Reduction Of 9-fluorenone To 9-fluorenol Mechanism

So, hey! Grab your mug. Let's chat about something kinda cool, right? We're gonna dive into how we take this little guy, 9-fluorenone, and turn it into its cooler, more chill cousin, 9-fluorenol. It's basically like giving it a spa day, you know? A little reduction, a little transformation. Easy peasy, lemon squeezy. Or, you know, maybe not always lemon squeezy, but we'll get there!
Think of 9-fluorenone like a fancy car with a slight dent. It’s got that ketone thing going on – a carbon double-bonded to an oxygen. Pretty standard, right? But 9-fluorenol? That’s the polished version. It’s got an alcohol group, an -OH. Way more friendly, in my opinion. And the magic behind this glow-up? A reduction reaction. Fancy term, I know, but it just means we’re adding electrons. Like giving it a little energy boost. Who doesn’t love a good energy boost?
The "Why" Behind the Transformation
Okay, so why would we even bother with this? Well, sometimes you need that alcohol. Maybe for making other cool chemicals, or for research. It's like having a different Lego brick. You need it to build something specific. And 9-fluorenol is a pretty useful brick. Plus, honestly, it’s a pretty neat demonstration of a fundamental organic chemistry concept. Like, “Ta-da! Chemistry happens!”
It’s not like we’re reinventing the wheel here, but understanding these basic reactions is, like, super important. It’s the building blocks for everything else. Think of it as learning your ABCs before you can write a novel. Except, you know, way more exciting than the alphabet. Definitely more exciting.
The Hero of Our Story: The Reducing Agent
Now, to make this happen, we need a sidekick, a superhero, if you will. And that’s our reducing agent. This is the dude (or dudette, or they!) that’s going to generously donate those electrons. They’re the generous givers of the chemical world. And in the case of 9-fluorenone, a super common and pretty chill reducing agent is sodium borohydride (NaBH4). Ever heard of it? It’s like the friendly neighborhood strongman of the lab. Not too aggressive, but gets the job done.
Other agents exist, of course! There’s lithium aluminum hydride (LiAlH4), which is, like, the super strong dude. Too strong for this particular job, sometimes. Like using a sledgehammer to crack a walnut. But NaBH4? It’s just right. It’s selective. It likes targeting that ketone and leaving other parts of the molecule alone. Smart!
So, NaBH4 is our go-to. It’s relatively safe, easy to handle (compared to its more explosive cousins), and it’s super effective for this specific kind of transformation. Think of it as the perfect tool for the job. You wouldn’t use a screwdriver to hammer a nail, right? No! You use the right tool. And NaBH4 is the right tool for reducing our 9-fluorenone.
The Step-by-Step Action: The Mechanism, Unveiled!
Alright, so let's get down to the nitty-gritty. How does this actually work? It’s not like magic, although sometimes it feels like it! It's a series of well-defined steps. Like a dance. A very, very tiny dance happening at the molecular level.
First off, we gotta get our players in the same room. So, we have our 9-fluorenone, chilling out, maybe feeling a bit stressed with that double bond. And then we introduce our sodium borohydride. In solution, usually water or an alcohol like methanol or ethanol. Gotta have a solvent, right? Think of it as the dance floor. They can’t just waltz around in a vacuum.
So, NaBH4 is this cool little cluster of atoms. It’s got a boron atom in the middle, and four hydrogen atoms attached to it. And here’s the kicker: that boron atom is kinda electron-deficient. It’s like it’s always looking for something. And the hydrogen atoms attached to it? They’re carrying extra electrons. Like little backpacks full of goodies.
Step 1: The Hydride Attack!
This is where the action really starts. One of those hydrogen atoms on the borohydride ion (that’s the [BH4]- part after it dissociates a bit) decides it’s had enough of sitting around. It’s got these electrons, and it sees that carbonyl carbon on the 9-fluorenone. That carbon, the one in the C=O group, is electrophilic. That’s a fancy way of saying it’s a bit positive, or electron-poor. It’s like a magnet for those negative electrons.
So, bam! The hydride ion (H-), with its extra electron baggage, attacks that electrophilic carbonyl carbon. It's a direct assault! The double bond between the carbon and oxygen breaks. The electrons from that double bond, which were shared, now get to spread out a bit. They go onto the oxygen atom. This makes the oxygen atom suddenly have a negative charge. It’s like, “Whoa! What just happened?!”

This first step is the crucial one. It’s the initiation of the whole process. Think of it as the first domino falling. Without this hydride attack, nothing else would happen. It’s the moment of truth!
Step 2: The Borate Intermediate – A Brief Encounter
Now we have this intermediate. It’s not quite fluorenol yet, but it’s getting there. The oxygen has a negative charge, and the boron is now attached to the carbon that used to be part of the double bond. It’s a pretty short-lived stage, this borate intermediate. It’s like a quick handshake before the main event. It’s not super stable, so it’s looking for its next move.
This intermediate is essentially an alkoxide, but with a boron fragment still attached. It’s like a temporary holding pattern. The negatively charged oxygen is still there, and the boron is now bonded to the original carbonyl carbon, and the hydrogen that attacked. So, it’s a new bond formed, and a new negative charge on the oxygen.
Step 3: The Protonation – Giving it a Hug
Our intermediate is still carrying that negative charge on the oxygen. It’s feeling a bit unbalanced. It needs to become neutral and happy again. And how do we do that? We introduce something that can give it a proton (an H+ ion). In our reaction mixture, we usually have excess solvent, which is often an alcohol. And alcohols, remember, have hydrogen atoms attached to oxygen. So, they can donate a proton!
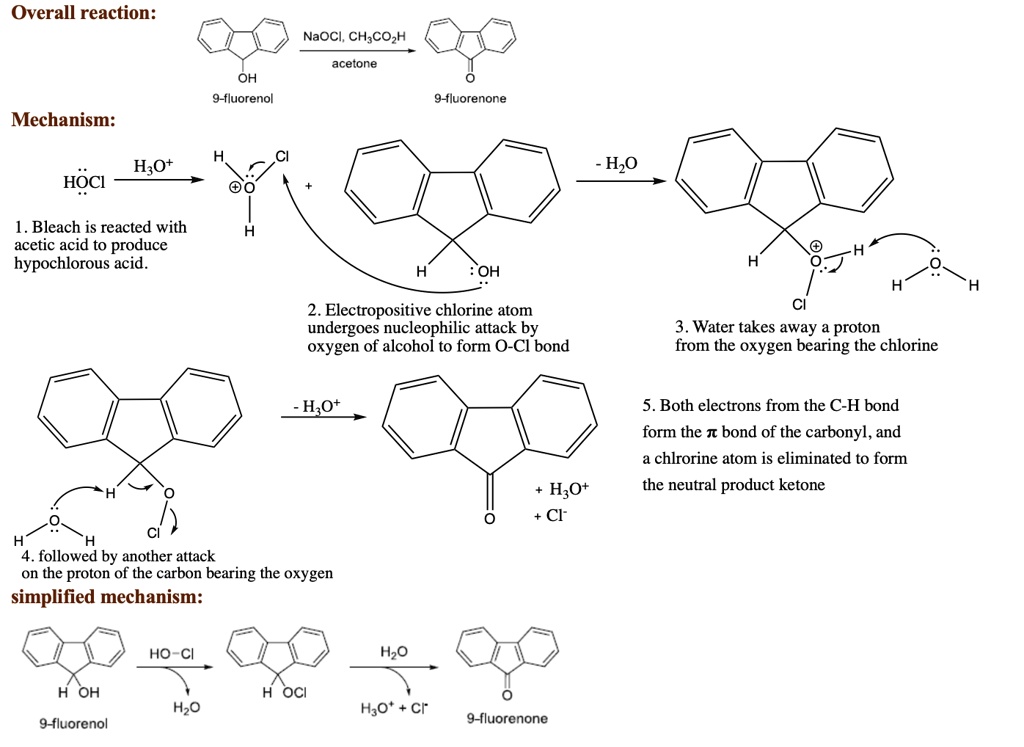
The negatively charged oxygen on our intermediate sees that proton hanging out in the solvent. It’s like, “Hey, you! I need you!” So, it grabs that proton. Poof! The negative charge on the oxygen is neutralized. And what do we end up with? The desired 9-fluorenol! Ta-da! We did it!
The boron part? It usually detaches and might pick up other things from the solvent, but the main goal is achieved: the ketone is turned into an alcohol. It’s a beautiful, elegant dance of electron pushing and proton grabbing. So satisfying to watch, even if you can't actually see it!
The Role of the Solvent – It’s Not Just Decoration!
Now, you might be thinking, “Does the solvent really matter?” And the answer is a resounding YES! The solvent is not just some pretty background décor. It plays a vital role in this whole process. For sodium borohydride reductions, we usually use protic solvents like water or alcohols (methanol, ethanol). Why? Because they can provide that crucial proton in the final step.
These protic solvents also help to dissolve both the sodium borohydride and the 9-fluorenone, so they can actually meet and mingle. If they’re not dissolved, they’re just sitting there, like strangers at a party who never get introduced. The solvent facilitates the reaction, making sure everyone can get up close and personal.
Think about it. If we used an aprotic solvent, like diethyl ether or hexane, we wouldn’t have those handy protons to help neutralize our alkoxide intermediate. The reaction might not go to completion, or it might take way longer. So, the choice of solvent is, like, a big deal. A really big deal.
Side Reactions and Other Fun Stuff (Not Always Fun!)
Of course, in chemistry, it’s not always a perfectly clean, one-way street. Sometimes, things can go a little… sideways. With NaBH4 and 9-fluorenone, it’s usually pretty straightforward. But, in general, reductions can have side reactions. For example, if you’re using a more reactive reducing agent or have other functional groups present, you might get unintended reductions. It’s like trying to do one thing, and accidentally doing five other things. Whoops!
In our specific case, the ketone in 9-fluorenone is pretty reactive towards NaBH4, and there aren’t many other easily reducible groups. So, it’s usually pretty selective. But it’s always good to be aware that chemistry can be a bit wild sometimes! Nature loves a good surprise, even if it messes with your yield.
And then there’s the work-up. After the reaction is done, you have to, you know, isolate your product. That usually involves quenching any remaining reducing agent (often with a bit more acid or water) and then extracting your 9-fluorenol. It's like cleaning up after a party. You gotta get rid of the mess and find your prized possession. Sometimes it's easy, sometimes it's a whole adventure!
The Big Picture: Why Does This Matter?
So, we’ve taken 9-fluorenone and turned it into 9-fluorenol using sodium borohydride. We’ve talked about the hydride attack, the borate intermediate, and the all-important protonation. It’s a beautiful, fundamental organic chemistry reaction. It teaches us about nucleophilic attack, the role of functional groups, and the importance of reducing agents.
This isn't just some random lab experiment for the sake of it. Understanding these mechanisms helps us design new reactions, create new materials, and solve problems in medicine, industry, and beyond. It's all about manipulating molecules to do our bidding. And that, my friends, is pretty darn cool. So, next time you see a ketone, remember that with a little help, it can become an alcohol. A little transformation, a little improvement. Just like us, really. Now, who needs a refill?
