Select All Of The Atoms Which Bear Lone Electron Pairs.
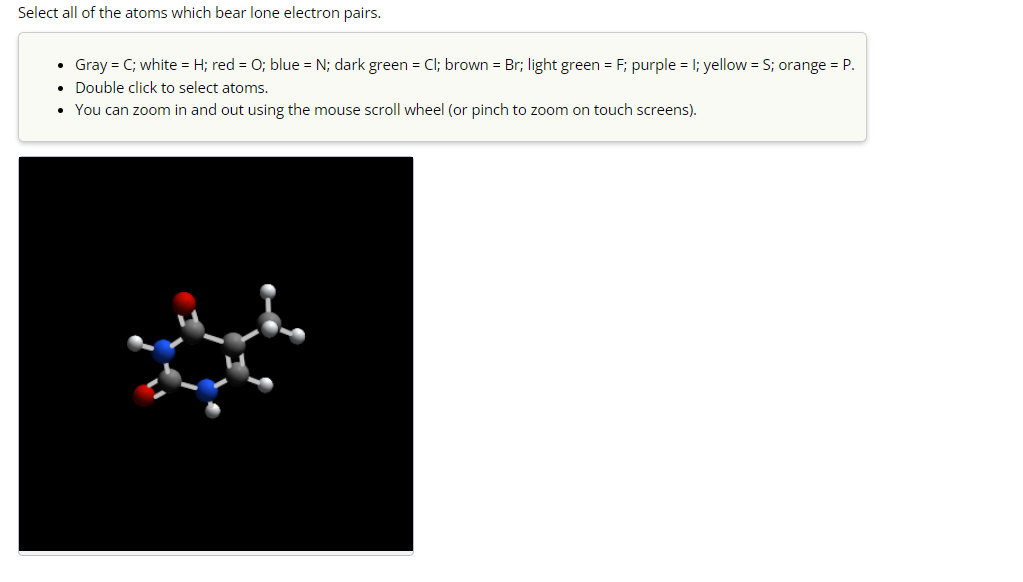
Hey there, science curious folks! Ever peeked at a molecule diagram and seen those little dots hanging out around some of the atoms? Those aren't just random decorations, oh no. They're actually tiny bundles of electron power called lone electron pairs, and they're the secret sauce behind a whole lot of fascinating chemistry. Today, we're going on a chill exploration of these hidden electron treasures. Think of it like a treasure hunt, but instead of gold doubloons, we're digging for lone pairs!
So, what exactly are these lone pairs? Imagine atoms as little characters in a cosmic play. They've got electrons, which are like their energy costumes. Most of the time, these electrons are busy holding hands with other atoms, forming the bonds that create everything around us – the air we breathe, the water we drink, even your comfy couch. But sometimes, an atom has a few leftover electrons that aren't playing nice and forming bonds. These are our lone pairs, chilling on their own, just waiting for something interesting to happen.
Why Should We Even Care About These Lone Pairs?
You might be thinking, "Okay, so some electrons are single. Big deal!" But trust me, these singletons are absolute game-changers. They're like the unexpected plot twists in your favorite movie. These lone pairs have a bit of a reputation for being… well, a little reactive. They've got this pent-up energy, and they love to get involved in chemical reactions. It's like they're always ready to jump into the mosh pit of molecular interactions!
Think about it like this: you know those quiet, unassuming folks who suddenly have the most insightful comment or the most brilliant idea? Lone pairs can be a bit like that. They might seem passive, but they hold immense potential for influencing how molecules behave and interact. They're the unsung heroes of chemical transformations.
The "Select All Of The Atoms Which Bear Lone Electron Pairs" Challenge
Now, the big question is, how do we find these lone pairs? It’s like playing a game of "Where's Waldo?" but with electrons. Usually, in chemistry, we're looking at Lewis structures, which are basically little drawings that show how atoms are connected and where their electrons are. When you draw a molecule, you'll see symbols for atoms, lines representing bonds (those are shared electron pairs), and then, bam! Those little dots hanging out around an atom? Those are your lone pairs.
To "select all of the atoms which bear lone electron pairs," you're essentially scanning these Lewis structures and ticking off the atoms that have these unbonded pairs of electrons. It's a visual task, really. You're looking for the atoms that have those little dots, usually in pairs, that aren't part of a bond line.
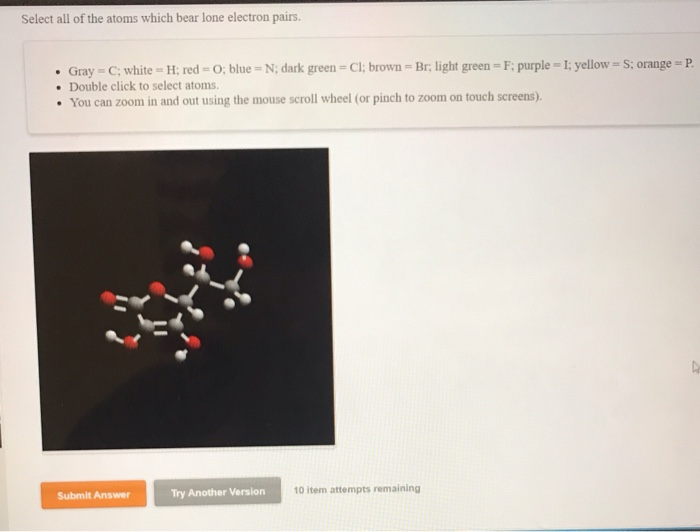
Let's Meet Some Lone Pair Enthusiasts!
So, which atoms are typically the ones you'll find sporting these cool electron accessories? Some common culprits include:
Oxygen (O): Oxygen is a bit of a lone pair champion. In molecules like water (H₂O), the oxygen atom has two lone pairs. It's like oxygen shows up to the party with two extra sets of groovy sunglasses. These pairs are super important for water's ability to dissolve things and its unique properties. Without those lone pairs, water might just be… well, a lot less interesting!
Nitrogen (N): Nitrogen, the main component of our atmosphere, also loves to carry lone pairs. In ammonia (NH₃), the nitrogen atom has one lone pair. This lone pair is key to ammonia's basicity, meaning it can readily accept protons (which are just hydrogen ions). It's like nitrogen is holding a little sign saying, "Protons welcome here!" This ability makes it crucial in biological processes and industrial applications.
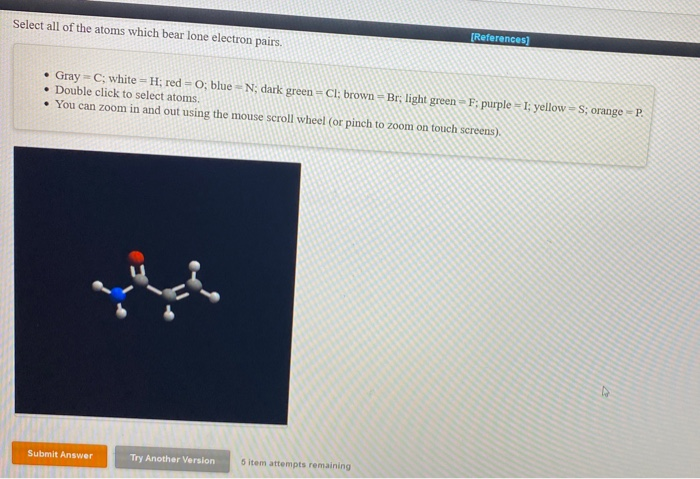
Halogens (F, Cl, Br, I): These elements, like fluorine, chlorine, bromine, and iodine, are practically drowning in lone pairs when they're not busy forming bonds. For instance, in a hydrogen halide molecule like HCl, the chlorine atom will have three lone pairs. That's a whole lot of unshared electron energy! They're like the shy, but powerful, kids on the block who can really throw a punch when needed.
Carbon (C) - Sometimes: Carbon is usually quite the team player, forming four bonds to satisfy its electron needs. However, in certain special cases, like in carbanions (where a carbon atom has a negative charge), it can end up with a lone pair. This is a bit less common but still a neat example of carbon’s versatility.
The Bigger Picture: Why Lone Pairs Matter So Much
These lone pairs aren't just hanging out for decoration. They have profound implications for how molecules behave. Their presence influences:

Molecular Shape: Those lone pairs take up space, and they don't like to be too close to other electron clouds (including other lone pairs and bonding pairs). This repulsion pushes and pulls on the bonds, dictating the molecule's three-dimensional shape. Think of it like trying to arrange a bunch of balloons in a tight space – they'll push each other around until they find a stable configuration. The shape of a molecule is super important because it determines how it interacts with other molecules, like how a lock fits a key.
Reactivity: As we touched on, lone pairs are often the launching pads for chemical reactions. They can be attracted to positively charged areas on other molecules or can even donate their electron density to form new bonds. They're the spark that ignites the chemical firework show!
Acidity and Basicity: The ability of an atom to accept or donate protons is heavily influenced by its lone pairs. Atoms with lone pairs that are readily available to bond with a proton are considered basic. Conversely, atoms that can stabilize a negative charge (often due to the presence of lone pairs) can influence acidity.

A Fun Analogy Time!
Imagine a dance floor. The bonded electrons are the couples dancing together, locked in a partner's embrace. The lone pairs are the dancers who are taking a break, standing around the edges of the dance floor. They might not be dancing, but they're observing, they're ready to jump in, and their presence influences the overall energy and flow of the room. Sometimes, a lone pair dancer might even tap a bonded dancer on the shoulder, initiating a new dance formation!
Or think about a social gathering. Most people are engaged in conversations (bonded electrons). But there are those individuals who are circulating, observing, and sometimes initiating new interactions – those are your lone pairs. They have the potential to change the dynamics of the whole party.
The Takeaway
So, the next time you see those little dots on a Lewis structure, don't just dismiss them as extra clutter. They're indicators of lone electron pairs, the energetic, reactive, and often misunderstood players in the world of chemistry. They are the secret ingredients that give molecules their unique personalities and drive the incredible transformations we see all around us.
Learning to "select all of the atoms which bear lone electron pairs" is a foundational step in understanding the 'why' behind so many chemical phenomena. It's about recognizing where the potential for action lies within a molecule. It's a key piece of the puzzle in unlocking the secrets of chemical behavior. Pretty cool, right? Keep those curious eyes peeled for those lone pairs – they're where the real magic often happens!
