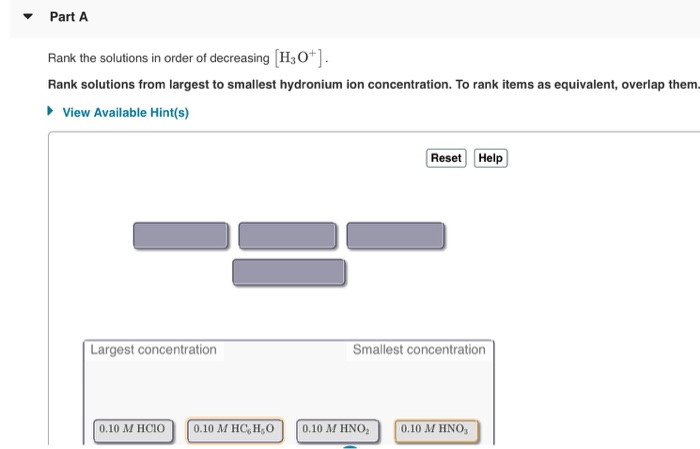
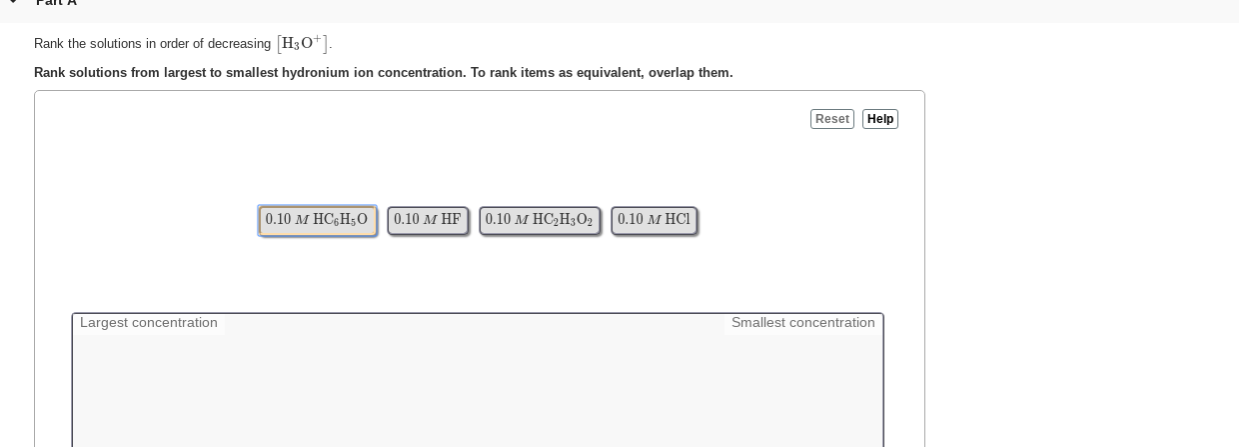
Rank The Solutions In Order Of Decreasing H3o+

Hey there, science curious peeps! Ever found yourself staring at a bottle of vinegar or a glass of lemonade and wondered what’s really going on under the hood? Like, what makes one sourer than the other? Well, today we’re diving into something super neat called hydronium ions – or H3O+ for the lab coat enthusiasts. Don't let the fancy name scare you; it’s all about how acidic something is, and understanding it is kinda like learning a secret code to the world around you.
Think about it. We’ve got all sorts of liquids in our lives, from the stuff we chug to stay hydrated to the cleaning agents we use to make our homes sparkle. And these liquids can behave so differently, right? Some are gentle, some are a bit… intense. This intensity, this tanginess, this sourness – it all boils down to these little H3O+ guys. They're basically the tiny, energetic troublemakers that make acids, well, acidic.
So, what exactly are these H3O+ characters? Imagine water (H2O) is like a chill, laid-back partygoer. And then an acid comes along, like a hydrogen ion (H+), and it’s a bit more… demanding. This demanding H+ ion crashes the water party and basically latches on, forming H3O+. It’s like the H+ ion found a new best friend in H2O and they decided to stick together. This combination, H3O+, is what we call a hydronium ion. And the more of these guys floating around in a solution, the more acidic that solution is.
Why is this so cool? Because it gives us a way to rank things! It’s like a popularity contest for acidity. We can actually put different substances in order based on how many H3O+ ions they’ve got. Pretty neat, huh? It’s like having a secret superpower to understand why your stomach might get a bit upset after too much spicy food (hello, stomach acid!) or why your grandmother’s secret lemon meringue pie recipe is so perfectly tart.
Let’s get to the main event: ranking solutions in order of decreasing H3O+. This means we’re going to start with the solution that has the most H3O+ ions (the most acidic) and work our way down to the one with the least H3O+ ions (the least acidic, or most neutral/basic). Think of it like going from a rock concert with thousands of screaming fans to a quiet library with just a few folks whispering.
The Super Acidic End of the Spectrum
At the very top, with the absolute highest concentration of H3O+ ions, you’ll find things that are incredibly strong. We’re talking about acids that can be downright dangerous if you’re not careful. Imagine these as the VIP section of the acidity club, packed to the brim!
One of the heavy hitters here is battery acid. Yep, the stuff that powers your car’s battery. This is some serious H3O+ power. It's so concentrated that it can eat through metal and is definitely not something to play around with. If H3O+ was a superhero, battery acid would be its strongest, most concentrated blast of power.
Right behind it, you’d likely find things like concentrated sulfuric acid and hydrochloric acid. These are the workhorses of many chemical labs. They’re used in all sorts of industrial processes, from making fertilizers to refining metals. They’ve got a massive amount of H3O+ zipping around, making them super reactive and excellent at breaking down other substances. Think of them as the professional bodybuilders of the H3O+ world – all muscle and energy.
The Moderately Acidic Zone
Moving down the ladder, we get to acids that are still strong but a bit more manageable. These are the ones you might encounter more in everyday life, though they still require a bit of respect.
A prime example here is stomach acid. Your stomach is a marvel of biological engineering, and its acidic environment (mostly hydrochloric acid) is crucial for digesting food and killing off harmful bacteria. It’s got a good concentration of H3O+, enough to do its job effectively without destroying your insides. It’s like the diligent security guard of your digestive system.
Then we have common household acids like vinegar. That sharp, tangy smell? That’s the H3O+ at work! Vinegar is a dilute solution of acetic acid, and while it’s great for salad dressings and cleaning, it’s nowhere near as potent as battery acid. It's more like the friendly neighborhood gym coach, strong but approachable.
Citrus fruits also bring the H3O+! Lemon juice and orange juice are packed with citric acid. They provide that characteristic sour punch that makes them so refreshing. They’re definitely acidic, but in a way that’s designed for our enjoyment (and for making delicious desserts!). They’re the zesty, energetic dancers of the H3O+ group.
The Mildly Acidic and Almost Neutral Territory
As we continue our descent, the H3O+ concentration gets lower and lower. These solutions are much gentler and often taste or feel less intense. They're the ones you might use for everyday tasks without much worry.
Consider rainwater. Pure water is neutral, but rainwater absorbs carbon dioxide from the atmosphere, which forms carbonic acid. This makes rainwater slightly acidic, but usually not enough to cause concern unless pollution levels are very high. It’s like a light breeze compared to a hurricane of acidity.
And then there’s milk. Milk is also slightly acidic, thanks to lactic acid. It’s a delicious and nutritious drink, and its mild acidity contributes to its overall properties. It's a comforting, familiar presence in the H3O+ lineup.
The Neutral Zone
This is the sweet spot, the land of balance. Here, the concentration of H3O+ is just right – not too much, not too little. These solutions are neither acidic nor basic.

The ultimate ruler of this kingdom? Pure water. When water is just H2O, with no added acids or bases, it's perfectly neutral. It’s the ultimate chill-out zone, the serene meditation retreat of the liquid world. It has a balanced amount of H3O+ and its counterpart, hydroxide ions (OH-).
You might also find things like certain saline solutions in this zone, depending on their exact composition. They’re carefully balanced and don’t lean towards being acidic or basic.
The Slightly Basic Side (where H3O+ is really taking a backseat)
Now, for the solutions where H3O+ ions are really scarce. These are moving away from acidity and towards being basic (or alkaline). In these solutions, there are actually more hydroxide ions (OH-) than hydronium ions (H3O+).
Think about baking soda dissolved in water. Baking soda is a base, meaning it tends to neutralize acids. So, when you mix it with water, it doesn't add a lot of H3O+; in fact, it can react with any stray H3O+ that might be present, making the solution less acidic. It’s like the helpful mediator, trying to bring things back to center.

As we move further into the basic territory, the H3O+ concentration drops even more significantly. Things like ammonia (household ammonia cleaners) have a low concentration of H3O+. They feel slippery and have a distinct smell because they are basic.
The Very Basic End of the Scale
Finally, at the far end of the spectrum, with the absolute lowest concentration of H3O+ ions, you’ll find the strongest bases. These are the opposite of the super acids!
Drain cleaner, often containing lye (sodium hydroxide), is a prime example. These are powerful substances that can be very corrosive. They have so few H3O+ ions that they can actually strip away organic material. They are the ultimate chillers, so low in H3O+ that they’re almost the anti-acidity!
So, there you have it! A journey from the incredibly acidic to the decidedly basic, all ranked by the mighty H3O+ ion. It’s a fun way to think about the world, right? The next time you’re sipping a fizzy drink or cleaning your countertops, you can have a little secret science chat with yourself about the H3O+ concentration.
Understanding these little ions helps us appreciate why things taste, smell, and react the way they do. It’s a small peek into the incredible chemical dance happening all around us, all the time. Pretty cool, wouldn’t you say?
