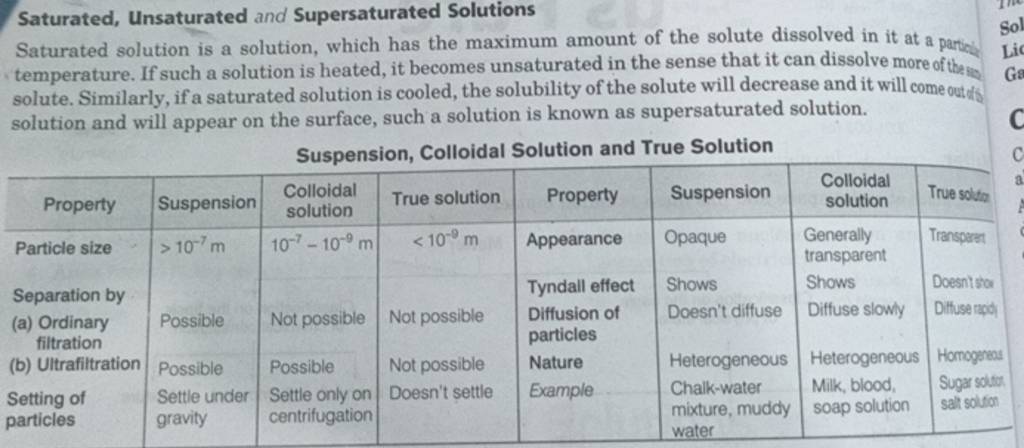
Saturated And Unsaturated Solutions Pogil Answers

Alright, settle in, grab your latte, and let's dive into a topic that sounds about as exciting as watching paint dry, but I promise, it’s way more fun than it sounds. We're talking about saturated and unsaturated solutions, or as I like to call them, the "stuff-holding-capacity Olympics" of the liquid world. Ever wondered why your Kool-Aid sometimes tastes like pure sugar water and other times like… well, just water? This is your answer, and it’s more dramatic than you think!
Imagine you've got a mug of hot water. It’s like a tiny, friendly apartment building for all sorts of little molecules. You start tossing in sugar cubes, one by one. At first, they happily dissolve, zipping around, making your drink all sweet and delightful. This is your unsaturated solution. Think of it as a party where there's plenty of room on the dance floor for everyone. There’s still plenty of space for more sugar to crash the party and dissolve without a fuss.
Now, keep adding sugar. Eventually, you’ll reach a point where you drop in another sugar cube, and it just… sits there. At the bottom of your mug, like that awkward guest who didn’t get the memo and is now just awkwardly standing in the corner. This, my friends, is the glorious moment you’ve created a saturated solution. Your apartment building is officially at full capacity. No more dancing room, no more dissolving guests. Anything you add from here on out will be the social pariah of the mug, the insoluble outcast.
So, what’s the magic number? The amount of "stuff" (we scientists call it a solute, fancy, right?) that can dissolve in another "stuff" (the liquid, or solvent) is called the solubility. It’s like each solvent has a personal limit, a personal space bubble for solutes. And guess what? This solubility isn’t a constant. It's a bit of a drama queen, and its mood swings are often dictated by temperature.
Generally, most things are like eager partygoers – they dissolve way better when things are heated up. Think of that hot chocolate you love. You can dump way more cocoa powder into hot milk than you ever could into cold milk, right? That’s because the hotter temperature gives the solvent molecules more energy to wiggle and jiggle, making more room for the solute molecules to come in and make themselves at home. It’s like the apartment building landlords turning up the disco music, making everyone dance faster and creating more chaotic, but ultimately more dissolved, space!

But wait, there’s a plot twist! Sometimes, things get really fancy. We can actually push the limits. Imagine you’ve made your saturated solution at a high temperature, and then you very carefully cool it down. If you’re super gentle, the solute might not realize the party’s over and it’s supposed to leave. It’ll hang around, even though there’s technically no room left. This is called a supersaturated solution. It’s like the ultimate, overcrowded, possibly-about-to-explode party. It’s inherently unstable, teetering on the edge of a solute-induced riot.
One tiny nudge, one little shake, or even dropping in a single, solitary crystal of the solute (which we call a seed crystal – think of it as the bouncer finally deciding enough is enough), and BAM! All the excess solute that wasn’t supposed to be there suddenly decides to leave the dance floor. They clump together, crystallize, and make a dramatic exit, often as a visible solid at the bottom. It’s the ultimate social media drama, playing out in your mug. One minute it's all a cozy, jam-packed party, the next it’s a mass exodus.

So, why should you care about this whole saturated, unsaturated, supersaturated hullabaloo? Well, besides understanding why your rock candy might be hiding more sugar than you expected, it has some real-world applications. Think about making rock candy itself. You create a supersaturated sugar solution, and as it cools, the sugar crystallizes out, forming those beautiful, sparkly gems. It’s basically controlled chaos for sweet treats!
Or consider making tea. If you’ve ever tried to steep a tea bag in lukewarm water, you’ll know it’s a sad, watery experience. Hot water is crucial because it can dissolve more of those tasty flavor compounds from the tea leaves. More dissolved flavor = a more delicious cuppa. It’s the solvent capacity of the water flexing its muscles!

Even in nature, this stuff is going on! Think about how minerals form in caves, creating stalactites and stalagmites. Water seeps through rocks, dissolving minerals. When the conditions change – maybe the water drips, or evaporates – it can lead to saturated or even supersaturated solutions, causing those minerals to precipitate out and build these incredible formations over millennia. So, next time you’re in a cave, you can whisper to the stalactites, "Ah, you too, were a victim of solubility limits!"
The POGIL (Process Oriented Guided Inquiry Learning) worksheets you might encounter this in are designed to make you discover these concepts yourself. They’re like little science treasure hunts where you’re the detective. You’ll be doing experiments, observing what happens, and piecing together the puzzle of why some things dissolve and others don't, and how temperature throws a wrench (or a helping hand) into the whole process. Instead of me just telling you, you get to be the "Eureka!" moment person. It's way more satisfying than just memorizing a definition, isn't it?
So, to recap our café chat: * Unsaturated: Plenty of room for more solute. The party is just getting started. * Saturated: No more room. The dance floor is full. Any more solute is just going to stand around awkwardly. * Supersaturated: The illegal, overcrowded, potentially explosive party. A tiny disturbance will cause a mass eviction.
Remember, the key players are the solute (the stuff being dissolved), the solvent (the liquid doing the dissolving), and solubility (the maximum amount of solute that can dissolve at a given temperature). And don't forget temperature, that fickle friend who can either boost solubility or make things dramatically fall out of solution. It’s all about how much the solvent can handle, and sometimes, how much it can pretend to handle before reality (and crystallization) sets in.
So, the next time you’re stirring sugar into your coffee, or admiring a rock candy, give a little nod to the fascinating world of saturated and unsaturated solutions. It’s a simple concept with a surprising amount of drama and utility. Now, who wants another coffee? I think my brain is now saturated with scientific facts and needs some caffeine to become unsaturated again. See? It all comes full circle!
