Select All The Intermolecular Forces Associated With Solid Nacl Salt.

Hey there, salt lovers! Did you know that the humble grain of NaCl, the stuff that makes your popcorn pop and your fries fly, is actually a tiny, bustling metropolis of magical forces? Yep, that's right! Even in its solid form, sitting innocently in your salt shaker, it's a whole world of attraction going on.
Think of it like a really, really, really well-behaved party. Everyone’s invited, and everyone’s sticking together. But what’s holding them all in this giant, crystalline hug? It’s not just a handshake, oh no.
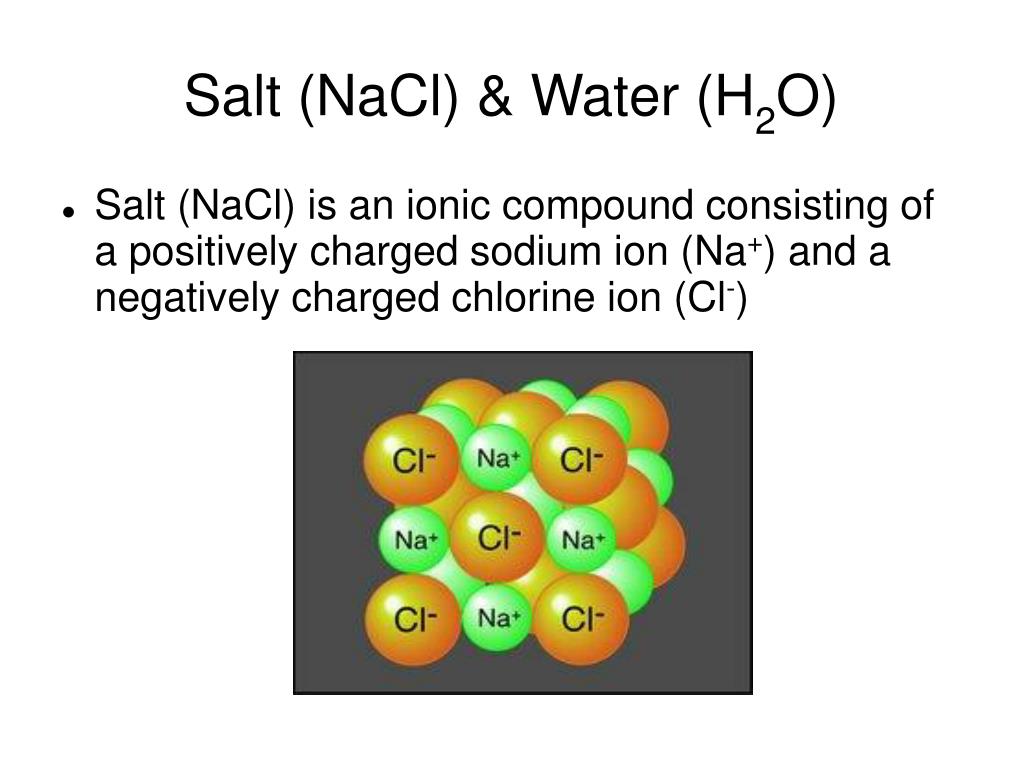
The biggest and most important guest at this party is the ionic bond. Imagine a super-strong magnets, but instead of iron, we've got little charged particles. We've got positively charged sodium ions (think of them as the cheerful, outgoing folks) and negatively charged chloride ions (these are the slightly more reserved, but equally important ones). They are so attracted to each other, like two best friends who just can't get enough of each other's company.
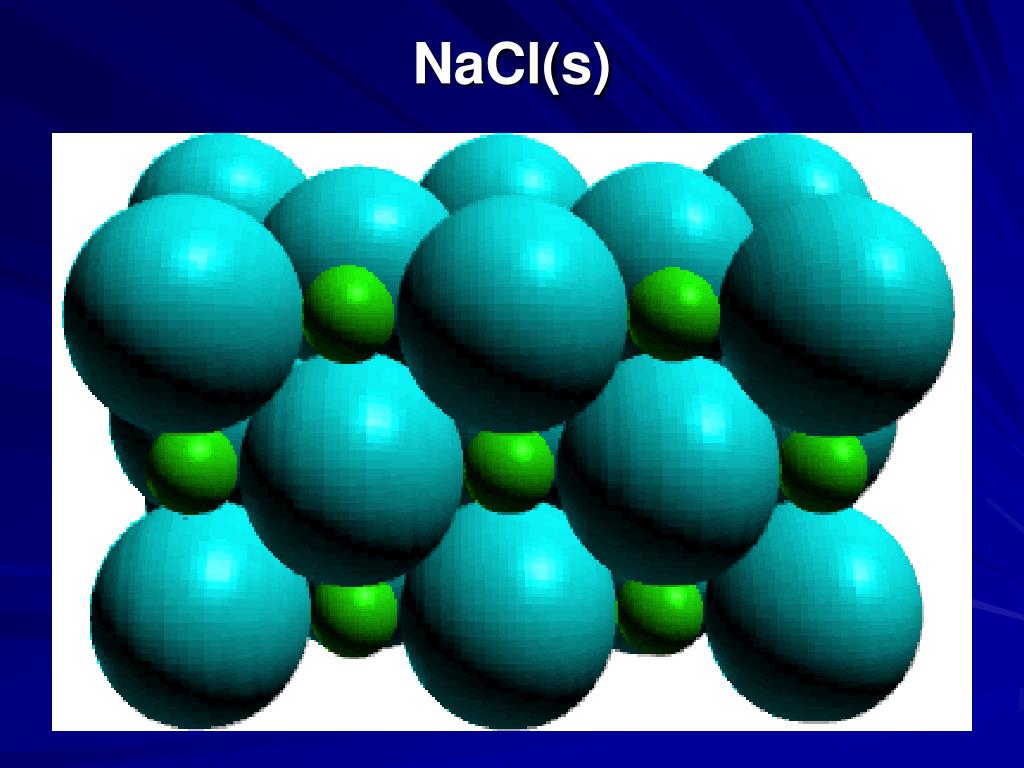
These sodium and chloride ions aren't just hanging out randomly. They arrange themselves in a super orderly fashion, forming a crystal lattice. It's like they've got a perfectly choreographed dance routine they do, all day, every day.
But wait, there's more! Even though the ionic bond is the main event, there are some quieter, more subtle forces at play too. These are the intermolecular forces, and they're like the background music at our salt party. They might not be as loud as the ionic bond, but they’re definitely there, adding to the overall vibe.
One of these subtle players is the dipole-dipole interaction. Now, you might think salt is neutral, and it is overall. But those individual sodium and chloride ions are charged. This creates little "poles" of charge within the crystal.

Think of it like a tiny tug-of-war. The positive ends of one ion are nudging the negative ends of another. It's a gentle pull, a friendly nudge, keeping everything cozy and close. It’s like when you’re in a crowd, and even if you don’t know everyone, there’s still a general sense of closeness and connection.
And then we have the unsung hero, the London dispersion force, sometimes called induced dipole-induced dipole forces. This one is a bit more shy. It’s like the quiet observer at the party, the one who might not say much but is still contributing to the atmosphere.
These forces arise from the constant jiggling and wiggling of electrons within the ions. Even though the ions are generally stable, their electrons can momentarily shift, creating tiny, fleeting imbalances of charge. These tiny fluctuations can then induce similar fluctuations in neighboring ions.
It’s like a ripple effect. One tiny electron wobble makes a neighbor wobble a tiny bit, and so on. It’s not a strong pull, but over millions and millions of ions, these little nudges add up. They’re the tiny whispers that keep the whole structure from flying apart.
So, when you look at a grain of salt, remember it’s not just a simple solid. It’s a vibrant, interconnected community held together by a complex web of attractions. The powerful ionic bonds are the main glue, the foundation of this crystalline city.
But the gentle dipole-dipole interactions and the subtle London dispersion forces are like the friendly nods and shared smiles that make this community truly stable and complete. They are the invisible threads that weave the fabric of the salt crystal.
It’s a testament to the fascinating world of chemistry, where even the most common things have hidden depths and surprising stories. The next time you reach for the salt shaker, give a little nod to these invisible forces. They’re doing a pretty amazing job keeping your seasoning in line!
Isn’t it wonderful that something so simple can be so complex and interesting? These forces are what give salt its solid structure, its ability to dissolve in water (that’s a whole other adventure!), and its unique properties. It's a tiny universe in every crystal.
So, to recap, in solid NaCl, we have a few key players keeping the peace. We’ve got the superstar ionic bond, the undisputed champion of attraction. Then, we have the charming dipole-dipole interactions, adding a bit of subtle charm.

And let’s not forget the ever-present, though often overlooked, London dispersion forces, the quiet guardians of the crystal. They all work together in perfect harmony, like a well-oiled machine, or perhaps more fittingly, a perfectly seasoned dish.
It’s like a microscopic city where everyone is connected, contributing to the overall strength and stability of their home. This intricate dance of attraction is what allows salt to be a solid, a fundamental part of our kitchens and our lives. Without these forces, salt would just be a confused pile of separate, unattached particles.
So, when you sprinkle that magical white stuff on your food, imagine the tiny, invisible party happening within each grain. Cheers to NaCl and all the marvelous forces that hold it together! It’s a little bit of everyday magic.
