Rank The Relative Nucleophilicity Of The Indicated Species In Ethanol
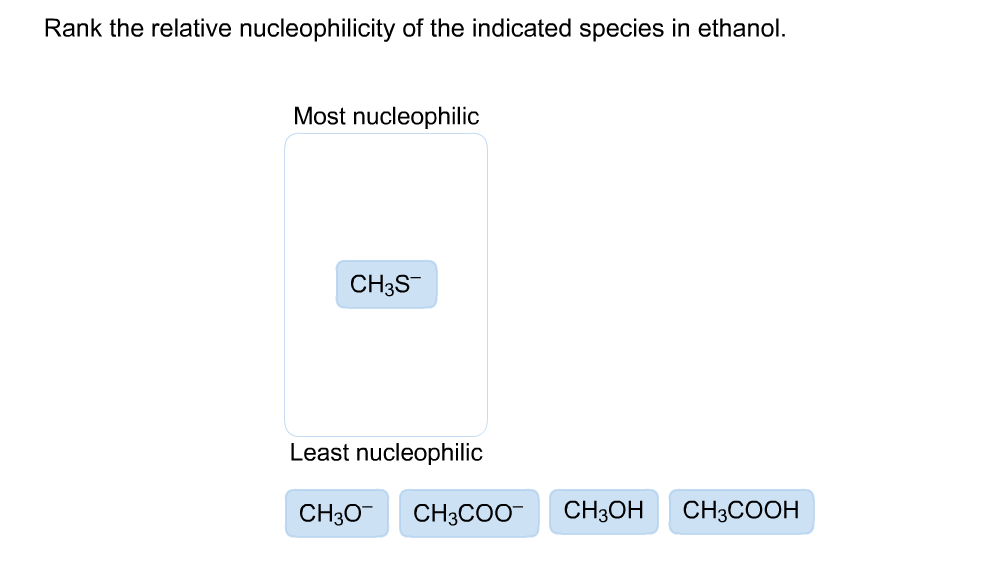
Hey there, chemistry curious folks! Ever wondered about the secret lives of tiny particles, those unsung heroes of all the stuff around us? Today, we're diving into a super fun, slightly dramatic world: the wild west of nucleophilicity in a place we'll call Ethanol-ville! Think of it like a bustling town where everyone's looking for a good time and a chance to share. And who are these party-goers? They're our amazing nucleophiles!
Now, "nucleophile" might sound like a fancy word for a spaceship captain, but it's really just a molecule that loves to give away a little something extra – usually an electron. It's like that generous friend who always has spare change for the vending machine or an extra cookie to share. These electron-givers are crucial for building all sorts of cool things, from the medicines that keep us healthy to the plastics that make our gadgets. And where do they do their best work? Well, today, we're setting the scene in Ethanol-ville!
Ethanol is basically rubbing alcohol, but imagine it as a lively, slightly crowded bar. It's got its own vibe, its own rules. And our nucleophiles are arriving, ready to mingle and make some chemical magic happen. We've got a few special guests tonight, and we're going to figure out who's the biggest, boldest, most eager giver of electrons – basically, who's the most nucleophilic! Think of it like ranking the most popular person at a party. Who's going to be the life of the conversation? Who's going to be the most sought-after?
Let's meet our contenders! We have hydroxide (OH-), which is like the seasoned veteran, a bit bossy but knows how to get things done. Then there's ethoxide (C2H5O-), who's basically hydroxide's cool cousin, a bit more relaxed and perhaps a tad more influential in Ethanol-ville. We've also got water (H2O), who's a bit shy, not quite as eager to share as the others. And finally, we have ethanol (C2H5OH) itself, who's sort of just hanging out, observing the scene, not quite as outgoing as the charged-up guests.
So, who's going to be the star of the show in Ethanol-ville? Who's going to be the most enthusiastically sharing their electron party favors? It all comes down to how much they want to give away those electrons. Some folks are just born givers, while others are a little more hesitant.
First up, let's talk about hydroxide (OH-). This little powerhouse is carrying a negative charge, which is like having a giant flashing neon sign that says "FREE ELECTRON OFFER INSIDE!" It's basically shouting, "I've got extra goodness to share, come and get it!" Because it has this extra electron hanging around, it's super keen to find a partner. It's like the person at the party who’s practically vibrating with excitement, eager to connect and form new bonds. Hydroxide is a major player. It's got that negative charge that makes it practically begging to react!
Now, let's bring in ethoxide (C2H5O-). This one is also sporting a negative charge, just like hydroxide. So, are they neck and neck? Well, sort of. Think of ethoxide as being in its natural habitat. Ethanol-ville is its stomping ground! It’s like the local celebrity who knows all the best spots and can easily make introductions. Because it's an organic molecule, the ethanol solvent is really comfortable with it, almost like a warm hug. This makes ethoxide feel even more comfortable and ready to share its electron bounty. It's got a bit of an edge because it's in its happy place, making it incredibly eager to jump into action. In fact, in Ethanol-ville, ethoxide often feels even more empowered and ready to share than its cousin, hydroxide!
Then we have water (H2O). Water is a pretty good molecule, it's involved in tons of reactions. But compared to our charged-up friends, it's a bit more reserved. It doesn't have that extra, easy-to-give-away electron like hydroxide or ethoxide. It's like someone at the party who’s enjoying themselves but not necessarily initiating every conversation. It can share, but it's not as desperate to do so. It's still a good nucleophile, don't get me wrong, but it's not the life of the electron-sharing party like the others.
And finally, we have ethanol (C2H5OH) itself. This is the solvent, the background music of our Ethanol-ville party. It's got an oxygen atom that has some electrons it could share, but it's not carrying that extra negative charge like hydroxide and ethoxide. It's like the person who's there, being friendly, but not actively looking to swap secrets or form deep connections right away. It's capable of being a nucleophile, but it's much, much less enthusiastic than its charged-up buddies. It's the quiet observer, the polite attendee.
So, if we were to rank our amazing nucleophiles on how eager they are to share their electron treasures in the lively atmosphere of Ethanol-ville, here’s the lowdown:

The absolute reigning champ, the king or queen of electron-giving, is ethoxide (C2H5O-)! It's in its element, feeling the good vibes of Ethanol-ville, and is just bursting with eagerness to react.
Close behind, with a huge amount of enthusiasm, is its powerful cousin, hydroxide (OH-). It’s got that negative charge that just screams "Let's party!" and it's a fantastic nucleophile.
Solved Rank the relative nucleophilicity of the indicated | Chegg.comNext in line, a reliable but slightly less boisterous participant, is water (H2O). It can definitely join in the fun and share, but it's not quite as driven.
And bringing up the rear, the chill observer of the scene, is ethanol (C2H5OH). It’s got the potential, but it’s not the most eager bee in the chemical bonnet.
Isn't that neat? It's all about who's feeling the most generous with their electron goodies. These little molecular interactions are the building blocks of everything we see and touch. So next time you’re enjoying a sip of something, or using a product, remember the unsung heroes, the nucleophiles, throwing their electron parties in places like Ethanol-ville!

