Rutherford's Nuclear Theory As It Was Originally Stated

Hey there, science adventurer! Ever heard of Rutherford? Ernest Rutherford. This guy was like the rockstar of early 20th-century physics. He wasn't just some stuffy dude in a lab coat. Oh no. He was a bit of a character, and his big idea? Totally blew people's minds.
So, picture this. It’s way back in the day, before we had smartphones and instant noodles. Scientists basically thought atoms were just... well, lumps. Like tiny, invisible, positively charged Jell-O molds. Everything was smooth, solid, and predictable. Pretty boring, right?
Then came Rutherford and his buddies, Geiger and Marsden. These guys were doing experiments. And let me tell you, their experiments were way cooler than anything you'd find on TikTok. They decided to shoot tiny, positively charged particles, called alpha particles, at a very, very thin sheet of gold foil. Think of it like throwing microscopic paintballs at a piece of aluminum foil. Easy peasy, right?
They expected the alpha particles to just whiz right through the gold. Like a hot knife through butter. They figured, "Yeah, it's gold, it's thin, they'll just go straight on through. No biggie." But science, as it often does, decided to be a mischievous little imp.
Most of the alpha particles did go straight through. Phew! Crisis averted. The lump theory was looking pretty good. But then, something… weird happened.
A tiny fraction of the alpha particles, like, a really, really small number, didn't go straight. Some got deflected. Not just a little nudge, but some got sent off at weird angles. And then, the kicker. A few of them, get this, actually bounced straight back. Like a boomerang! Imagine throwing a marshmallow at a brick wall and having it bounce right back into your face. You'd be like, "What in the actual proton just happened?!"
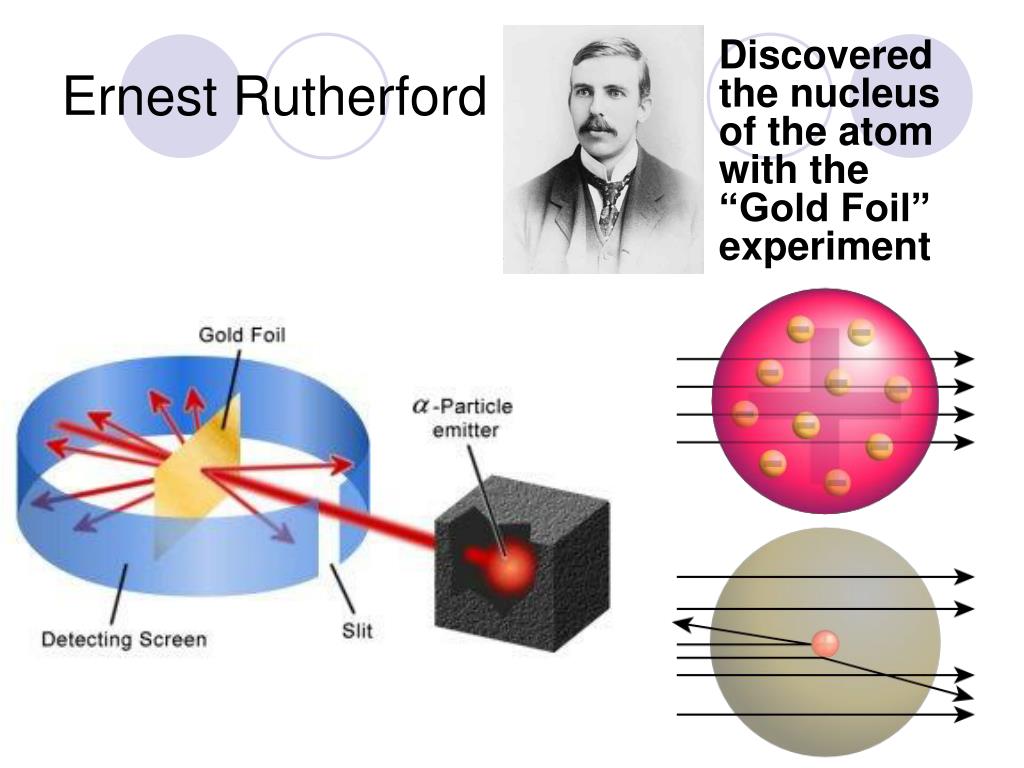
Rutherford himself famously said it was "quite the most incredible event that has ever happened to me in my life. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you." Tissue paper! He was that shocked. You can practically hear the cogs turning in his brilliant, slightly singed, brain.
So, what did this mean? The lump theory? Dead in the water. If atoms were just evenly spread lumps of positive charge, those alpha particles should have sailed through like they were in a ghost convention. But they didn't. Something else was going on inside those atoms.
Rutherford, being the clever cookie he was, started piecing it together. He realized that for a few particles to get deflected so drastically, and some even bounce back, there had to be something in the atom that was really, really dense and positively charged. Something tiny, but with a serious punch.

He proposed that the atom wasn't a lumpy Jell-O at all. Instead, it was mostly empty space. Yep, you heard that right. Most of an atom is just… nada. Zip. Zilch. An impressive amount of nothingness!
And at the very center of this vast emptiness, he imagined a super-tiny, super-dense core. This core, he said, held all the positive charge and almost all of the atom's mass. He called this the nucleus. Pretty catchy, right?
And where were the negatively charged electrons? Well, they were zipping around this nucleus. Kind of like planets orbiting a sun, but way, way smaller and way, way faster. He called this the "nuclear model" of the atom. It was a total game-changer.
So, Rutherford's theory, as he first stated it, basically boiled down to this:

The Nuclear Theory of the Atom: Rutherford's Original Thoughts
1. Atoms are mostly empty space. Seriously, like 99.99999% empty. Mind-boggling, isn't it?
2. There's a tiny, dense, positively charged core at the center of the atom. This is the nucleus. It’s where all the good stuff (mass and positive charge) is packed in.
3. The negatively charged electrons whiz around this nucleus, occupying the vast expanse of empty space. Think of them as tiny, energetic race cars on a giant, empty racetrack.
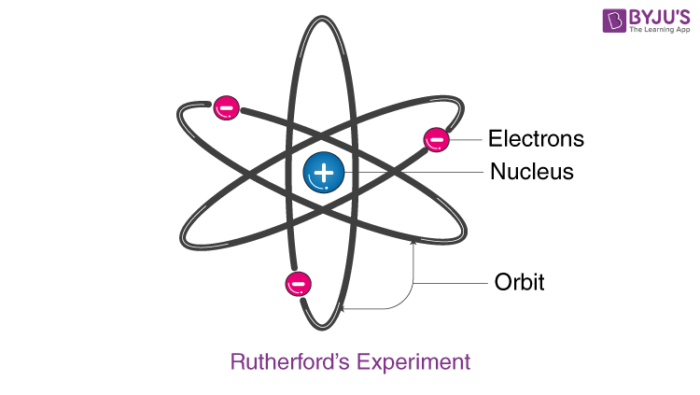
This was HUGE. Before Rutherford, the atom was like a featureless blob. Now, it had structure. It had a heart. A tiny, powerful heart. It was like discovering a whole hidden city inside what you thought was just a single building.
And the best part? This whole idea came from a little bit of gold foil and some really persistent alpha particles. It wasn't some grand, philosophical debate. It was good old-fashioned experimentation and a willingness to say, "Okay, what I thought was true… isn't."
It’s the kind of scientific discovery that makes you want to go out and poke things with a stick (safely, of course!). It shows that even when you think you've got it all figured out, the universe is always ready to surprise you. And sometimes, the most amazing discoveries come from expecting one thing and getting something completely, wonderfully, unexpectedly different.
So next time you’re looking at something, anything, remember that it’s all made of these tiny, mostly empty buildings with incredibly dense, energetic centers. It’s a wild thought, and it all started with Ernest Rutherford and a little bit of gold foil magic. Pretty cool, huh?
