Rank The Structures In Order Of Decreasing Electrophile Strength.
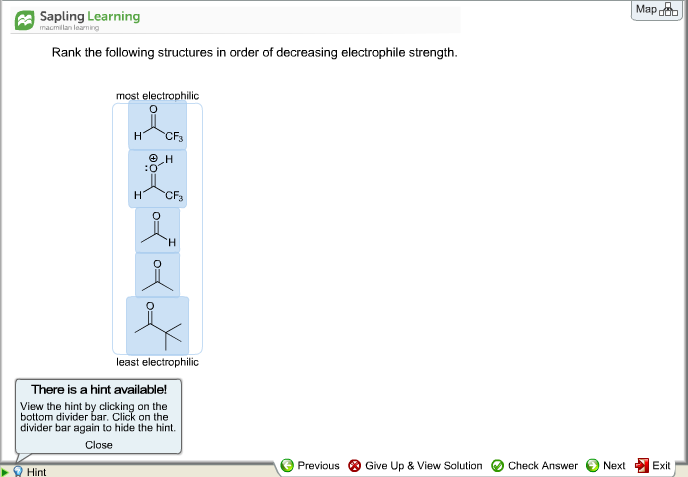
Imagine a giant game of chemical tag, where some molecules are the super-fast runners, and others are the eager chasers. Today, we're diving into the exciting world of these chemical tag players, specifically the ones who are always looking for a dance partner – the electrophiles! These guys are like the life of the party, always ready to grab onto something with extra electrons. But just like in any party, some are more popular and more outgoing than others.
Think of it this way: some electrophiles are so popular, they're practically shoving their way to the front of the line to make a connection. Others are a bit more shy, hanging back and waiting for the perfect moment. We're going to rank these enthusiastic molecules, not by how much they bench-press or how many followers they have on social media, but by how good they are at attracting electrons. It’s all about their electrophile strength – how desperately they crave those electron-rich pals.
Let's start with the absolute rockstars, the ones who practically vibrate with eagerness. At the very top of our list, ready to swoop in and make a connection faster than you can say "chemical reaction," are molecules that are practically begging for electrons. These are the heavy hitters, the ones with a super strong positive charge or a really electron-hungry atom just itching to bond. They're the ones who will make other molecules feel like the most popular kid in school, instantly drawing them in.
These are the molecules that mean business. They're not playing around; they're here to make friends, and they're not afraid to show it!
As we move down the list, the eagerness might calm down just a tiny bit. It's like going from a mosh pit to a slightly more organized dance floor. These next electrophiles are still very much looking for a partner, but perhaps they’re a tad more discerning. They have that electron-seeking ability, but it's not quite as overwhelming as the top tier. They’re still the life of the party, just maybe with a slightly more curated guest list.
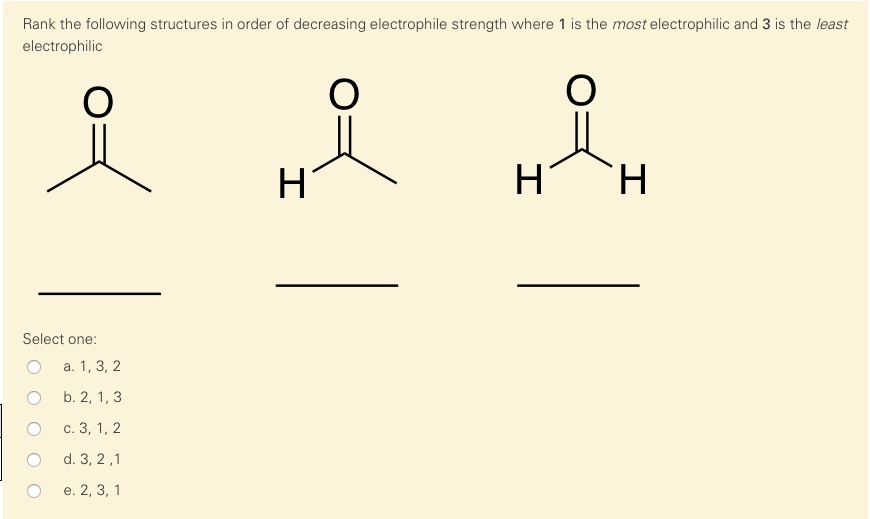
Think of a molecule that has a carbon atom that's just a little bit positive. It's not a full-on rave, but it's definitely a lively gathering. This carbon is still super keen to find an electron-rich neighbor. It's like that friend who’s always up for an adventure, but they might check the weather forecast first. They’re enthusiastic, but with a touch of sensible planning. They’re still incredibly important players in the chemical world, driving many of the transformations we see.
Now, let’s talk about the ones that are still definitely looking for a connection, but maybe they’re a bit more laid-back about it. These are the molecules where the "electrophile" vibe is present, but it's more of a subtle hint than a neon sign. They might have an atom that could attract electrons, but it's not as aggressively seeking them out. It’s like that person who’s happy to chat if you come over to them, but they’re not going to chase you down across the room.

These molecules are still valuable participants, but their desire for electron buddies is more of a gentle hum than a loud declaration.
Sometimes, the way a molecule is structured, or the atoms it's partnered with, can make it seem like it should be a super strong electrophile, but in reality, it's a bit of a poser. These are the ones we have to watch out for! They might look the part, with all the right atoms in place, but their electron-grabbing power is actually quite weak. They're the ones who might show up to the party but spend most of their time in a corner, observing.
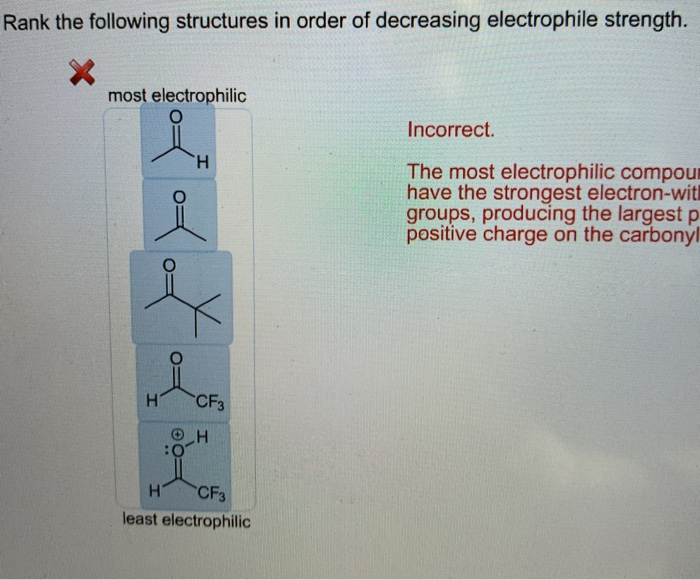
This is where things get really interesting. You might see a structure and think, "Wow, that looks super reactive!" but then, upon closer inspection, you realize its true electrophilic nature is a bit… subdued. It’s like judging a book by its cover – sometimes the most unassuming molecules are the ones with the most hidden depths, and vice versa. These weaker electrophiles still play a role, of course, but they require a bit more encouragement to get involved in the chemical dance.
Finally, we have the molecules that are barely in the running. They have the potential to act as electrophiles, but it's a very, very mild potential. They’re the ones who might only react under very specific, energetic circumstances, or with an incredibly powerful electron donor. Think of them as the quiet observers at the back of the hall, who might only get pulled onto the dance floor if there's a sudden outbreak of everyone doing the Macarena. Their electrophile strength is so low, it’s almost negligible in many everyday chemical scenarios. But even these can have their moments of surprising reactivity!
So, as we rank these molecules from the most eager dancers to the most reserved wallflowers, remember that even the shyest ones have a story to tell. It’s a constant interplay of attraction and connection, a beautiful chemical ballet happening all around us, and understanding who's leading the dance and who's waiting their turn gives us a whole new appreciation for the hidden world of chemistry!
