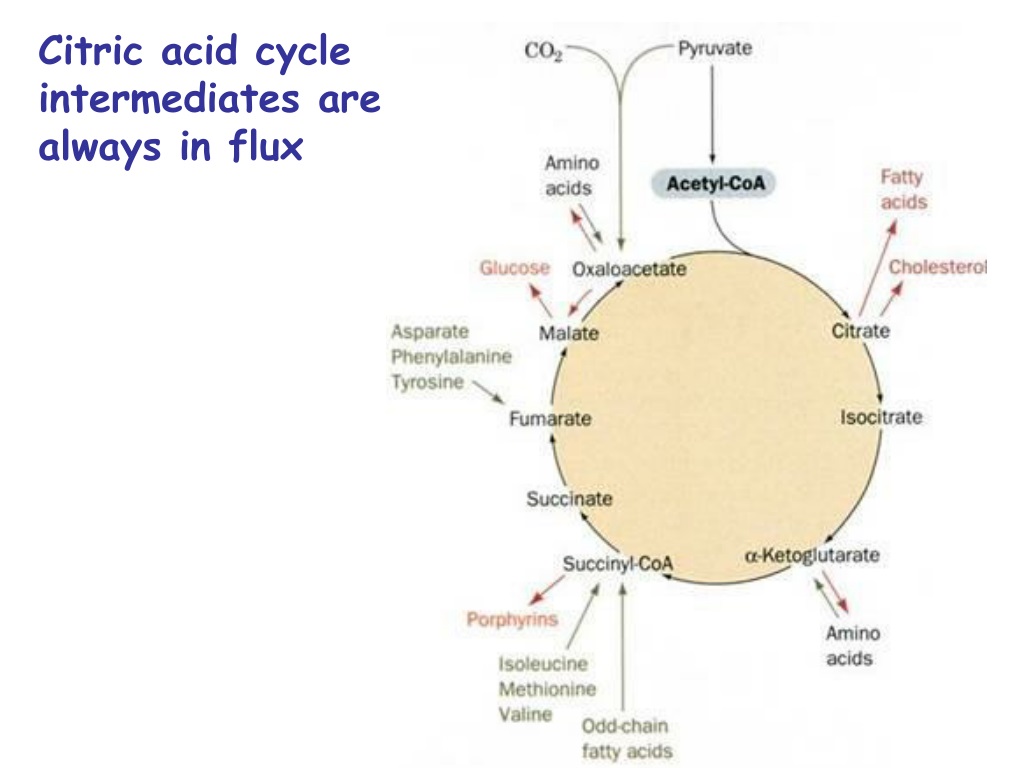
Reactions Drain The Citric Acid Cycle Of Intermediates.
Ever wondered what happens after you take that first bite of a delicious, zesty orange? Or how your body extracts the energy from that sweet strawberry? It’s a microscopic ballet of fascinating chemical reactions, and one of the main performers is a brilliant cycle called the Citric Acid Cycle, also known as the Krebs Cycle or the TCA Cycle. Think of it as the central processing unit for energy production in your cells. It’s not just crucial for survival; understanding this cycle gives us a peek into the intricate workings of life itself, making it a surprisingly fun and useful topic to explore!
The Powerhouse's Playlist: What the Citric Acid Cycle Does for You
So, what’s the big deal about the Citric Acid Cycle? Its primary purpose is to generate the molecules that fuel almost everything your body does. From the twitch of a muscle to the firing of a neuron, it all relies on the energy produced here. It’s like the body’s personal power plant, churning out the “ATP” (adenosine triphosphate), the universal energy currency of cells. But it’s not just about ATP directly. The cycle also produces electron carriers, specifically NADH and FADH2. These are like energized couriers, carrying high-energy electrons to another important stage of energy production called the electron transport chain. Without these couriers, the real ATP-making party can't even get started!
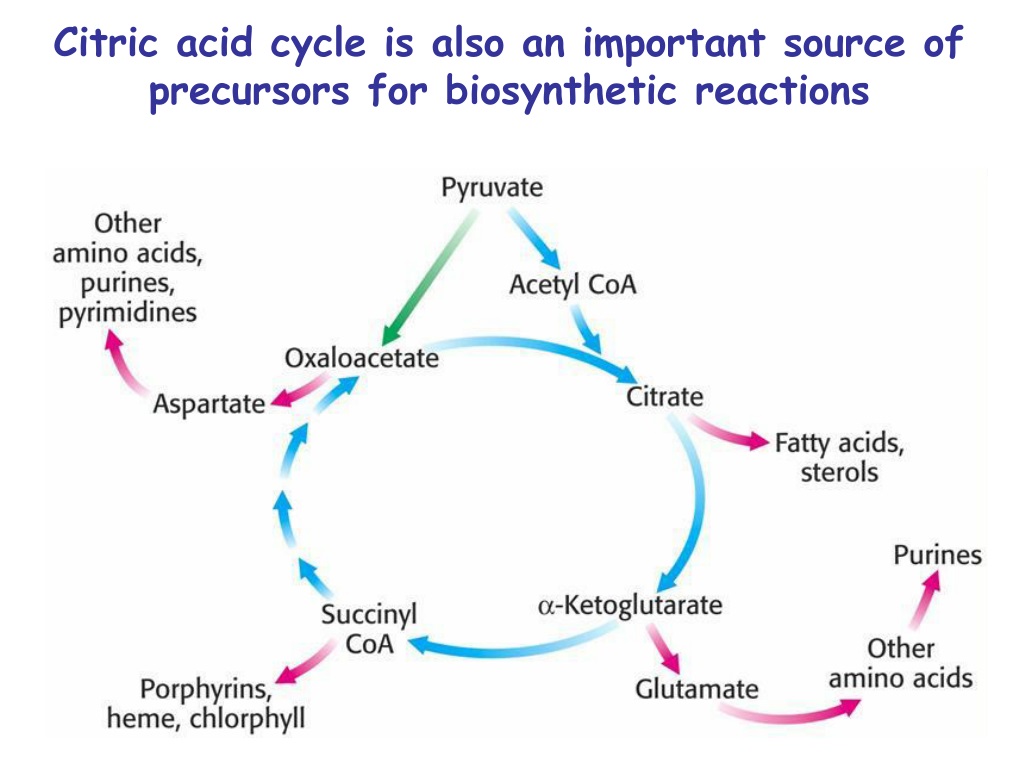
The benefits are profound. A well-functioning Citric Acid Cycle means efficient energy production, which translates to sustained energy levels throughout your day. It’s essential for metabolic health, helping your body break down carbohydrates, fats, and proteins to extract their energy. Furthermore, the cycle’s intermediates (the molecules that are formed and then transformed as the cycle progresses) are not just confined to making energy. They are also vital building blocks for other essential molecules your body needs, like amino acids (the components of proteins) and heme (a key part of hemoglobin, which carries oxygen in your blood).
When the Cycle Gets a Little Too Popular: Reactions Drain the Intermediates!
Now, here’s where things get really interesting and, dare we say, a little dramatic! While the Citric Acid Cycle is a wonderfully self-sustaining loop when it’s just doing its energy-making job, life isn’t always that simple. Sometimes, the molecules that are supposed to be circulating within the cycle get borrowed for other important tasks. These “borrowed” molecules are the cycle intermediates. Think of them like the popular kids at a school dance; everyone wants to talk to them and involve them in other activities.
Imagine the Citric Acid Cycle as a merry-go-round. The intermediates are the horses that go around and around. But sometimes, a horse is taken off the merry-go-round to be used in a parade! That’s what happens when reactions drain the intermediates.
PPT - Citric acid cycle Krebs cycle, tricarboxylic acid cycle TCA
What kind of reactions would be so important that they’d “borrow” from our beloved Citric Acid Cycle? Well, a major one is the synthesis of glucose, the primary sugar your body uses for energy, especially in situations where you haven’t eaten for a while. The process of making new glucose, called gluconeogenesis, taps into some of the cycle’s intermediates. For instance, a molecule called oxaloacetate, a key starting and ending point for the cycle, can be pulled out to help build glucose. This is super important because your brain, in particular, relies heavily on glucose.
Another significant drain comes from the production of certain amino acids. Remember how we said the intermediates are building blocks? Well, some amino acids are directly synthesized from these cycle components. For example, the intermediate α-ketoglutarate is essential for making glutamate and other related amino acids. Similarly, succinyl-CoA can be diverted to help form porphyrins, which are crucial for making heme.
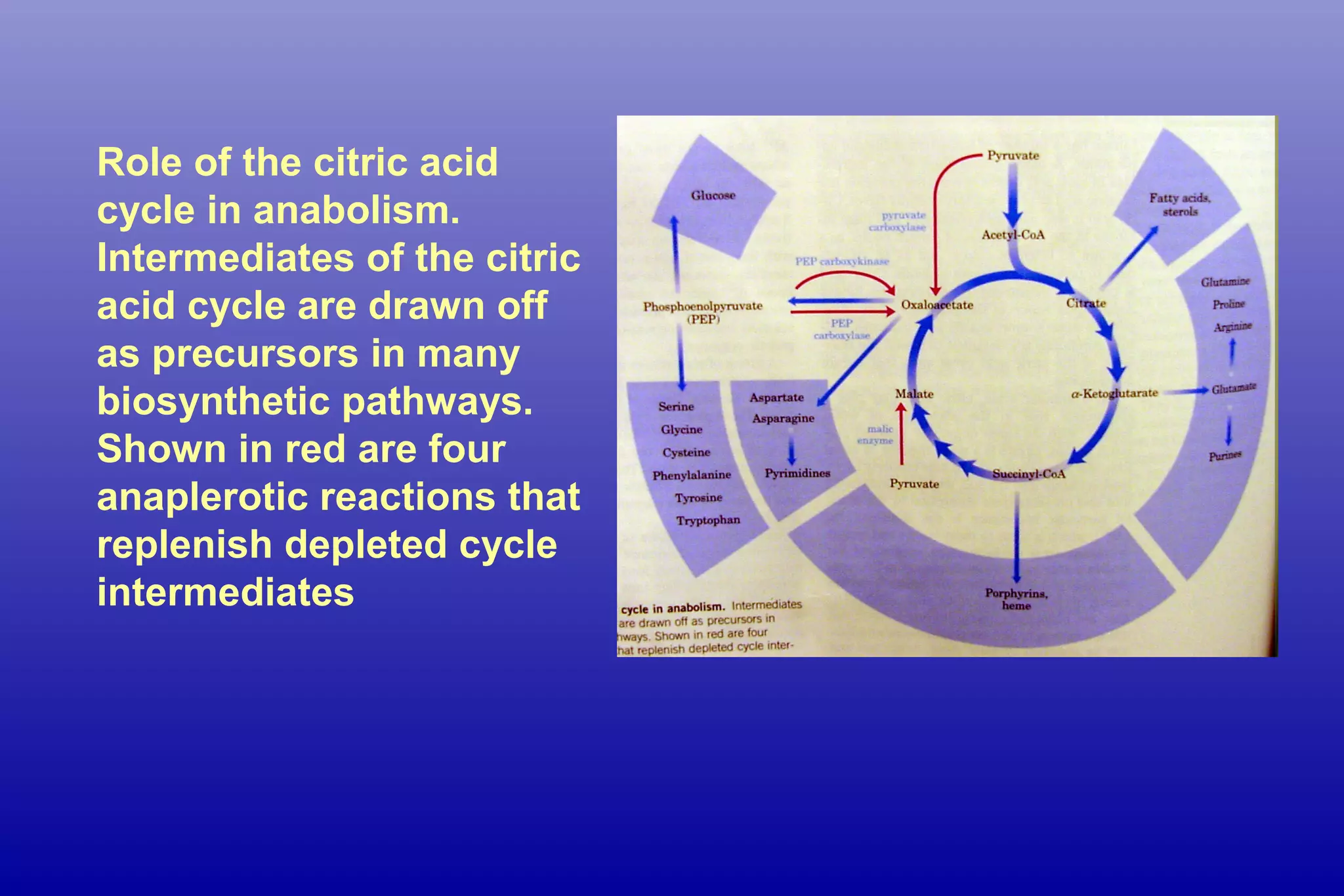
These "draining" reactions aren't necessarily bad; they highlight the incredible interconnectedness of our cellular machinery. The body is incredibly smart about balancing its needs. When glucose is low, the body will prioritize making it. When the body needs to build more proteins, it will utilize the intermediates. However, this diversion means that the Citric Acid Cycle can’t keep spinning at its usual speed if its intermediates are constantly being taken away. If too many intermediates are removed for these other vital synthesis pathways, the cycle can slow down.
This slowdown can have consequences. A less efficient Citric Acid Cycle means less ATP is produced, potentially leading to fatigue or reduced metabolic activity. It’s a delicate balancing act that your body performs constantly. The body has ways to replenish these intermediates, often by bringing in new fuel molecules to restart the cycle. But understanding these drains gives us a clearer picture of how dynamic and responsive our cellular energy systems truly are. It’s a testament to the elegant, yet sometimes demanding, choreography of life at the molecular level, making the Citric Acid Cycle and its intricate dance with other metabolic pathways a truly fascinating area of biology!