Reduction Of Benzophenone With Sodium Borohydride Discussion

Hey there, lab buddies! Let's chat about something super cool that happens in the wonderful world of chemistry: the reduction of benzophenone with sodium borohydride. Sounds a bit fancy, right? But trust me, it's actually pretty straightforward and, dare I say, a little bit fun.
So, imagine you’ve got this molecule, benzophenone. It's got a couple of phenyl rings attached to a carbonyl group. Think of it like a little molecular handshake between two fancy benzene rings, all holding hands with a double-bonded oxygen. Pretty neat, eh?
Now, what’s a “reduction” in chemistry terms? It’s basically like giving a molecule a little bit of extra electron-y goodness. Think of it as the opposite of oxidation, where a molecule might lose electrons. In this case, we're going to add some electrons to that carbonyl group in benzophenone. And guess who’s the superhero of this electron-adding mission? That’s right, sodium borohydride (NaBH₄)!
Sodium borohydride is like the gentle giant of reducing agents. It's super useful because it's not as aggressive as some of its more hardcore buddies (like lithium aluminum hydride, which is a whole other story and can be a bit of a drama queen in the lab!). NaBH₄ is known for being relatively safe and easy to handle, which is always a win in my book. After all, we want to make cool stuff, not become the cool stuff, right?
So, the basic idea is that sodium borohydride has these special “hydride” ions (that’s a hydrogen atom with an extra electron, H⁻). These hydride ions are the ones doing the heavy lifting. They’re like little microscopic delivery trucks, bringing their electron cargo to the party.
When you mix benzophenone with sodium borohydride, usually in a suitable solvent like ethanol or methanol (think of these as the cozy little rooms where our reaction can hang out), something magical happens. The hydride ion from NaBH₄ is attracted to the positively charged carbon atom in the benzophenone’s carbonyl group. Remember, that double-bonded oxygen is a bit of an electron hog, so it makes the carbon it's attached to a little bit electron-deficient, or electrophilic, if you want to get technical.
The hydride ion, with its extra electron, swoops in and attacks that electron-hungry carbon. Poof! The double bond between the carbon and oxygen breaks, and the hydrogen from the hydride ion attaches itself to the oxygen. The carbon now has a single bond to the oxygen, and a new hydrogen attached to it. The oxygen, being a bit of a diva, usually grabs a proton (a hydrogen ion, H⁺) from the solvent later on, completing the transformation.
The Grand Transformation: Benzophenone to Benzhydrol
What do we get at the end of this whole kerfuffle? We get benzhydrol! Or, if you want to be more formal, diphenylmethanol. This is an alcohol. And what’s an alcohol? It’s a molecule with an –OH group. So, that double-bonded oxygen in benzophenone has now become a lovely hydroxyl group in benzhydrol. It’s like benzophenone went through a spa day and came out as a more hydrated, more hydroxylated version of itself!
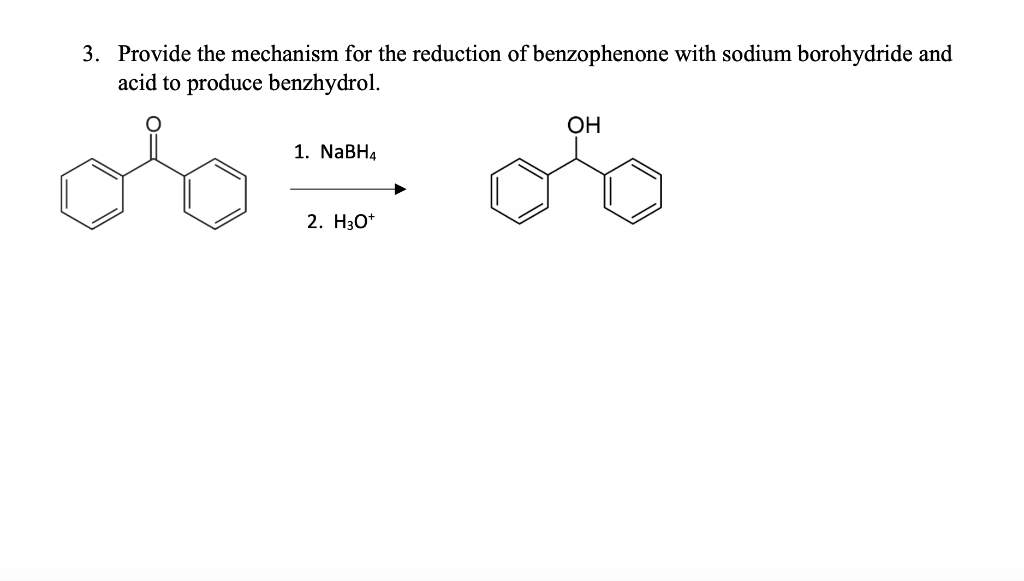
Think of it this way: benzophenone is a ketone. Ketones have a carbonyl group where the carbon is bonded to two other carbon atoms. Alcohols, on the other hand, have a hydroxyl group attached to a carbon. So, we’ve essentially converted a ketone into a secondary alcohol. Secondary means that the carbon bearing the –OH group is attached to two other carbon atoms. Pretty neat, right?
The beauty of using sodium borohydride for this is its selectivity. It's really good at reducing carbonyl groups (like the one in ketones and aldehydes) but it tends to leave other functional groups alone. So, if your benzophenone had, say, an ester or a nitro group hanging around, NaBH₄ would probably ignore them, letting them do their own thing. This is what chemists call chemoselectivity, and it’s super important when you're trying to make specific molecules without accidentally messing up other parts.
Why is this Reaction So Cool? (Besides the Cool Name)
Okay, so beyond the satisfying chemical transformation, why do we even bother with the reduction of benzophenone? Well, for starters, it's a classic example that’s often used in introductory organic chemistry labs. It’s a fantastic way to learn the ropes of using reducing agents, understanding reaction mechanisms, and practicing purification techniques. You get to see firsthand how a little bit of reagent can completely change the nature of a molecule.
Plus, benzhydrol itself is a pretty useful building block. It's an intermediate in the synthesis of various pharmaceuticals and other organic compounds. So, this seemingly simple reaction is actually a stepping stone to creating more complex and valuable molecules. It's like learning your ABCs before you can write a novel!
And let's not forget the joy of watching a reaction happen! Sometimes, you can see the benzophenone (which is usually a solid) dissolve, and then after adding the NaBH₄ and a bit of stirring, you might see the benzhydrol precipitate out or get it by quenching the reaction and isolating it. It's like a little chemical magic show happening in your flask.

The Nitty-Gritty: A Peek Under the Hood
For those of you who love a bit more detail (you know who you are!), let's dive a little deeper into the mechanism. It’s not super complicated, I promise!
Step 1: Hydride Attack. As we mentioned, the hydride ion (H⁻) from NaBH₄ acts as a nucleophile. It attacks the electrophilic carbon of the carbonyl group in benzophenone. This breaks the pi bond (the weaker of the two bonds in the double bond) and forms a new carbon-hydrogen sigma bond. The oxygen atom now carries a negative charge, making it an alkoxide intermediate.
Step 2: Protonation. The alkoxide intermediate is quite reactive. When the reaction mixture is quenched with an acid (like dilute HCl or even just water, if the solvent is protic like ethanol), the negatively charged oxygen atom picks up a proton (H⁺) from the acid or solvent. This forms the final alcohol product: benzhydrol.
The sodium ion (Na⁺) from the sodium borohydride just chills out as a spectator ion for most of the reaction, forming ionic bonds with the negatively charged intermediates and then with the leaving groups. It’s the unsung hero, providing the counter-ion support!
Safety First (Always!)
Now, while sodium borohydride is considered relatively safe, it's still a chemical, and we should always treat it with respect. It can react with water (especially acidic water) to release hydrogen gas, which is flammable. So, you’ll always be working in a well-ventilated area or under a fume hood, and avoiding open flames. Lab coats, safety glasses, and gloves are your best friends in the lab – they’re like your superhero costume!

When working with solvents like ethanol or methanol, remember they are also flammable. So, always be mindful of your surroundings and follow proper lab safety protocols. It’s all about being a responsible and awesome chemist!
Tips and Tricks for Success
When you're performing this reaction, a few things can help make it go smoothly:
- Solvent Choice: Ethanol and methanol are common and work well because they can dissolve both benzophenone and sodium borohydride to some extent, and they can also provide the necessary protons for the final step.
- Temperature: Often, this reaction can be done at room temperature. Sometimes, a little cooling might be beneficial, especially if the reaction is very exothermic (gives off a lot of heat), but for benzophenone, room temperature is usually just dandy.
- Stoichiometry: You don't need a huge excess of sodium borohydride. A slight molar excess is usually sufficient to ensure that all the benzophenone is reduced. Don't go overboard; more isn't always better!
- Work-up: After the reaction is complete, you’ll need to "quench" it to destroy any remaining sodium borohydride and then isolate your benzhydrol. This usually involves adding water or dilute acid, and then often extraction with an organic solvent, followed by drying and evaporation.
- Purification: Depending on how pure you need your benzhydrol to be, you might need to purify it further, perhaps by recrystallization or chromatography. This is where you get to play detective and make sure you've got the good stuff!
Don't be discouraged if your first attempt isn't perfect. Chemistry is all about trial and error, learning from mistakes, and celebrating the successes. Every experiment is a learning opportunity, and even a slightly messy outcome teaches you something valuable.
The "Why" Behind the "What"
So, to recap, we’re taking a molecule with a carbonyl group (benzophenone) and using sodium borohydride to add a hydride ion and then a proton, turning it into an alcohol (benzhydrol). This is a fundamental organic reaction that showcases the power of reducing agents and the elegance of chemical transformations. It’s a cornerstone for many more complex syntheses.
It's like learning to tie your shoelaces before you can run a marathon. This simple reduction is a vital skill in the chemist’s toolkit. It allows us to manipulate molecules, creating new compounds with desired properties. It's the bedrock upon which more intricate molecular architectures are built.
Think about all the medicines we rely on, the materials we use every day – many of them start with simple, fundamental reactions like this one. It’s a reminder that even the most complex things often have their roots in something beautifully simple.
A Little Bit of History and Fun Facts
Did you know that sodium borohydride was first synthesized in the 1940s? It was developed by chemist HH{" "}Adler and E.{" "}C.{" "}Stelling as part of research for the Manhattan Project. Talk about a surprising origin story! From nuclear research to everyday organic labs, it's had quite the journey.
And benzophenone itself? It’s a pretty common organic compound. It’s used as a photoinitiator in UV-curing applications, meaning it helps start polymerization reactions when exposed to UV light. So, it’s not just a pretty molecule; it’s got some practical applications too!
The conversion of a ketone to a secondary alcohol is a very common and important reaction in organic synthesis. It’s a fundamental step that allows chemists to modify the properties and reactivity of organic molecules. This ability to finely tune molecular structure is what drives innovation in so many fields, from pharmaceuticals to materials science.
Bringing It All Together: The Joy of Discovery
So, there you have it! The reduction of benzophenone with sodium borohydride. It’s a process that’s both elegant and accessible, a true testament to the power of controlled chemical reactions. It’s a moment where abstract concepts like electron transfer and functional group transformation become tangible realities in a flask.
Every time you perform this reaction, or any reaction for that matter, you’re participating in a long and rich history of scientific exploration. You’re adding your own small piece to the grand puzzle of understanding and manipulating the molecular world. It’s a journey filled with curiosity, challenges, and the immense satisfaction of discovery. So, embrace the process, enjoy the learning, and never forget the sheer wonder of making new things happen in the lab! Go forth and reduce, my friends! And may your yields be high and your purifications be swift!
