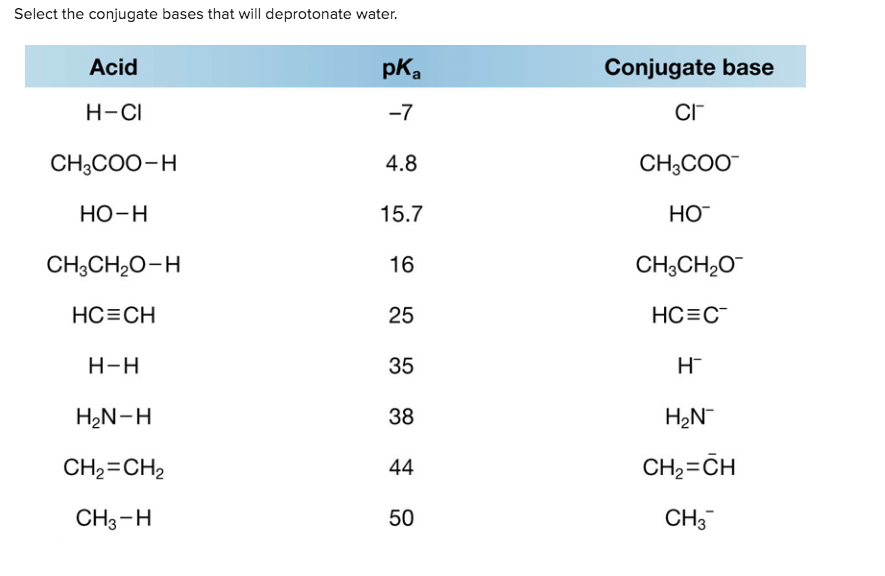
Select The Conjugate Bases That Will Deprotonate Water
Hey there, chemistry curious folks! Ever wonder what happens when you mix a few things together and watch them dance? Well, today we're diving into a super cool, slightly dramatic, and totally fascinating corner of chemistry. We're going to talk about choosing the right partners to coax a tiny bit of drama out of water. Yep, you heard that right. Water, the stuff we drink every day, can be coaxed into giving up a proton – that's like a little hydrogen superhero – if you ask it very nicely with the right kind of friend.
Imagine water molecules as little shy introverts. They're perfectly happy being H₂O, holding hands and minding their own business. But sometimes, a really, really strong extrovert comes along and says, "Hey, water! Wanna let loose for a sec?" That extrovert is what we call a conjugate base. And not just any conjugate base will do the trick. We need the right ones, the ones with enough persuasive charm to convince a water molecule to shed its hydrogen.
This whole process is like picking the perfect contestant for a reality TV show. You've got a bunch of potential contenders, but only a select few have the personality, the power, and the oomph to actually get the job done. And the job here? Well, it's to deprotonate water. Sounds fancy, right? But think of it as water having a little molehill moment, where it temporarily lends a hydrogen. It’s like water saying, "Okay, okay, you convinced me! Here, take it!"
So, what makes one conjugate base a superstar at this game, and another a total wallflower? It all comes down to how badly they want that hydrogen. Think of it like this: some things are super, super hungry for something, and they'll go to great lengths to get it. Other things are pretty chill and aren't bothered if they don't get what they want.
In the world of chemistry, this "hunger" is related to something called acidity. A strong acid is like a really demanding personality. It's desperate to get rid of its proton. Its conjugate base, then, is like that person who is completely unfazed when their proton-leaving friend does just that. They're perfectly happy without it, and they're not going to go chasing after someone else’s proton to replace it.

Now, here's where it gets really fun. Water is, in its own way, a bit of a diva. It doesn't just hand over its hydrogen to anyone. It needs someone who is even less attached to their own proton than water is. This means we're looking for conjugate bases that come from acids that are weaker than water itself. Confusing? Let’s break it down with a fun analogy.
Imagine you're at a party, and you've got two cookies. You're pretty happy with your cookies. Now, someone comes up to you, let’s call them "The Polite Requestor". This person is like a strong acid. They're so eager for a cookie, they practically demand one. You're likely to say, "Sure, here you go!" because they’re so persuasive and frankly, you have plenty.

But then, another person, "The Super Casual Friend", comes by. They might like cookies, but they're not dying for one. They're perfectly content with whatever they have. If you offer them a cookie, great! If not, no biggie. This "Super Casual Friend" is like a weaker acid. Their conjugate base (that's the friend after they've potentially given away a cookie, if that were a thing) is also pretty chill.
When we want to deprotonate water, we need a chemical friend who is like "The Super Casual Friend", but even more so! We need a conjugate base that is so incredibly stable and content without an extra hydrogen that it’ll happily accept one from water. This means the original acid that formed this conjugate base has to be really, really weak. So weak, in fact, that it's weaker than water's own tendency to hold onto its proton. It's like finding someone who is less likely to grab a cookie than you are to offer one!
So, when you're looking at a list of potential conjugate bases and you're asked to pick the ones that can deprotonate water, you're on a mission to find the ultimate chill-masters. You're looking for those chemical entities that are born from the weakest of acids. These are the stars of our show, the true proton-persuaders!
Think of it like a popularity contest. The conjugate bases that win the "deprotonate water" award are the ones whose parent acids were so unpopular (in terms of giving away protons) that the conjugate bases are just super happy being what they are and aren't desperate for more!
Solved Select the conjugate bases that will deprotonate | Chegg.com
It’s a bit of a mind-bender, but that’s the magic of chemistry! We're selecting the conjugate bases that have a stronger pull towards that little hydrogen than water does. They’re like magnets, but for protons, and they’ve got to be really strong magnets to get water to budge.
The key is to remember that strong acids make weak conjugate bases. And conversely, weak acids make strong conjugate bases. So, if we want a conjugate base that is strong enough to steal a proton from water, it means the acid that formed it must be weaker than water itself. It's a game of relative strengths, and it’s quite exhilarating to figure out who’s who.
So, next time you see "select the conjugate bases that will deprotonate water," don't get bogged down. Just picture that party scenario, the cookies, and the incredibly laid-back friends. You're looking for the ultimate chill vibes in a molecule! It’s a delightful puzzle, and the satisfaction of picking the right players is truly rewarding. Happy selecting!