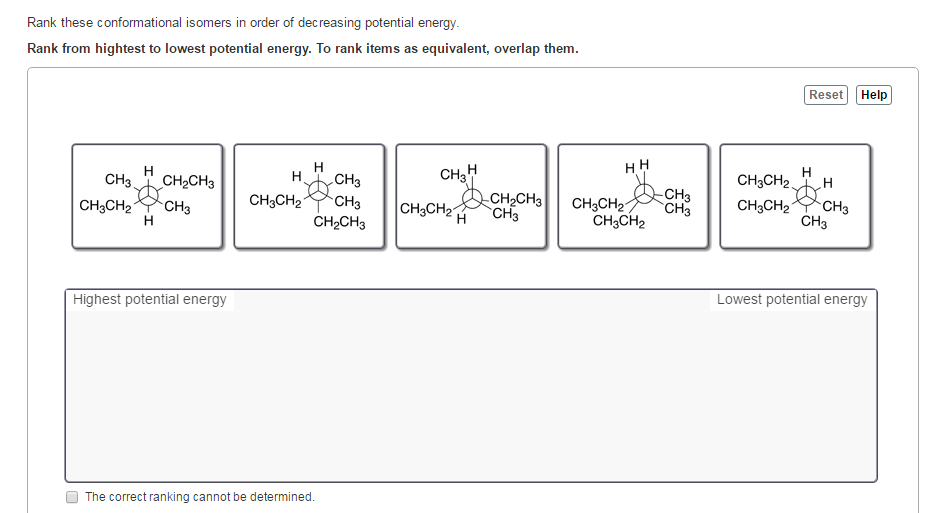
Rank These Conformational Isomers In Order Of Decreasing Potential Energy
Ever wondered why some molecules just 'prefer' to be in a certain shape? It's a bit like choosing your favorite comfy couch position – some are just more stable and less… wobbly. Today, we're diving into the fun world of conformational isomers, which are basically different 3D arrangements a molecule can adopt by simply rotating around its single bonds. Think of it like twisting a flexible toy – it can bend and twist into various shapes, but it's still the same toy! Understanding these shapes isn't just for super-smart scientists; it's a surprisingly practical and even enjoyable puzzle for anyone curious about how things work at a tiny level.
So, why should you care about ranking molecules by their potential energy? For beginners, it’s a fantastic way to build intuition about molecular stability. You'll start to see that some arrangements require more effort (energy) to maintain, like holding your arm out straight for a long time versus letting it rest by your side. For families, this can be a fun, hands-on activity. Imagine using pipe cleaners or Lego bricks to build simple molecules and then twisting them around to see which shapes are easiest to hold. It's a great way to introduce basic chemistry concepts in a tangible way. Hobbyists, especially those interested in molecular modeling, drug design, or even understanding how certain flavors and scents work, will find this knowledge incredibly valuable. It helps explain why one isomer might be more reactive, more stable, or have different properties than another.
Let's take a peek at a common example: ethane. This is a simple molecule with two carbon atoms connected by a single bond. The carbons can rotate freely, leading to different arrangements. Imagine looking down the carbon-carbon bond. One arrangement is eclipsed, where the hydrogen atoms on one carbon are directly in front of the hydrogen atoms on the other. This is like trying to stand with your feet perfectly aligned with someone else's – it feels a bit crowded and unstable, right? Then there's the staggered arrangement, where the hydrogen atoms are as far apart as possible. This is much more comfortable and stable, like standing with your feet shoulder-width apart. The staggered conformation has lower potential energy than the eclipsed one.
Here's a simple way to get started: Grab some marshmallows and toothpicks! Make simple molecules like ethane or butane. You can then physically twist the "bonds" (toothpicks) to see how the "atoms" (marshmallows) arrange themselves. Notice how some arrangements cause the marshmallows to bump into each other – those are the higher energy, less stable ones. Always remember that the goal is to minimize these 'crowded' interactions. You can even draw these out on paper, imagining the 3D space.
Ranking conformational isomers by potential energy is essentially a molecular popularity contest, where the most comfortable and least crowded shapes win by having the lowest energy. It’s a puzzle that unlocks a deeper understanding of the invisible world around us, making chemistry more accessible and, dare we say, fun!
