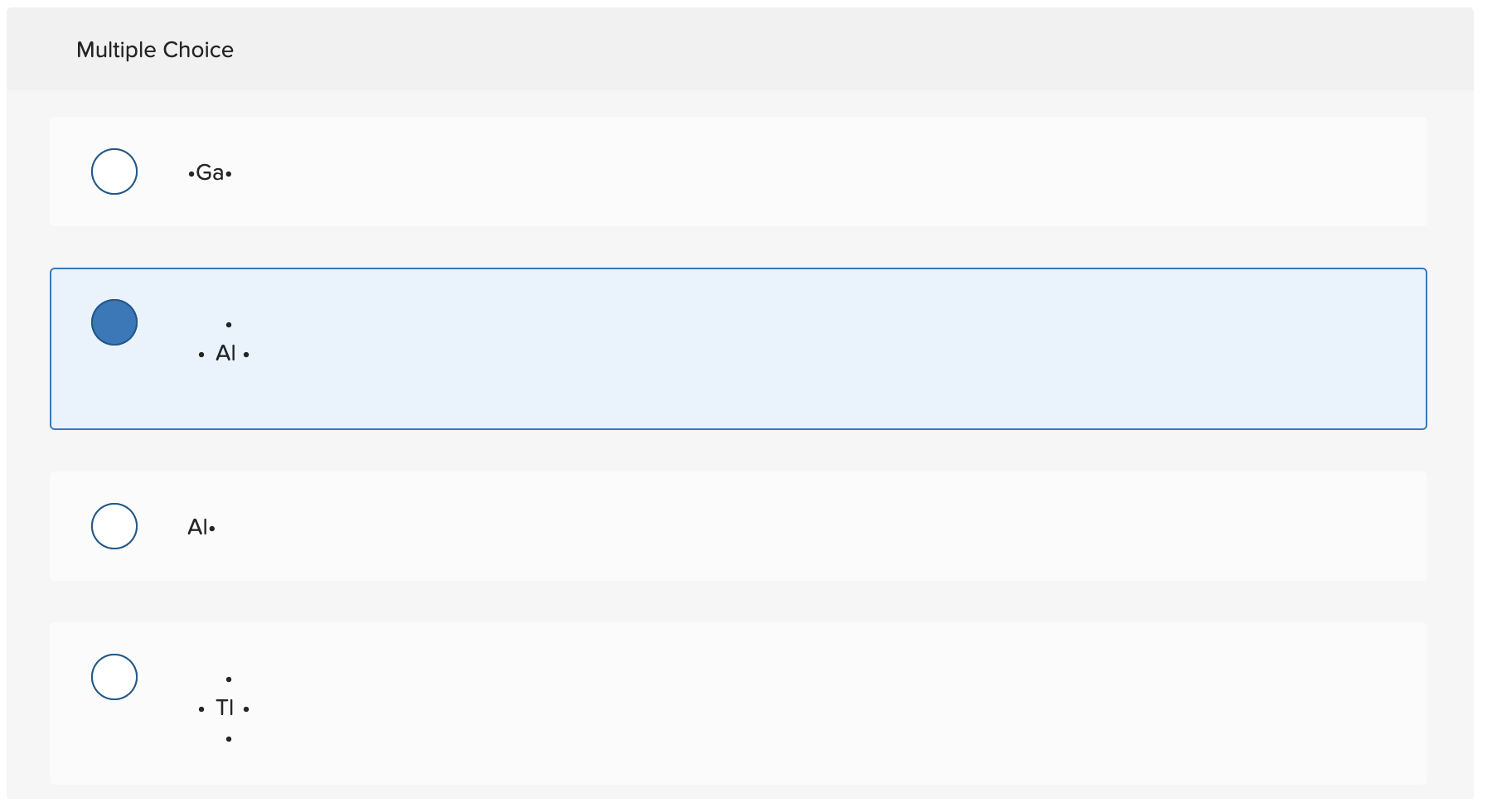
Select The Element Whose Lewis Symbol Is Correct

Alright, so imagine you're at a potluck, right? Everyone's brought their signature dish, and there's this one Jell-O mold that's just... spectacular. It's got all the fruits perfectly suspended, the wobble is just right, the color is chef's kiss. And then there's another Jell-O mold, and let's just say it looks like it gave up halfway through. Maybe the gelatin didn't set, maybe a rogue grape escaped and is doing its own thing. You just look at it and think, "Yeah, that's not quite hitting the mark, is it?"
Well, in the wacky world of chemistry, we've got something similar going on. It's called a Lewis symbol, and it's basically a tiny little drawing that shows how atoms like to hang out and share their electrons. Think of electrons as those little sprinkles on top of your favorite cupcake – they're the bits that get passed around, the bits that make things stick together. And the Lewis symbol is like a blueprint, showing you exactly which sprinkles are where and how they're arranged.
Now, just like with those potluck Jell-O molds, some Lewis symbols are spot-on, perfectly representing how an element actually behaves. They’re the showstoppers, the ones that make you go, "Ah, yes, that’s it!" And then, you’ve got the other ones. The ones that are… well, let’s just say they’re trying their best, but they’re a bit off. Like that Jell-O mold that’s more of a liquid puddle. They might look like they’re supposed to be Jell-O, but they’re just not quite there.
So, our mission, should we choose to accept it (and honestly, it’s not that dramatic), is to become super sleuths of the atomic world and select the element whose Lewis symbol is correct. It’s like being the judge at a Jell-O mold competition, but instead of tasting, we’re using our brains and a little bit of atomic know-how.
Let’s break down what makes a Lewis symbol so special. It’s named after a brilliant scientist, G.N. Lewis, who basically figured out that atoms are all about those outer electrons, the ones they use for… well, for everything! He said, "Let's represent the atom with its chemical symbol, and then let’s surround it with dots. Each dot is an electron in the outermost shell." Simple, right? Like drawing little smiley faces around your friends to show who’s got the most friends.
The key thing to remember is that atoms generally want to have a full outer shell of electrons. It’s like wanting a full set of matching LEGO bricks so your structure is super stable. They want to achieve what we call an octet, which means eight outer electrons. Some smaller atoms are happy with just two, like they’re content with just a small handful of sprinkles. It’s their own little version of atomic bliss.
So, when we look at a Lewis symbol, we’re looking for the element's symbol, and then dots around it. These dots represent those precious outer electrons. The number of dots has to match the number of valence electrons the atom actually has. This is the golden rule, the non-negotiable commandment of Lewis symbols. Mess this up, and your symbol is about as useful as a screen door on a submarine.

Let’s take an example. Imagine you see a symbol for Oxygen. Oxygen, on the periodic table, is in Group 16. That '6' is a big clue! It means Oxygen has six valence electrons. So, a correct Lewis symbol for Oxygen should have the symbol 'O' surrounded by six dots. These dots can be arranged in pairs or individually, but the total number must be six. Think of it like packing a lunchbox – you need to make sure you have the right number of sandwiches, not too many, not too few.
Now, what if you see an Oxygen symbol with, say, seven dots? Or maybe only four dots? Those are the equivalent of showing up to the potluck with just a single, sad carrot stick. It’s not wrong in the sense that it’s poisonous, but it’s definitely not what was expected, and it doesn’t quite fit the bill. The Lewis symbol is just wrong because it doesn't accurately reflect the atom’s electron count.
The periodic table is your best friend in this whole operation. It’s like a cheat sheet for the universe! The group number (for the main groups) often tells you directly how many valence electrons an element has. For instance, elements in Group 1 (like Sodium, Na) have 1 valence electron. So, their Lewis symbol is just 'Na' with one dot. Easy peasy, lemon squeezy. They’re like the minimalist of the atomic world, keeping things simple.
Elements in Group 2 (like Magnesium, Mg) have 2 valence electrons. So, you’d see 'Mg' with two dots. They’re a bit more enthusiastic with their sprinkles, but still keeping it contained. Then you jump over to Groups 13 through 18. For Group 13, it's 3 valence electrons. Group 14, 4 valence electrons. Group 15, 5 valence electrons. Group 16, 6 valence electrons. Group 17, 7 valence electrons. And finally, Group 18, the noble gases, are famously stable with 8 valence electrons (except for Helium, which is happy with 2, like a special case). These guys are the rockstars of stability, already having their full octet, so they don’t really need to share or borrow electrons. They're the folks who show up to the party already fully prepared and don't need anything from anyone.
So, when you're presented with a question like, "Select the element whose Lewis symbol is correct," you're essentially being asked to play detective. You’ll be given a few options, each with an element symbol and a set of dots. Your job is to:
- Identify the element: What element is it?
- Find its position on the periodic table: Where does it hang out with its atomic buddies?
- Determine its number of valence electrons: How many outer electrons does it actually have?
- Count the dots: How many dots are around the symbol in the given Lewis symbol?
- Compare! Does the number of dots match the number of valence electrons? If yes, BAM! You’ve found your winner. If no, move on to the next contender.
It’s like a game of "Spot the Difference," but with atoms. You’re looking for the one that’s exactly right, the one that tells the true story of that element’s electron situation.
Let’s imagine some scenarios. You might see:
Scenario A: Sodium (Na) with 2 dots.
Okay, Sodium is in Group 1. It has 1 valence electron. The symbol has 2 dots. Mismatch! This is like a recipe calling for 2 cups of flour and you only put in 1. The cake’s not gonna turn out right.
Scenario B: Chlorine (Cl) with 7 dots.
Chlorine is in Group 17. It has 7 valence electrons. The symbol has 7 dots. Match! Bingo! This is the Jell-O mold that’s absolutely perfect. It’s got the right wobble, the right fruit, the right everything.
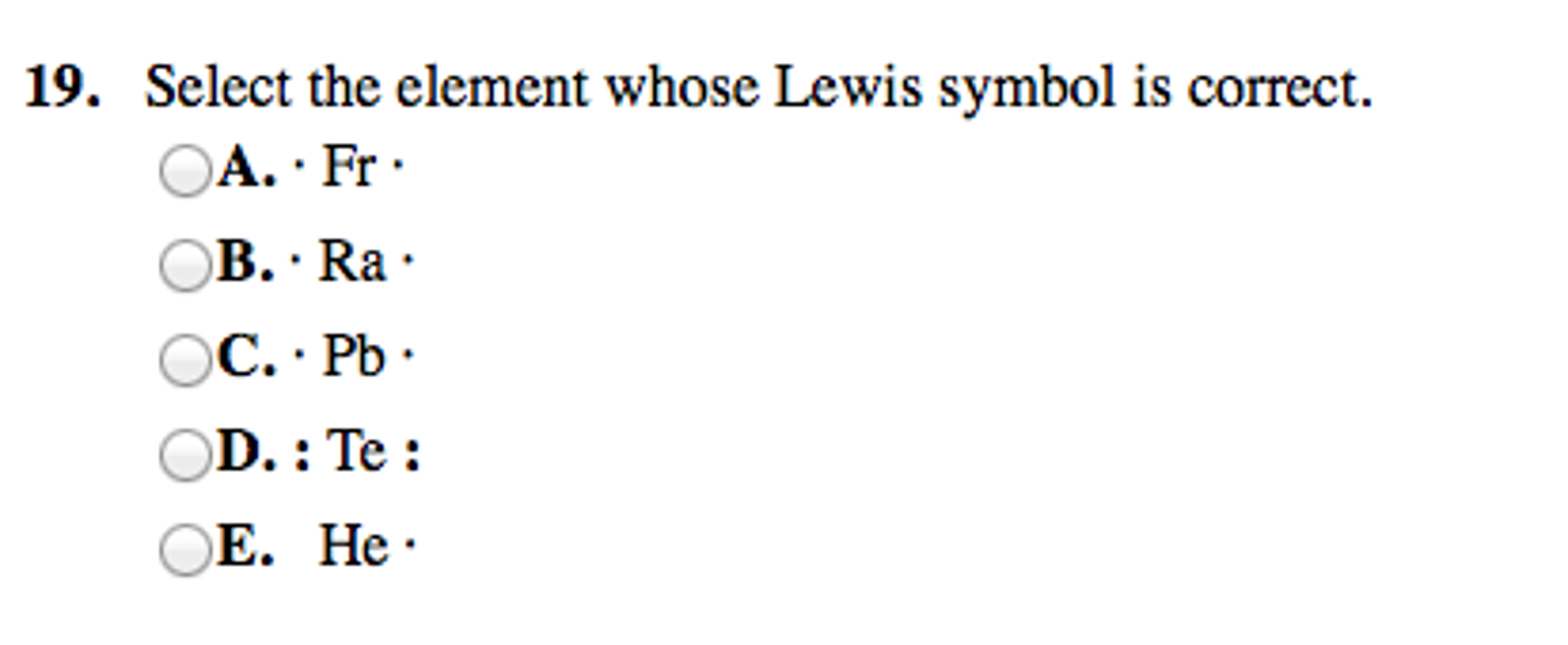
Scenario C: Carbon (C) with 3 dots.
Carbon is in Group 14. It has 4 valence electrons. The symbol has 3 dots. Mismatch! Not enough sprinkles for this cupcake. It’s missing a crucial element.
Scenario D: Neon (Ne) with 8 dots.
Neon is in Group 18. It has 8 valence electrons (it’s a noble gas, super stable!). The symbol has 8 dots. Match! Another winner. This is the element that’s already perfectly content with its electron arrangement.
The trick is to not get flustered. Take your time, consult your trusty periodic table, and do the simple comparison. It’s not about memorizing every single element’s electron configuration on the spot (though that comes with practice!). It’s about understanding the principle behind Lewis symbols and applying it.
Sometimes, you might see dots arranged in a specific way. For example, single dots might be shown before pairing up. This is because atoms often try to spread out their electrons as much as possible before they start pairing them up. Think of it like people getting on a bus – they’ll take individual seats before they start sitting next to strangers. But for the purpose of simply checking if the number of dots is correct, the arrangement is secondary. The primary check is the count.
It's important to remember that Lewis symbols are a simplified model. They're not the whole story of how atoms behave, but they're an incredibly useful way to visualize and understand bonding. They’re like the basic outline of a drawing – you can see what it’s supposed to be, even if it doesn’t have all the shading and detail yet.
So, when you’re faced with this task, just take a deep breath. Think of it as a little puzzle, a brain teaser. You’re the detective, the periodic table is your clue board, and the Lewis symbols are your suspects. You’re looking for the one that’s telling the truth about its valence electrons.
Don't be intimidated by fancy-sounding terms. "Valence electrons" just means the electrons on the outside, the ones that do the interacting. "Octet" just means eight. Lewis symbols are simply a visual shorthand. They’re like the emojis of the chemistry world, conveying information in a quick and easy-to-understand way, as long as you know what the emojis mean!
Practice makes perfect, as they say. The more you look at different elements and their Lewis symbols, the quicker you’ll become at spotting the correct ones. You’ll start to recognize the patterns, and it’ll feel less like a test and more like a friendly chat with the elements themselves. You'll be able to say, "Ah, yes, that’s the Lithium with its one lonely electron, just like it should be!" or "Look at Nitrogen, with its five dots, ready to get cozy with some other atoms."
So, go forth, fellow element detectives! Armed with your periodic table and a keen eye for dot counting, you are ready to conquer the challenge of selecting the element whose Lewis symbol is correct. May your dots be accurate and your electron counts always balanced!
