Rate Law Determination Of The Crystal Violet Reaction Answers

Ah, the Crystal Violet reaction. You know, the one that makes pretty purple solutions slowly fade away. It sounds like a spa treatment for your lab bench, doesn't it? But behind that pretty fade lies a whole lot of chemical detective work. And let me tell you, some of that detective work can feel like trying to find a needle in a haystack, blindfolded.
We're talking about figuring out the "rate law." It's basically the secret handshake of a chemical reaction. It tells you how fast things are going to happen, and what ingredients are the real stars of the show. Think of it like a recipe for speed.
And the Crystal Violet reaction? It's famous for being the ultimate lab test subject. It's like the guinea pig of kinetics. Every chemistry student has to wrestle with it. It's practically a rite of passage.
The Plot Thickens (or Fades, in this case)
So, imagine you're staring at this purple liquid. It's gorgeous, right? But then it starts to lighten. You're supposed to measure how fast it's fading. This is where the fun (and sometimes, the mild panic) begins.
You've got your beakers, your timers, your spectrophotometer – which sounds fancy, but it's basically a light-measuring gadget. You're looking at the concentration of this stuff, crystal violet. You're watching it disappear. It's a race against time, but a very, very slow race.
And you're doing this, like, a million times. Or at least it feels like it. You're changing things up. You're adding more of one reactant, less of another. You're trying to see what makes the purple fade faster or slower. It's like being a mad scientist, but with less dramatic lightning.

Unraveling the Mystery: The "Rate Law" Revealed
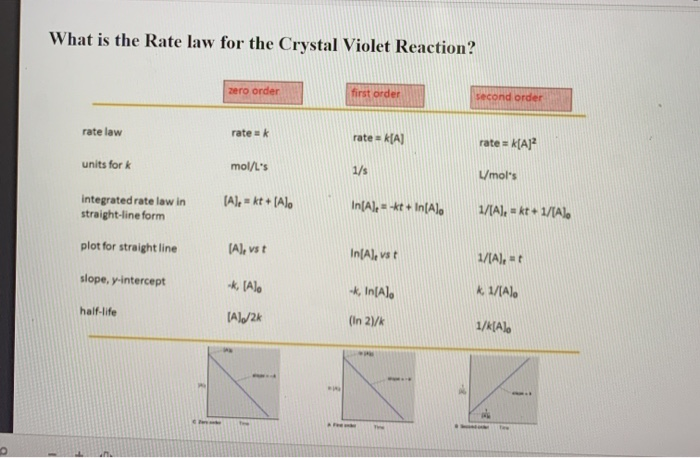
The whole point is to get to the rate law. It's usually in the form of: Rate = k[Crystal Violet]^m[Other Reactant]^n. See those little letters, 'm' and 'n'? Those are the exponents we're trying to discover. They tell us how much each ingredient matters.
For Crystal Violet, things get a little... predictable. After all the tinkering, the graphs, and the frantic calculations, you usually find that the exponents are pretty simple. Like, really simple. So simple, it makes you wonder if all that work was strictly necessary.
My unpopular opinion? Sometimes the Crystal Violet reaction's rate law feels like being told 2+2=4 after building a supercomputer. It's correct, absolutely. But you can't help but feel like there was a shortcut. Or maybe I'm just saying that because I spent too long staring at faded purple.
"The answer is often simpler than the journey there."
You'll often see that the rate law for the Crystal Violet reaction is first-order with respect to crystal violet. That means the rate of fading is directly proportional to how much crystal violet you have. Double the crystal violet, double the fading speed. Simple, right?

And the other reactant? Usually, it's something like sodium hydroxide. Guess what? The exponent for that is often zero. Zero! That means it barely matters how much of it you throw in there. It's like adding a tiny sprinkle of fairy dust; it doesn't change the outcome much.
So, the rate law often boils down to something like: Rate = k[Crystal Violet]^1. That's it. Just the crystal violet itself matters. All that painstaking work, all those fiddly measurements, and the answer is staring you in the face. Well, not literally staring, but it's definitely there in your data.
You might be sitting there, nodding along, remembering those lab sessions. You might even be feeling a little bit of that old frustration creep back in. "Why did I have to do all that graphing?" you might be thinking. I'm right there with you.
It's like learning to drive. You spend ages practicing parallel parking, all the while knowing that most of the time you'll just be going in a straight line. The skill is important, but the everyday application is… less dramatic.
The "Answers" You're Looking For
So, what are the actual answers? What are the exponents? For the classic Crystal Violet reaction experiment you'll do in introductory chemistry, the rate law is typically:
Rate = k[Crystal Violet]^1 [Sodium Hydroxide]^0
Yes, that little '0' next to sodium hydroxide is the punchline. It’s the chemical equivalent of a shrug. It means that specific reactant, in this common experimental setup, is essentially a spectator. It’s there, but it’s not driving the speed.
And that '1' next to crystal violet? That means it's a first-order reaction with respect to crystal violet. The speed of the fading directly matches the amount of purple dye present. Pretty straightforward once you peel back the layers.
The 'k' is the rate constant. It's a number that tells you how fast the reaction is at a specific temperature. It's like the speed limit for that particular chemical interaction. You'll calculate that too, of course. More calculations!

But the exponents, 'm' and 'n' (which are 1 and 0 in this case), those are the real meat of the rate law determination. They're what you're trying to nail down with all those experiments.
It’s funny, isn't it? You spend hours meticulously collecting data, plotting graphs, and doing calculations, all to confirm that the reaction is, in fact, first-order with respect to the main ingredient and zero-order with respect to another. It’s like conducting a grand scientific investigation to discover that water is, indeed, wet.
But that's the beauty of science, I suppose. Even the "obvious" needs to be proven. And even if the answer feels a little anticlimactic after all that effort, there's a certain satisfaction in cracking the code. You’ve figured out the reaction's secret speed formula.
So next time you see that purple fade, you can smuggle a knowing smile. You understand the underlying kinetics. You know it's not just magic; it's chemistry. And sometimes, that chemistry is surprisingly straightforward. Even if getting there felt like a bit of an epic quest.
