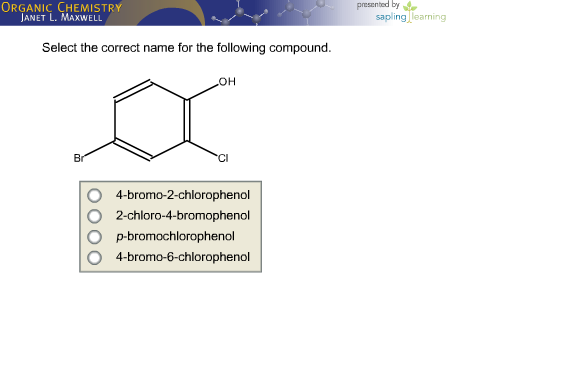
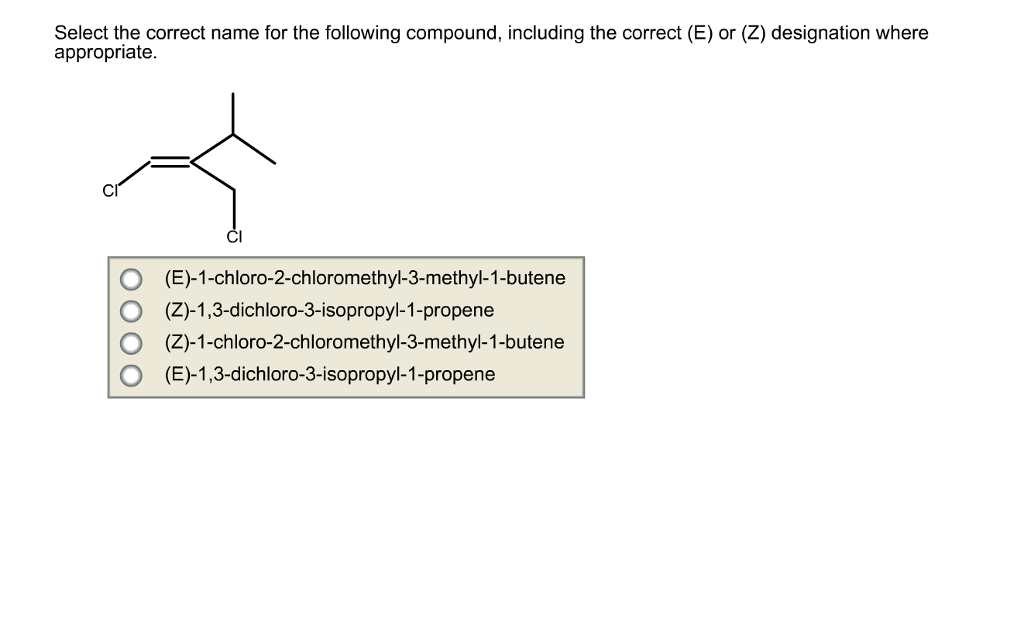
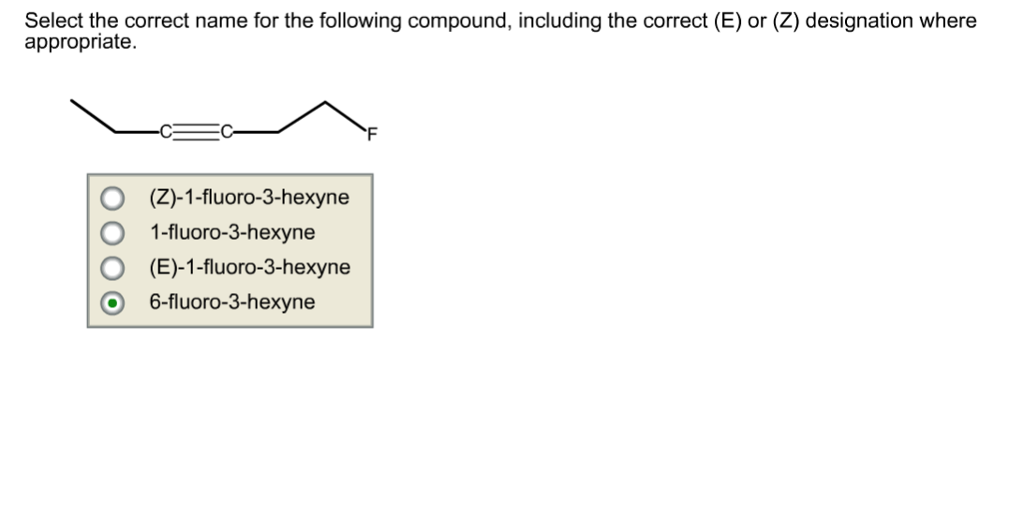
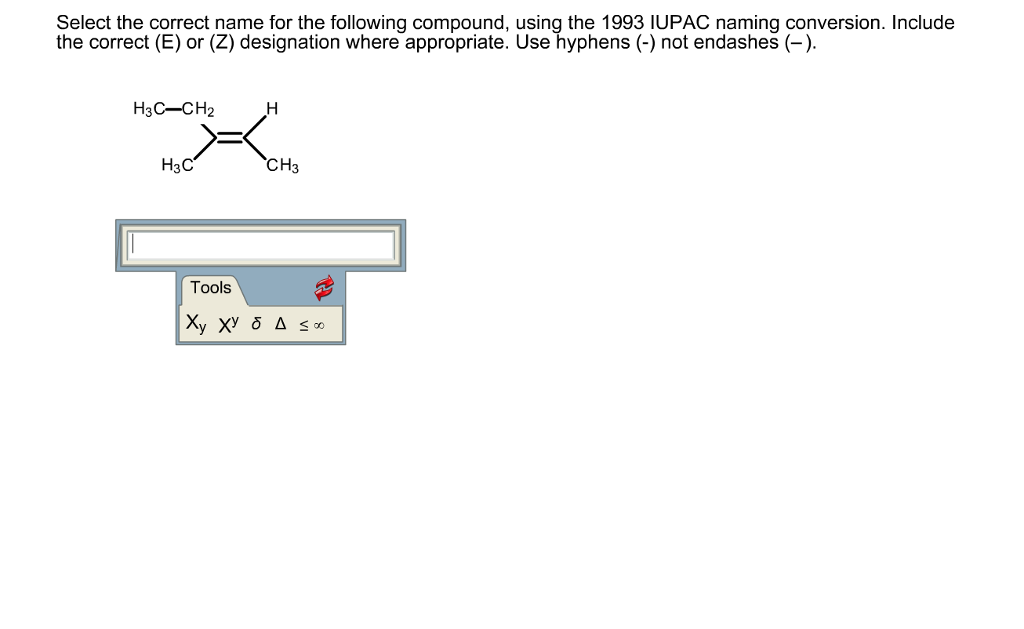
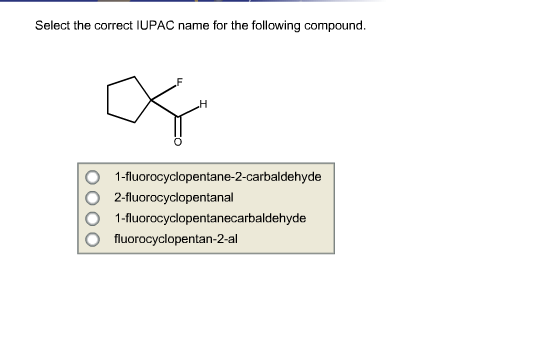
Select The Correct Name For The Following Compound
Ever found yourself staring at a jumble of letters and numbers, feeling like you've stumbled upon a secret code? Yep, we’re talking about chemical names. It’s not exactly beach reads or binge-watching, is it? But hey, understanding how things get their official monikers can be surprisingly satisfying. Think of it like knowing the real name behind your favorite celebrity’s stage name – it adds a layer of depth, a little “aha!” moment.
Today, we’re diving into the wonderfully weird world of naming chemical compounds. It might sound intimidating, like trying to decipher an ancient scroll, but trust me, it’s more like putting together a really clever puzzle. And once you get the hang of it, you’ll feel like a bona fide science guru, effortlessly gliding through the lab or the grocery store aisle, mentally naming everything in sight.
Let’s get this party started with a little context. Why do we even need these elaborate names? Well, imagine trying to order a specific type of artisanal coffee without a name. “Yeah, can I get that dark, roasted bean from that place with the mountain logo, you know, the one that smells a bit like chocolate?” Chaos! Chemical naming is all about precision and clarity. It ensures that no matter where you are in the world, or who you’re talking to, you’re referring to the exact same substance. It’s the universal language of chemistry, and it’s pretty darn cool.
Now, the prompt is a bit of a tease: "Select The Correct Name For The Following Compound." But without the compound itself, we’re flying blind! So, let’s make this a masterclass. We'll equip you with the tools and insights to tackle any compound name thrown your way. Consider this your cheat sheet, your secret weapon, your backstage pass to the glamorous (okay, maybe not glamorous, but definitely fascinating) world of chemical nomenclature.
Unlocking the Secrets: The Logic Behind the Labels
The beauty of chemical naming lies in its systematic approach. It's not random scribbling; it's a carefully constructed system, largely governed by the International Union of Pure and Applied Chemistry (IUPAC). Think of IUPAC as the ultimate style guide for chemists, ensuring everyone’s on the same page. Their rules are designed to be logical, predictable, and, importantly, unambiguous.
We're going to focus on the most common types of compounds you'll encounter in everyday life and in introductory chemistry. This includes ionic compounds and molecular compounds. Each has its own set of rules, like different etiquette for different social events. But don't worry, they're not that complicated. With a little practice, they'll become second nature.
Ionic Compounds: The "Salt" of the Earth (and So Much More!)
Ionic compounds are formed when atoms that tend to lose electrons (metals) bond with atoms that tend to gain electrons (nonmetals). This creates charged particles called ions, which then attract each other like tiny, charged magnets. Think of it as a cosmic dance of electron exchange!
The naming convention for simple ionic compounds is usually straightforward. You take the name of the metal and combine it with the name of the nonmetal, but you change the ending of the nonmetal to "-ide."
For example, let's take a classic: Sodium and Chlorine. Sodium (Na) is a metal, and Chlorine (Cl) is a nonmetal. Sodium tends to lose one electron, becoming Na+, and Chlorine tends to gain one electron, becoming Cl-. They come together to form Sodium Chloride – the stuff you sprinkle on your fries! Pretty neat, right?
Practical Tip: When you see a compound name ending in "-ide" and the first part sounds like a metal (like "sodium," "potassium," "calcium"), you're likely looking at an ionic compound. The second part, with the "-ide" ending, will be a nonmetal (like "chloride," "oxide," "sulfide").
What about compounds with more than one atom of a particular element? This is where prefixes come in. For molecular compounds (which we'll get to next), prefixes like "di-" (two), "tri-" (three), "tetra-" (four), and so on, are used. However, for simple ionic compounds, the charges already dictate the ratio, so you don't usually need prefixes. For instance, Calcium Chloride is CaCl2. Calcium (Ca) forms a +2 ion (Ca2+), and Chlorine (Cl) forms a -1 ion (Cl-). You need two chloride ions to balance out the one calcium ion. But we don't call it "calcium dichloride." The name "calcium chloride" is sufficient because the charges are understood.
Cultural Reference: Think of common salts you know. Table salt (Sodium Chloride), Epsom salt (Magnesium Sulfate – we'll get to polyatomic ions in a sec!), and even the baking soda in your kitchen (Sodium Bicarbonate). These are all ionic compounds, and their names, while scientific, often have familiar roots.
Now, things get a tiny bit more complex with metals that can form multiple different charges. These are called transition metals. For example, Iron can be Fe2+ or Fe3+. To distinguish between Iron(II) Chloride (FeCl2) and Iron(III) Chloride (FeCl3), we use Roman numerals in parentheses after the metal's name. This is called the Stock system and is incredibly useful.
Fun Fact: The Roman numeral system, while ancient, is still a vital part of scientific communication. Imagine trying to explain Iron-2 versus Iron-3 without it – it would be a linguistic nightmare!
Molecular Compounds: The "Sharing is Caring" Crew
Molecular compounds, also known as covalent compounds, are formed when nonmetals share electrons. This happens because the nonmetals have similar tendencies to attract electrons, so rather than one atom completely taking an electron from another, they form a bond by sharing. It’s a more collaborative approach to chemical bonding.
The naming for molecular compounds is where those prefixes we mentioned earlier really shine. They tell you exactly how many atoms of each element are present in the molecule. The rule is to use a prefix to denote the number of atoms of the first element, and then use a prefix and the "-ide" ending for the second element (just like with ionic compounds).
Here’s how it generally works:
- Mono-: one
- Di-: two
- Tri-: three
- Tetra-: four
- Penta-: five
- Hexa-: six
- Hepta-: seven
- Octa-: eight
- Nona-: nine
- Deca-: ten
Let's look at some examples. Water, H2O. It’s two hydrogen atoms and one oxygen atom. The name is dihydrogen monoxide. The "di-" tells you there are two hydrogens, and "mono-" tells you there's one oxygen. However, water is such a common compound that we’ve all agreed to call it by its trivial name, "water." This is a bit like how everyone knows what "Coke" is, even though its official name might be more complex.
Another common one: carbon dioxide, CO2. "Di-" for two oxygens, and "mono-" for one carbon. Wait, why isn't it "monocarbon dioxide"? Ah, there’s a little quirk: If the first element only has one atom, you often drop the "mono-" prefix. So, it’s "carbon dioxide," not "monocarbon dioxide." This makes the name shorter and more elegant.
Let’s try another. Sulfur dioxide, SO2. "Di-" for oxygen, and "mono-" for sulfur. Again, we drop the "mono-" for sulfur, so it's sulfur dioxide. If we had sulfur trioxide, SO3, it would be sulfur trioxide. Simple, right?
What about sulfur tetrachloride, SCl4? "Tetra-" for four chlorines, and "mono-" for one sulfur. Following our rule, we drop the "mono-" for sulfur, so it becomes sulfur tetrachloride. See how the prefixes clearly define the composition?
Practical Tip: When identifying a molecular compound, look for a name that starts with prefixes (other than "mono-" for the first element) and ends in "-ide." It's a strong indicator that electrons are being shared.
Cultural Reference: Think about the air we breathe. Nitrogen (N2) and Oxygen (O2) are diatomic molecules, meaning they exist as two atoms bonded together. We don't usually name them as "dinitrogen" and "dioxygen" in everyday conversation, but in a scientific context, those are their molecular names.
Polyatomic Ions: The "Team Players" of Chemistry
Things get a little more interesting when we have groups of atoms that stick together and behave as a single charged unit. These are called polyatomic ions. They have their own special names, and you just have to memorize them, kind of like learning the names of your favorite band members. Some common ones include sulfate (SO42-), nitrate (NO3-), and ammonium (NH4+).
When these polyatomic ions bond with metals, the naming follows similar rules, but you use the full name of the polyatomic ion. For example, Sodium Sulfate would be Na2SO4. Sodium (Na+) and Sulfate (SO42-). You need two sodium ions to balance the -2 charge of the sulfate ion. The name is simply sodium sulfate. No "-ide" ending on sulfate, because it's the name of the ion itself.
Similarly, Ammonium Nitrate is NH4NO3. Ammonium (NH4+) and Nitrate (NO3-). Here, the +1 charge of ammonium balances the -1 charge of nitrate, so you have a 1:1 ratio.
Fun Fact: Some polyatomic ions have names that sound quite dramatic, like "chlorate" or "permanganate." They're like the rock stars of the ion world, with their own distinct identities!
Putting It All Together: The Naming Detective Work
So, when you’re faced with a compound name and need to select the correct one, here’s your mental checklist:
- Identify the elements involved. Are they metals, nonmetals, or both?
- Look for prefixes. If you see "di-," "tri-," "tetra-," etc., it's likely a molecular compound.
- Check for "-ide" endings. This is common in both ionic and molecular compounds, but if it's a nonmetal with an "-ide" ending, and there are no prefixes (or only one atom of the first element), it’s often ionic.
- Watch out for polyatomic ion names. If you see names like "sulfate," "nitrate," "carbonate," "hydroxide," "ammonium," you're dealing with a polyatomic ion.
- Consider Roman numerals. If a metal name is followed by a Roman numeral in parentheses (e.g., Iron(III)), it indicates the charge of the metal ion.
Let's imagine we have a compound and we're given a few options for its name. For instance, let's say the compound is composed of Aluminum (Al) and Oxygen (O). Aluminum is a metal in Group 13, typically forming a +3 ion (Al3+). Oxygen is a nonmetal, typically forming a -2 ion (O2-).
To balance the charges, we need two aluminum ions (2 x +3 = +6) and three oxygen ions (3 x -2 = -6). This would give us the formula Al2O3.
Now, how do we name this? It's an ionic compound formed between a metal and a nonmetal. So, we take the metal name "aluminum" and the nonmetal name "oxide." Since aluminum in this case consistently forms a +3 ion, we don't need Roman numerals. The correct name is aluminum oxide.
If we were given options like:
- Aluminum oxide
- Dialuminum trioxide
- Aluminum trioxide
- Aluminum oxyde
We'd systematically eliminate the incorrect ones.
"Dialuminum trioxide" and "Aluminum trioxide" use prefixes, which are typically for molecular compounds. While the ratios are correct, this compound is ionic. So, these are incorrect.
"Aluminum oxyde" is just a misspelling of oxide. Common mistake, but not the correct scientific term.
This leaves aluminum oxide as the correct name for the ionic compound Al2O3.
Pro Tip: When in doubt, think about the type of bonding. Metals and nonmetals usually mean ionic. Two nonmetals usually mean molecular.
The ability to name compounds correctly is like having a key to unlock a treasure chest of chemical knowledge. It’s not just about memorizing facts; it’s about understanding the underlying principles and patterns. It's about appreciating the elegance and order that chemists have brought to the seemingly chaotic world of molecules.
So, the next time you see a chemical name, don't shy away from it. Break it down, look for the clues, and remember the systematic rules. You're not just reading a name; you're reading a story about how atoms have come together. It’s a story of attraction, sharing, and balance. And in a world that often feels complex, finding clarity and order in something like chemical nomenclature can be surprisingly… grounding. It's a little bit of science that makes the world around us, from the air we breathe to the food we eat, just a little bit more understandable, one name at a time.
