Select The Complexes That Exhibit Geometric Isomerism

Hey there, science enthusiasts and curious minds! Ever felt like life sometimes throws you the same old ingredients but presents them in a slightly different way? Well, buckle up, because today we're diving into a world where even in the realm of molecules, things can get spicy with a little twist! We're talking about a super cool concept in chemistry called geometric isomerism. Sounds a bit fancy, right? But trust me, it’s less about complicated formulas and more about how the universe likes to play with its building blocks.
So, what exactly is geometric isomerism? Think of it like this: imagine you have four friends, all with the same personality (let’s call them Alex, Ben, Chloe, and David). Now, imagine you're trying to arrange them around a small, square table. There are a few ways you can do this, right? Some friends might be sitting next to each other, others across from each other. Geometric isomerism is kind of like that, but for molecules! It happens when molecules have the same chemical formula, meaning they’re made of the exact same atoms, but these atoms are arranged in different spatial configurations. It’s like having the same ingredients for a cake, but you can bake it in a round pan or a square pan – the cake is still a cake, but it looks and feels a bit different!
The key ingredient for geometric isomerism, the thing that makes this spatial dance possible, is often a double bond. You know, those strong connections between atoms? When atoms are linked by a double bond, they can't just spin around freely like they can with a single bond. This rigidity forces other groups attached to those atoms into specific positions. It's like having a dance floor where the music has a strict beat – you have to move in certain ways! Another common culprit is a ring structure, where the atoms are locked into a cycle, again limiting their freedom to rotate. So, if you see a double bond or a ring, your antenna should start twitching for geometric isomers. Exciting, isn't it? The potential for variation is already building!
Now, when we talk about geometric isomers, we usually hear about two main flavors: cis and trans. Let's break these down, shall we? Imagine our double bond is like a fence. The atoms attached to either side of this fence are like little garden gnomes. In the cis isomer, the two "main" gnomes (let’s say the ones we’re most interested in) are on the same side of the fence. They’re cozy neighbors, hanging out together. In the trans isomer, however, these same two gnomes are on opposite sides of the fence. They’re a bit more spread out, keeping their distance. See the difference? It’s all about that relative positioning!
So, how do we actually select these complexes that exhibit geometric isomerism? It's like being a detective, looking for clues. The most common scenario we encounter this in is with alkenes. Remember those double bonds we talked about? Alkenes are hydrocarbons that have at least one carbon-carbon double bond. If each of the carbon atoms involved in the double bond is attached to two different groups, then you’ve got a golden ticket to geometric isomerism! For example, take 2-butene. It has a double bond between the second and third carbon atoms. The second carbon is attached to a hydrogen and a methyl group, and the third carbon is also attached to a hydrogen and a methyl group. Since both carbons have two different things attached, you can have a cis-2-butene (where the two methyl groups are on the same side of the double bond) and a trans-2-butene (where they are on opposite sides). Boom! Two different molecules from the same ingredients!

But it’s not just simple alkenes! This phenomenon extends to cyclic compounds too. Think about a cyclohexane ring. If you have substituents (those extra little bits attached to the ring) on adjacent carbon atoms, they can be on the same side of the ring (cis) or on opposite sides (trans). It’s like having two people on a merry-go-round – they can be next to each other or diametrically opposed! The possibilities just keep on unfolding, making chemistry a delightful puzzle.
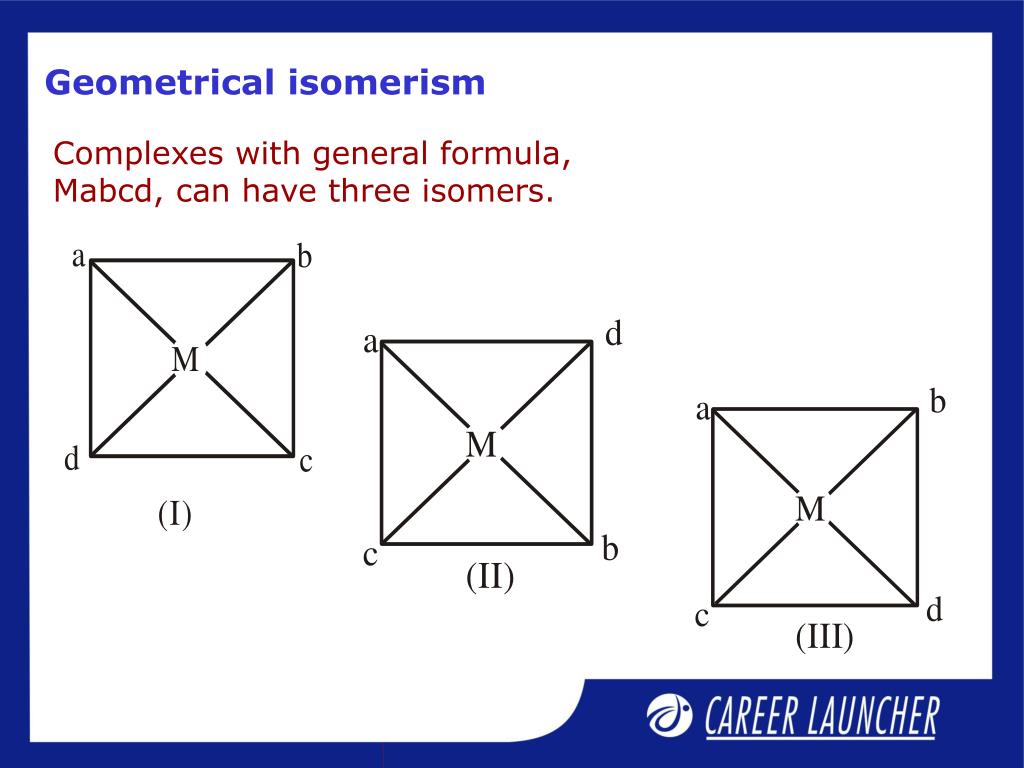
And let's not forget about the glamorous world of coordination complexes! These are the intricate structures formed when a central metal atom is surrounded by ligands (molecules or ions that bind to the metal). When a metal atom has multiple ligands, and especially when some of those ligands are identical, geometric isomerism can pop up. For instance, consider a complex with the formula MA₂B₂, where M is the central metal, A and B are different ligands, and there are two of each. If these ligands are arranged around the metal in a square planar or octahedral geometry, you can get cis and trans isomers. In the cis isomer, the two A ligands are next to each other, and in the trans isomer, they are opposite each other. It’s like arranging flowers in a vase – you can group them or spread them out for a totally different look!
Why should you care about this? Because it’s not just abstract chemistry! The difference between cis and trans isomers can have real-world consequences. Think about vitamins. Vitamin A exists in two geometric forms, cis and trans. The trans form is the one our bodies can use for vision. Or consider pharmaceuticals. Sometimes, only one isomer of a drug is effective, while the other might be inactive or even harmful. It’s a subtle difference in shape that can lead to dramatically different outcomes. How cool is that? The universe isn't just throwing random atoms around; it's got a sense of style!
Learning to identify these geometric isomers is like unlocking a secret code of the universe. It’s a way of appreciating the diversity and elegance that exists even at the molecular level. It encourages us to look closer, to ask "how?" and "why?". It’s about understanding that even with the same components, the arrangement can lead to entirely new properties and functionalities. This isn't just about memorizing facts; it's about developing a keen eye for structure and an appreciation for the subtle nuances that make the world so fascinating.
So, the next time you encounter a molecule with a double bond or a ring structure, or even a complex coordination compound, remember to pause and think: "Could this be a case of geometric isomerism?" It’s a wonderful way to engage with chemistry, to see it not as a daunting subject, but as an exciting exploration of spatial relationships and molecular architecture. Every time you identify a potential for cis and trans isomers, you’re not just doing chemistry; you’re appreciating a tiny, elegant dance that the molecules are performing. Keep exploring, keep questioning, and let the fascinating world of molecular shapes inspire your curiosity!
