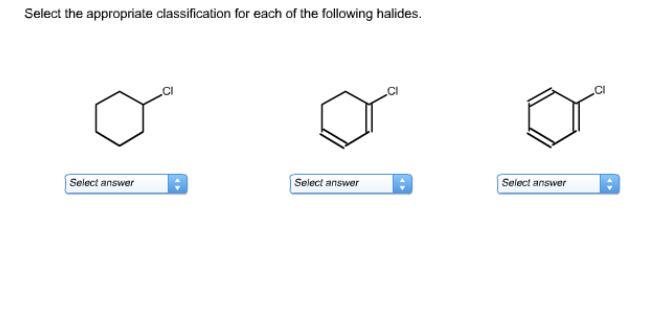
Select The Appropriate Classification For Each Of The Halides.
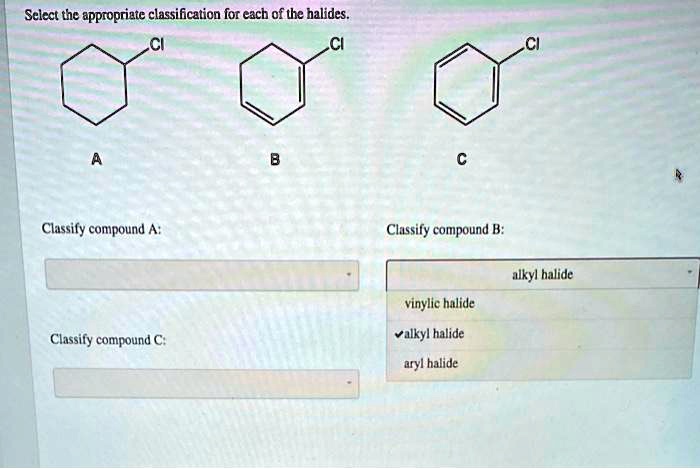
Hey there, chemistry enthusiast (or soon-to-be)! So, you’ve stumbled upon the wonderful world of halides and you’re wondering, "What’s what?" Don’t worry, it’s not as complicated as it sounds. Think of it like sorting your LEGOs – you’ve got different shapes and sizes, and knowing where each one goes makes building way more fun and, you know, possible.
Today, we’re going to chat about how to pick the appropriate classification for each of these nifty halide compounds. We’re talking about those molecules where a halogen – you know, fluorine (F), chlorine (Cl), bromine (Br), or iodine (I) – decides to get cozy with some carbon. Pretty neat, right? They’re like the little black dresses of the organic chemistry world – versatile and always making an appearance.
Before we dive headfirst into the classifying fun, let’s do a super quick refresher on what halides actually are. Basically, a halide is any chemical compound that contains a halogen atom bonded to a carbon atom. That’s the core idea. Think of the halogen as the guest of honor at a party, and the carbon is the host. They’re linked up, and that bond is what makes them special.
Now, the reason we need classifications is because these carbon-halogen bonds can behave in different ways. And when they behave differently, they can do different things in reactions. It’s all about predicting their personality, you know? Like knowing if your friend is more likely to suggest a quiet night in or a spontaneous road trip. Understanding their classification helps us understand their reactivity.
So, let’s break down the main ways we classify these guys. It mostly comes down to where the halogen is attached to the carbon atom. Think of it like assigning a zip code to your carbon atom. Does it have a lot of friends (other carbon atoms) nearby, or is it more of a loner?
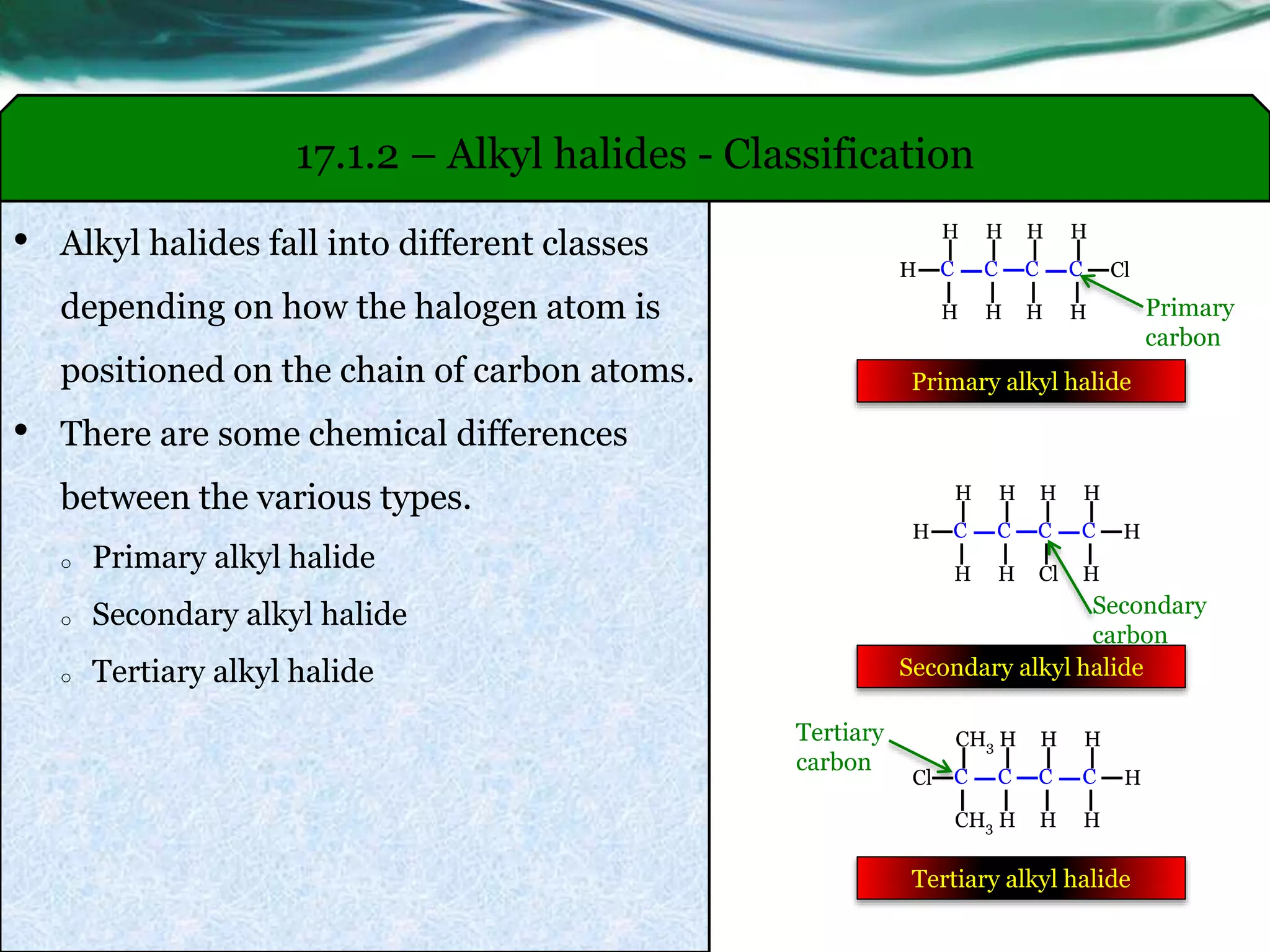
The Big Three: Primary, Secondary, and Tertiary Halides
These are your bread and butter, your classic classifications. It’s all about how many carbon atoms are directly attached to the carbon atom bearing the halogen. This is the most fundamental way to categorize them.
Primary Halides (1°)
Imagine the carbon atom that’s holding onto our halogen friend. If that particular carbon atom is only attached to one other carbon atom, then congratulations, you’ve got yourself a primary halide! It’s like the carbon is saying, "Yep, I've got one buddy over here, and that's it!"
Let’s visualize it. You’ll see something like this:
R–CH2–X
Here, R represents another carbon-containing group (or even a hydrogen, but we’re focusing on carbons for this classification). The carbon with the ‘X’ (our halogen) is bonded to only one ‘R’ group. Simple as that!
Think of methyl chloride (CH3Cl). The carbon here is bonded to zero other carbons, but for the purpose of this classification, we consider it like a primary. It’s the ultimate minimalist carbon!
Then you have ethyl chloride (CH3CH2Cl). The carbon attached to the chlorine is only attached to one other carbon. See? Easy peasy!
These primary halides are generally pretty chill. They’re often involved in reactions where they can be easily nudged out of their spot. Think of them as the welcoming committee – ready to let new people join the party.
Secondary Halides (2°)
Now, if the carbon atom holding the halogen is attached to two other carbon atoms, we’re stepping into secondary halide territory. This carbon is a bit more popular, you could say. It’s got two carbon buddies keeping it company.
The general structure looks like:

R–CH(X)–R’
Here, both R and R’ are carbon-containing groups. The carbon with ‘X’ has two direct links to other carbons. Notice the middle carbon has one hydrogen atom remaining, while the others have none (they’re fully saturated with carbon connections).
A good example is isopropyl chloride (CH3CHClCH3). The carbon atom attached to the chlorine is bonded to two methyl groups (which are CH3, so they contain carbon). So, two other carbons are directly linked to our halocarbon.
Secondary halides are a bit more in the middle. They can be persuaded to react, but maybe not as readily as their primary cousins. They have a little more 'stuff' around them, making them a tad more stable and a little less eager to jump into things.
Tertiary Halides (3°)
And finally, the life of the party! If the carbon atom holding the halogen is attached to three other carbon atoms, then we have a tertiary halide. This carbon is totally surrounded by other carbons. It’s like a VIP booth at the club – not easily accessible!
The structure is like this:
R–C(X)(R’)(R’’)
All R, R’, and R’’ are carbon-containing groups. See how the carbon with ‘X’ has no hydrogen atoms attached? It’s completely swamped by other carbons.
A classic example is tert-butyl chloride ((CH3)3CCl). The carbon attached to the chlorine is bonded to three separate methyl groups. That’s three carbons it’s directly connected to!
Tertiary halides are the most stable of the three because of this surrounding carbon power. The ‘electron-donating’ effect of those extra carbons helps to stabilize the positive charge that can form on the carbon when the halogen leaves. They’re the ones who are a bit more hesitant to react unless the conditions are just right. They’ve got a lot to lose, so to speak!
Beyond the Basics: Allylic and Benzylic Halides
Now, sometimes, the location of the halogen relative to a double bond or an aromatic ring gives it some extra oomph. These are special cases, and they are super important in organic chemistry. Think of them as having superpowers!
Allylic Halides
An allylic halide has a halogen atom attached to a carbon atom that is directly next to a carbon-carbon double bond. So, the carbon holding the halogen is adjacent to a C=C. It’s like the halogen is chilling right next to the party zone!
The structure looks something like:
C=C–CH2–X
Or:
C=C–CH(X)–R
Or even:
C=C–C(X)(R)(R’)
The key is that the carbon bonded to X is attached to a carbon that’s part of a double bond.
Why is this cool? Because that double bond is full of electrons, and those electrons can influence the carbon bearing the halogen. It makes allylic halides particularly reactive. They’re prone to reactions where the halogen can be easily replaced, and the double bond can rearrange or participate. They’re like the social butterflies of the halide world, ready to mingle!
An example is allyl chloride (CH2=CH–CH2Cl). See? The chlorine is on a carbon that’s right next to the C=C double bond.
Benzylic Halides
Similar to allylic halides, benzylic halides have a halogen atom attached to a carbon atom that is directly attached to an aromatic ring (like a benzene ring). It doesn’t have to be on the ring itself, but on a carbon that’s connected to the ring.
Think of it like this:
Ring–CH2–X
Or:
Ring–CH(X)–R

Or even:
Ring–C(X)(R)(R’)
The crucial part is that the carbon with the halogen is directly bonded to a carbon that’s part of the aromatic system. That aromatic ring is super stable and has a special electron cloud.
This arrangement also makes benzylic halides very reactive. The stability of the aromatic ring helps to stabilize the intermediates formed during reactions, making them more likely to happen. Benzylic halides are like the sophisticated cousins of allylic halides, with the elegance of the aromatic ring giving them extra flair and reactivity.
A perfect example is benzyl chloride (C6H5CH2Cl). The CH2Cl group is attached to the benzene ring (C6H5).
The "It Depends" Cases: Vinyl and Aryl Halides
Now, these next two are a bit like the rebels of the halide group. Their classification is a little different, and they tend to be less reactive than you might expect. They’re the ones who say, "Nah, I'm good, I like it here."
Vinyl Halides
A vinyl halide has a halogen atom directly attached to a carbon atom that is part of a carbon-carbon double bond. So, the halogen is directly on the double bond itself. No neighbors here, it's right there in the thick of it.
The structure is like:
C=C(X)–R
Or:
C=C(X)(R’)
See how the ‘X’ is directly on one of the carbons involved in the double bond?
These are called vinyl halides because they're related to the vinyl group (CH2=CH–). Examples include vinyl chloride (CH2=CHCl), the building block for PVC! Pretty important stuff, huh?
The reason they’re less reactive is because the C=C double bond is strong, and the bond between the carbon and the halogen gets a bit of a 'partial double bond' character due to electron sharing. It’s like the halogen is really, really comfortable where it is and doesn't want to move. Breaking that bond is tough!
Aryl Halides
An aryl halide has a halogen atom directly attached to a carbon atom that is part of an aromatic ring. This means the halogen is directly on the benzene ring or another aromatic system.
Like this:
Ring–X
Where ‘Ring’ is the aromatic system.
The classic example is chlorobenzene (C6H5Cl). The chlorine is directly attached to the benzene ring.
Just like vinyl halides, aryl halides are generally unreactive under typical conditions. The aromatic ring is super stable, and the direct attachment makes the carbon-halogen bond very strong and resistant to breaking. These guys are content with their aromatic lifestyle and aren’t looking to switch things up easily. They're the sophisticated homebodies.
Putting It All Together: The Cheat Sheet!
So, how do you pick the right classification? It’s a bit like being a detective. You look at the molecule and ask yourself a few key questions:
- Is there a halogen (F, Cl, Br, I)? If not, it’s not a halide, and we can all go home and eat cookies.
- What is the halogen attached to? Is it a carbon atom?
- If it’s a carbon, how many other carbon atoms is that carbon attached to? This gives you primary (1°), secondary (2°), or tertiary (3°).
- Is that carbon atom (the one with the halogen) next to a double bond? If yes, and the halogen is on the adjacent carbon, it’s allylic.
- Is that carbon atom directly attached to an aromatic ring? If yes, and the halogen is on the adjacent carbon, it’s benzylic.
- Is the halogen directly on a carbon that’s part of a double bond? If yes, it’s a vinyl halide.
- Is the halogen directly on a carbon that’s part of an aromatic ring? If yes, it’s an aryl halide.
Sometimes, a molecule might fit into more than one general category. For example, a tertiary halide could also be a benzylic halide if its structure aligns. In such cases, the more specific classification (like benzylic) often takes precedence because it highlights a particular type of reactivity. Think of it as a nickname that's more descriptive than just 'person'.
The key is to look at the immediate environment of the carbon atom bonded to the halogen. Is it surrounded by other carbons? Is it near a pi system (double or triple bond, aromatic ring)? These clues will guide you to the correct classification.
And there you have it! You’ve navigated the fascinating landscape of halide classifications. From the simple primary, secondary, and tertiary to the super-powered allylic and benzylic, and the stoic vinyl and aryl halides, you now have the tools to sort them all out.
Remember, understanding these classifications isn’t just about memorizing terms; it’s about unlocking the secret language of chemical reactivity. It helps chemists predict how these molecules will behave, what reactions they’ll participate in, and how we can use them to create all sorts of amazing things, from medicines to materials.
So next time you encounter a halide, don't just see a bunch of letters and numbers. See a molecule with a personality, a role to play. And that, my friends, is the truly exciting part of chemistry. You’re not just learning facts; you’re learning to read the stories molecules have to tell. Keep exploring, keep questioning, and keep that spark of curiosity alive. The world of chemistry is waiting for you to discover its wonders, one classification at a time. And that, I think, is something to smile about!
