Select The Correct Statement About Equilibrium

Hey there, folks! Ever feel like life's a bit of a balancing act? Like you're juggling flaming torches while riding a unicycle on a tightrope? Well, you're not alone. That feeling, that sweet spot where everything just sort of works, even if it's a little wobbly, is what we're talking about today: equilibrium. Yep, that fancy word we hear in science class, but turns out, it's lurking around us more than you think. It's the secret sauce to why your morning coffee doesn't instantly evaporate, or why your perfectly stacked pizza slices don't just slide off the plate. Let's dive into the wonderfully relatable world of things being... well, balanced.
Think about it. You're making breakfast. You've got the milk carton on the counter, and you're pouring it into your cereal. The milk is flowing out, right? But it’s not like the whole carton is just going to poof and be empty instantly. There's a certain rate at which it's coming out. Now, imagine you also have a magic faucet above the cereal bowl that's adding milk at the exact same speed. Suddenly, the amount of milk in the bowl stays pretty darn constant. It’s like a tiny milk river, flowing in and flowing out at the same pace. That, my friends, is a beautiful, albeit slightly hypothetical, illustration of dynamic equilibrium. The milk is still moving, but the overall level isn't changing. It’s a constant flow, a never-ending dance of molecules.
This isn't just for breakfast beverages. Consider your favorite comfy couch. When you plop down on it, it squishes, right? That’s the couch’s springs or cushions pushing back against your weight. You’re applying a downward force (gravity pulling you), and the couch is applying an upward force. When you’re sitting there, not sinking further into the abyss of relaxation, it means those forces are pretty much balanced. You've reached a kind of static equilibrium. You're not moving, the couch isn't collapsing (hopefully!), and all is right with the world. It’s the universe’s way of saying, “Yep, you’re comfy, and the couch can handle it.”
Now, let’s get a tiny bit nerdy, but in a fun way. In science, when we talk about equilibrium, especially in chemical reactions, it’s not about everything grinding to a halt. Oh no. That would be a bit boring, wouldn't it? Imagine a crowded party where everyone is politely standing still. No fun! In a chemical reaction at equilibrium, it’s more like a bustling dance floor. The dancers (molecules) are still moving, mingling, and changing partners (reacting). But here’s the kicker: for every molecule that pairs up to form something new (a product), another molecule of that new thing breaks apart to go back to being what it was before (a reactant). It's a constant back-and-forth, a ping-pong match of molecules.
So, what’s the big deal about equilibrium? Why should you care that your coffee is in a state of not-quite-equilibrium (because it's cooling, and stuff is evaporating)? Well, it's all about stability and predictability. When something is in equilibrium, it's in a relatively stable state. It’s not going to suddenly decide to become something else without a good reason. Think of it like your diet. You're trying to maintain a healthy weight. If you’re eating more calories than you’re burning, you’re not in equilibrium – you’re gaining weight. If you’re burning more than you’re eating, again, not equilibrium – you’re losing weight. When you find that sweet spot where your calorie intake and output are roughly equal, you’re approaching a kind of bodily equilibrium. Your weight stays relatively stable. It's the universe's way of saying, "Alright, we've found a groove here, let's keep it going."
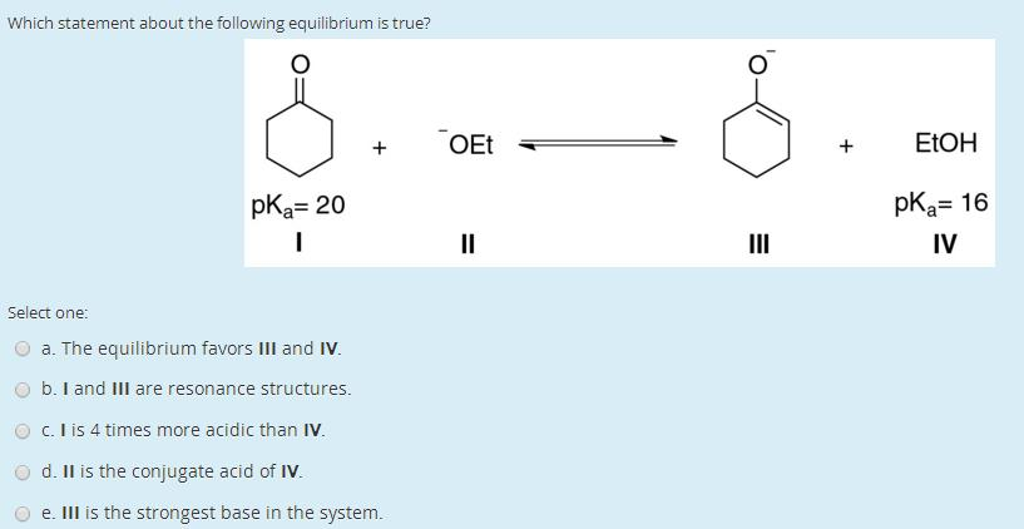
Let’s look at a few statements about equilibrium. We’ll try to pick the one that just makes sense, the one that feels right in your gut, like knowing exactly how much sugar to put in your tea. Imagine a sealed bottle of soda. We all know what happens when you shake that bad boy up, right? Fizz goes everywhere! But what if that bottle has been sitting on the shelf for a while? There's a certain amount of carbon dioxide gas dissolved in the liquid, and a certain amount of it hanging out in the space above the liquid. These two are in a kind of equilibrium. The gas molecules are constantly hopping from the liquid into the gas phase, and from the gas phase back into the liquid. It's a busy little exchange happening, but the overall amount of gas in the liquid and in the headspace stays pretty much the same. It’s like a tiny molecular revolving door.
Now, let's consider some statements about this whole equilibrium thing.

Statement A: When a system is in equilibrium, all chemical reactions have stopped.
Does this sound right? Remember our dancing molecules? If they stopped dancing, there'd be no more changing partners, no more creating new things. That’s more like a funeral than a party. So, this statement feels a bit... dead. Like a perfectly still pond. While a pond can be still, the molecules within it are definitely not. So,
Statement B: Equilibrium is reached when the rates of the forward and reverse reactions are equal.
Hmmm, let’s think about our soda bottle again. The gas molecules are leaving the liquid and entering the gas phase (forward reaction). The gas molecules are also rejoining the liquid (reverse reaction). If the number of molecules leaving the liquid per second is exactly the same as the number of molecules returning to the liquid per second, then the amount of dissolved gas and gas in the headspace will stay constant. This sounds an awful lot like our revolving door analogy! It’s a state of dynamic balance. This statement feels like it’s got some legs. It’s describing that constant, yet balanced, activity we talked about.
Statement C: Equilibrium can only be achieved in open systems.
What’s an open system? Think of a campfire. Smoke and heat escape into the air. Stuff is constantly coming and going. Now, what about our sealed soda bottle? It's a closed system. Nothing really gets in or out. Yet, we established that there’s an equilibrium happening inside. If a system is constantly losing or gaining things, it’s going to be hard to find a stable balance, right? Imagine trying to keep your room tidy if people were constantly coming in and taking your stuff and bringing in random junk. It would be chaos! Equilibrium needs a certain level of containment, at least for the reactants and products involved. So,

Statement D: The concentration of reactants and products must be equal at equilibrium.
Okay, let’s go back to our party analogy. Does everyone at the party have to be wearing the same outfit? No way! You can have a bunch of people in blue shirts and a few in red shirts, and it's still a rocking party. Similarly, in a chemical reaction at equilibrium, you can have a lot more of the product than the reactant, or vice versa. As long as the rates of forming and breaking down are equal, you’ve got equilibrium. It’s not about the amount being the same, but the speed of the changes being the same. Imagine a perfectly balanced tug-of-war. One team might have more people, but if the other team is stronger and they’re pulling with equal force, the rope stays in the middle. It’s not that both teams have the same number of people, it’s that they exert the same pull. So,
So, when we sift through these statements, one of them just rings true. It’s the one that captures the essence of that constant, yet balanced, activity. It's like finding the perfect rhythm when you're dancing – you're moving, but you're in sync. You're not frozen in place, but you're not flailing wildly either. You've found your groove.
Let’s take Statement B again: Equilibrium is reached when the rates of the forward and reverse reactions are equal. Think about it. If the forward reaction (making products) is happening faster than the reverse reaction (making reactants), you’ll just keep making more products, and you’ll never reach a steady state. You'll run out of reactants! Conversely, if the reverse reaction is faster, you'll just keep breaking down your products until you're back to where you started. It’s only when those two speeds perfectly match up that the amounts of reactants and products will stop changing. It’s like a perfectly calibrated seesaw. If one side goes down faster than the other comes up, it’ll just keep tipping. But when they’re moving in perfect opposition, the seesaw stays level. That’s equilibrium, folks! The sweet spot where the hustle and bustle of chemical change perfectly cancel each other out, leaving a state of apparent stillness, even though the action is still very much alive.
It’s the reason why when you open a bottle of water, it doesn’t immediately freeze or boil away. There’s an equilibrium between the liquid water and the water vapor in the air around it. Of course, temperature and pressure play a role in this equilibrium, nudging it this way or that, but at any given moment, there's a balance. It’s like the universe whispering, “We’re good here for now.”
So, to sum it up, equilibrium isn't about things grinding to a halt. It's about a dynamic, ongoing process where the opposing forces or reactions are happening at the same speed. It’s the calm on the surface of a churning sea, the steady hum of a well-oiled machine, the contented sigh after a perfectly balanced meal. It’s the universe’s way of finding a comfortable middle ground, and understanding it helps us understand why so many things in our world, from the air we breathe to the reactions in our bodies, behave the way they do. It’s a little bit of science that makes a whole lot of everyday sense!
