Select The Correct Statement About Osmolarity.

Alright, gather 'round, my caffeinated comrades! Today, we're diving into the wonderfully weird world of osmolarity. Now, I know what you're thinking: "Osmolarity? Sounds like something a wizard would sneeze out after a particularly potent potion." And you wouldn't be entirely wrong! But fear not, this isn't some dusty textbook lecture. We're talking about what makes your cells happy, your body hum, and why sometimes, after a marathon of salty snacks, you feel like a dried-out prune. Think of it as nature's ultimate balancing act, but with more water and less tightrope walking.
So, what is this mystical osmolarity? Imagine you have a bunch of tiny, crowded apartments – those are your cells. Now, imagine some of those apartments have a lot of stuff packed inside, like furniture, knick-knacks, and maybe even a rogue dust bunny convention. Other apartments might be more minimalist, with just the essentials. Osmolarity is basically a way of measuring how much stuff (we're talking solutes, folks, like salts and sugars) is crammed into a given volume of liquid, usually within your body fluids.
It's like comparing a tiny studio apartment filled to the brim with IKEA furniture to a spacious loft with just a beanbag chair. The studio is hyperosmolar, and the loft is hypoosmolar. And then there’s the Goldilocks zone, where everything is just right – that’s isosmolar. Easy, right? Well, the magic happens when these different "apartments" interact. Because nature, bless its quirky heart, hates an imbalance. It's like a cosmic neat freak. It wants everything to be perfectly, deliciously, and sometimes alarmingly, balanced.
And how does it achieve this balance? Through the magical, mystical, and frankly, sometimes annoying, force of osmosis. Osmosis is the star of the show here, the unsung hero (or villain, depending on your perspective) of osmolarity. It's the process where water decides to pack its bags and move from an area of low solute concentration to an area of high solute concentration. Think of water as that really nice friend who always brings extra snacks to a party. It’s trying to dilute the crowded apartment, to even things out, to make everyone feel a bit more comfortable.
So, let's say you've just downed a whole bag of those ridiculously addictive, salt-laden potato chips. Your blood, suddenly, becomes a tad more crowded with salt molecules. It's become hyperosmolar compared to the inside of your cells. What does our ever-helpful water do? It says, "Whoa, too much salt over there! I'm heading over to even this party out!" So, water rushes out of your cells and into your bloodstream to dilute that salty situation. The result? Your cells shrink, and you feel like a dehydrated husk contemplating the existential dread of thirst. It's like your cells are saying, "Hey, you left us high and dry! We need water!"
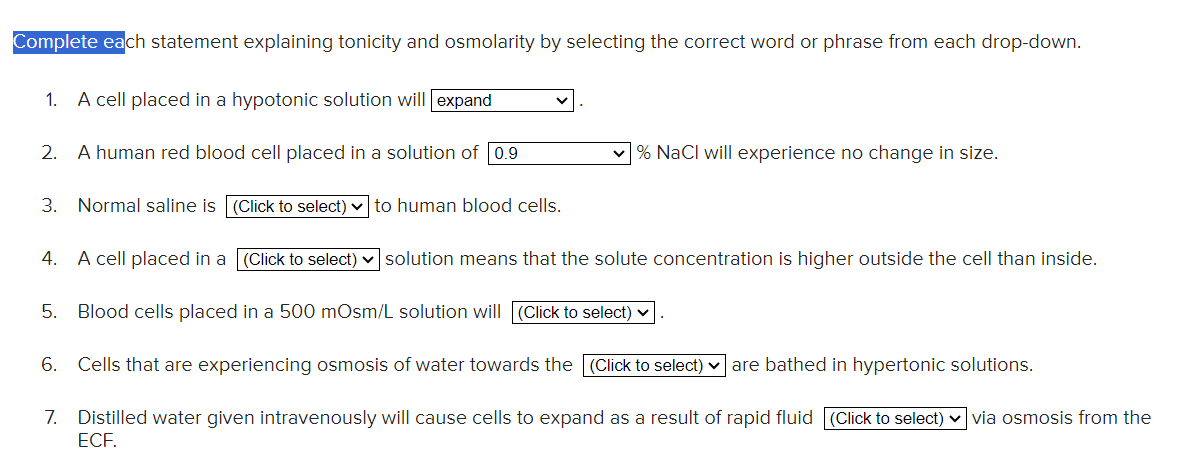
Conversely, if you chug a massive amount of plain water without any electrolytes (yes, even water can be too "plain" sometimes, in a weird way), your blood might become hypoosmolar. The water molecules will say, "Whoa, too much empty space over here! We need to go in and fill it up!" So, water rushes into your cells. If this happens too drastically, your cells can swell up like overinflated balloons. Imagine your cells looking like they’ve just had a questionable facial involving way too much hyaluronic acid. Not pretty. This is why drinking way too much water too quickly can be a problem, though thankfully, our bodies are pretty good at managing this for the most part. Still, a good reminder that balance is key, even in hydration!
Now, here's where it gets really interesting. When we talk about selecting the correct statement about osmolarity, we're essentially looking for the one that accurately describes how this whole water-shuffling game works. Let's break down some common misconceptions, shall we? Think of it as a pop quiz, but with less pressure and more potential for giggles.
Statement A: "Osmolarity refers to the concentration of only sodium in a solution."

Haha, nope! While sodium is a big player, it's not the only baddie (or goodie, depending on how you look at it). Osmolarity considers all the dissolved particles – salts, sugars, urea, you name it. It’s a whole party, not just a solo act. So, if you see this statement, give it a polite, but firm, "Bless your heart, but no."
Statement B: "Water moves from an area of lower osmolarity to an area of higher osmolarity."

BINGO! This is the money shot. This is the fundamental principle of osmosis driven by osmolarity. Water, in its infinite wisdom and desire for equilibrium, always heads towards the more crowded side, the side with more dissolved stuff, to dilute it. It’s the ultimate act of peacekeeping in the cellular world. This statement gets a standing ovation and maybe a sprinkle of glitter.
Statement C: "A hyperosmolar solution has fewer solute particles than another solution."
Oh, this one's a trickster! Remember our apartment analogy? Hyperosmolar means more stuff, not less! If a solution has fewer solute particles, it's actually the hypoosmolar one. It’s like saying a packed concert has fewer people than an empty park. Doesn't quite add up, does it? So, this statement is as wrong as wearing socks with sandals to a formal event – a fashion faux pas of biological proportions.
Statement D: "Osmolarity is only important for kidney function."
While the kidneys are absolute rockstars at managing osmolarity and keeping our fluid balance in check (seriously, give your kidneys a high-five next time you pee!), they're not the only ones playing this game. Every single cell in your body is constantly dealing with osmolarity. From your brain cells trying to keep their cool to your muscle cells flexing their brawn, they all rely on proper osmolarity to function. It’s a body-wide phenomenon, not just a kidney thing. Think of the kidneys as the head chefs, but the entire restaurant staff is involved in keeping things running smoothly.
So, there you have it! Osmolarity, in all its water-moving, cell-swelling, and salt-balancing glory. It's a fundamental concept, and understanding it helps us appreciate just how intricate and amazing our bodies are. It’s the silent, unseen force that keeps us from becoming either shriveled raisins or waterlogged balloons. Pretty neat, right? Now, who wants another coffee? I think my brain just ran a marathon calculating solute concentrations!
