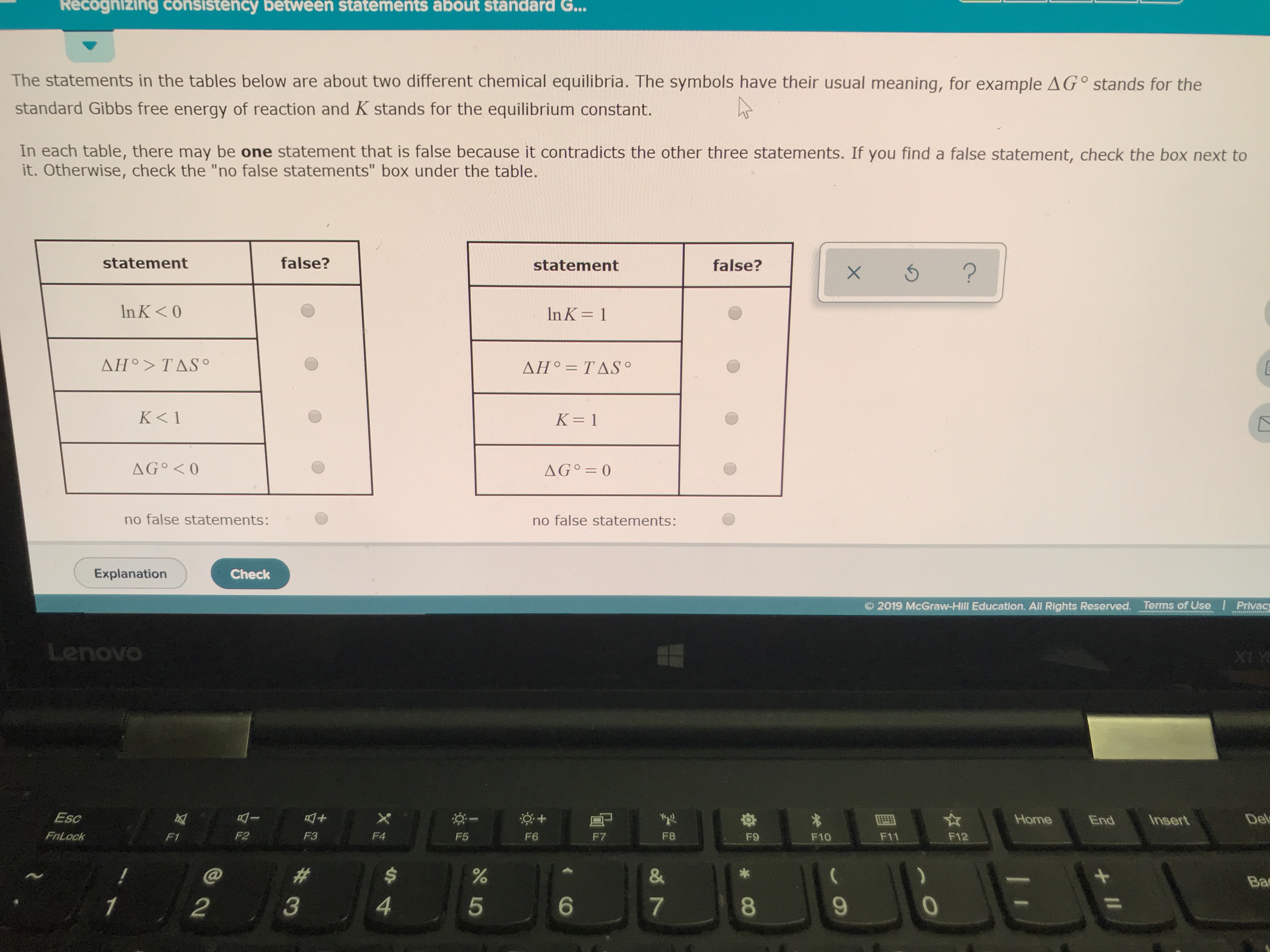
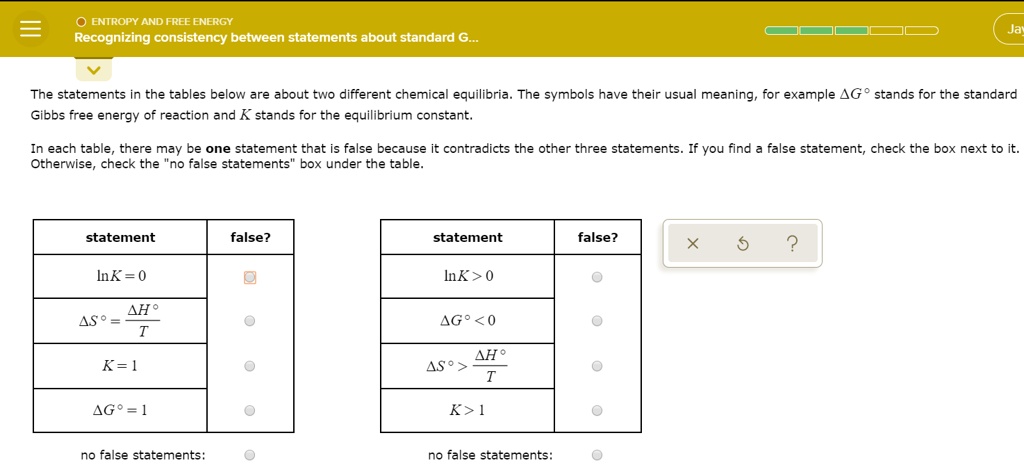
Recognizing Consistency Between Statements About Standard Gibbs Free Energy

Hey there, science nerds and curious cats! Ever feel like chemistry is just a bunch of grumpy professors muttering about reactions? Well, buckle up, because we’re about to dive into something super cool. We're talking about standard Gibbs free energy. Sounds fancy, right? But trust me, it’s like a secret handshake for understanding if a reaction is going to be a party or a total flop. And the best part? Spotting its consistency is way more fun than it sounds.
So, what’s the big deal with this “Gibbs free energy” thing? Think of it like this: it’s the usable energy in a system. Not the heat that just gets lost, but the energy that can actually do work. Like making a balloon inflate, or your muscles move. Pretty neat, huh?
And “standard”? That just means we’re talking about it under super specific, neat-freak conditions. Like, a temperature of 25 degrees Celsius (that’s 298 Kelvin for you math wizards!) and a pressure of 1 atmosphere. It’s like a perfectly controlled science experiment, ready for its close-up.
Now, the real fun kicks in when we start looking at consistency. Imagine you’re comparing notes with a friend about a recipe. You both agree that adding more sugar makes cookies sweeter. That’s consistency! In chemistry, it’s the same idea, but with energy. We're looking for whether different statements or calculations about standard Gibbs free energy agree with each other.
Why is this consistency so important? Because it tells us if our understanding is solid. If different scientists, using different methods, arrive at similar Gibbs free energy values for the same reaction, then we’re on the right track! It’s like finding out you and your buddy independently figured out the best way to fold a fitted sheet. High fives all around!
Let’s get a little quirky. Did you know that J. Willard Gibbs, the guy this energy is named after, was kind of a hermit? He was a brilliant mathematician and physicist, but he mostly stayed in his study. He probably would have loved online forums to discuss thermodynamics. Imagine him arguing about enthalpy versus entropy with strangers from his armchair!
So, how do we spot this consistency? It’s all about the numbers. When you see values for standard Gibbs free energy, they’ll usually have a little degree symbol (°) next to the delta (Δ), looking like ΔG°. This little guy means “standard conditions.” And the unit? It's usually kilojoules per mole (kJ/mol). Think of it as the "oomph" a reaction has, measured in tiny energy packets for every bunch of particles.
One of the coolest ways consistency shows up is through Hess's Law. This law is like a superhero move for thermochemistry. It says that the total change in enthalpy (another energy measurement, but we're focusing on Gibbs here!) for a reaction is the same, no matter how you get there. And guess what? A similar principle applies to Gibbs free energy! You can break down a big, scary reaction into smaller, manageable ones. If the Gibbs free energy values for those smaller steps add up to the Gibbs free energy for the overall reaction, bingo! Consistency detected.

Imagine you want to know the Gibbs free energy for making a peanut butter sandwich. You could try to measure it directly, which is a nightmare. Or, you could figure out the energy to gather the bread, the energy to get the peanut butter, and the energy to combine them. If those smaller energy costs add up to the total sandwich-making energy, then your calculations are consistent. See? It’s relatable!
Another place to look for consistency is in standard free energy of formation (ΔG°f). This is the Gibbs free energy change when one mole of a compound is formed from its constituent elements in their standard states. Think of it as the "building block cost" for molecules. If you can find these formation values for all the reactants and products in a reaction, you can calculate the overall ΔG° for that reaction. Then, you can compare that calculated value to a value found elsewhere. If they match? Boom! Consistency confirmed. It's like finding out your favorite pizza place uses the same secret ingredient in all its pizzas.
And here’s a funny thought: sometimes, the most minor differences in reported Gibbs free energy values can be a clue. Maybe one table used a slightly different temperature (like 24.9 °C instead of 25 °C), or a pressure that was a hair off. These tiny discrepancies aren’t usually errors, but they highlight how precise we need to be. It’s like a detective noticing a single scuff mark on a shoe. It might not be a smoking gun, but it’s a detail worth investigating.

What about those moments when things don't seem consistent? Don't panic! It's not necessarily a cosmic error. It could be that the "standard conditions" were interpreted slightly differently. Or maybe one source is using older data. It's a chance to learn more, not less. It’s like when your GPS recalculates your route – it’s not broken, it’s just finding a better way!
The beauty of standard Gibbs free energy is its predictive power. A negative ΔG° means the reaction is spontaneous. It'll happen on its own, given enough time. Think of a ball rolling downhill – it doesn’t need a push. A positive ΔG° means the reaction needs an energy input to happen. It’s like pushing that ball uphill. And ΔG° equal to zero? That’s equilibrium. The reaction is happy to go either way, like a perfectly balanced seesaw.
So, when you see statements about standard Gibbs free energy, ask yourself: Do these numbers tell the same story? Do they predict spontaneity in the same way? Are they calculated using consistent methods? This isn't about memorizing a million numbers. It's about understanding the language. It's about recognizing the patterns.
Think of it like recognizing a familiar tune. You might not know all the notes, but you can tell when it's the same song. Similarly, you can recognize the consistency in Gibbs free energy statements by looking at the sign (positive, negative, or zero) and the general magnitude of the values. Do they both suggest the reaction is a party animal (negative ΔG°) or a couch potato (positive ΔG°)?
And here’s a little secret: this stuff is actually used everywhere! From designing new drugs to understanding how our bodies work, to developing cleaner energy sources. So, when you’re spotting that consistency in Gibbs free energy, you’re not just doing a science exercise. You’re peeking behind the curtain of how the world operates.
So next time you stumble upon a mention of standard Gibbs free energy, don't shy away. Lean in! Look for the ΔG°. See if the numbers tell a coherent story. Are they all singing from the same hymn sheet? It’s a fun little detective game, and the reward is a deeper understanding of the universe’s energy budget. Happy calculating, and may your reactions always be spontaneous (when you want them to be, of course)!
