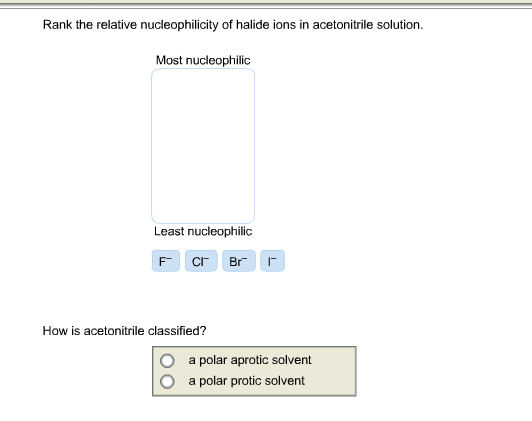
Rank The Relative Nucleophilicity Of Halide Ions In Acetonitrile Solution
Alright, gather 'round, folks! Ever feel like some people just have a knack for showing up exactly when you need them? Like that friend who always brings the perfect snack, or the one who knows how to fix your wonky Wi-Fi without you even asking? Well, in the wonderfully weird world of chemistry, we have something similar. We call them nucleophiles. Think of them as the chemical equivalent of those super helpful people. They’re eager to share their electrons, like a generous soul offering you their last cookie. And today, we’re going to chat about a specific crew of these electron-donating heroes: the halide ions.
Now, when we say "halide ions," we're talking about the plain old halogens – fluorine (F), chlorine (Cl), bromine (Br), and iodine (I) – but they’ve gone and grabbed an extra electron. So, they’re F-, Cl-, Br-, and I-. They're like the friendly neighborhood characters of the periodic table, always ready to lend a hand (or, you know, an electron). But here's the kicker: they don't all play the same game. Some are way more enthusiastic about sharing than others.
And where are they doing all this sharing? Well, we’re not talking about a bustling party scene here. Our setting is a solvent called acetonitrile. Imagine it as a particularly chill hangout spot for these ions. Acetonitrile is a bit like that coffee shop where everyone knows your name and the Wi-Fi is surprisingly reliable. It’s polar, meaning it has a little bit of a positive and a little bit of a negative end, kind of like a tiny magnet. This polarity helps it to solvate, or surround, these ions, making them feel comfortable and, crucially, influencing how they interact with each other and with other molecules.
So, why do we even care about ranking their nucleophilicity? It’s not just some abstract lab experiment for nerds in white coats, I promise! Understanding this ranking helps us predict how chemical reactions will go. Think of it like knowing which of your friends is most likely to volunteer for the awkward task, or which one will always volunteer to drive. In chemistry, it tells us which halide ion is going to be the most aggressive, the most eager to attack a molecule that’s looking a little electron-deficient. This is super important for designing new materials, making medicines, or even just figuring out how stuff breaks down in the environment.
Now, the common misconception, the one that might pop into your head if you’re just starting out, is that the smaller the ion, the more it should be able to pack a punch. Like a tiny but fierce chihuahua. You'd think fluorine, being the smallest halogen ion, would be the absolute champion nucleophile. It's got all its negative charge concentrated in a tiny little package, right? It should be able to get in there and electron-share like nobody's business!
But alas, dear reader, that's where the universe loves to throw us a curveball. In the cozy confines of acetonitrile, this neat little idea about size and nucleophilicity goes out the window. It's like bringing your chihuahua to a dog park and finding out it's terrified of the bouncy castle, while the big, goofy Golden Retriever is jumping on it with glee. The actual ranking is a bit of a surprise, and it all comes down to how well our solvent, acetonitrile, plays its part.
The Solvent Factor: Acetonitrile's Cozy Embrace
This is where acetonitrile shows its true colors. Acetonitrile is a polar aprotic solvent. Now, that’s a mouthful, I know. Let's break it down. "Polar" we covered – tiny magnet. "Aprotic" means it doesn't have any really "acidic" hydrogens hanging around. Think of it like a solvent that's really good at cuddling, but doesn't have any sharp edges to get in the way. It’s like a really comfy sofa for these ions.
When these halide ions are floating around in acetonitrile, the polar nature of the solvent starts to interact with them. The positive ends of the acetonitrile molecules are attracted to the negative charges of the halide ions. This is called solvation, and it's like giving each ion a warm, fuzzy hug. But here’s the twist: not all ions get the same kind of hug.
Smaller ions, like fluoride (F-), have their negative charge packed much more tightly. Imagine trying to hug a basketball versus trying to hug a bowling ball. You can get your arms right around the basketball. Acetonitrile molecules can get in super close to the fluoride ion and give it a really strong, tight solvation shell. It’s like being wrapped up in a thousand tiny blankets. This tight hugging, this strong solvation, actually hinders the fluoride ion. It’s so cozy and comfortable, it’s a bit lazy to go out and share its electrons.
Think of it like this: you’re at a party, and you’re wearing your absolute favorite, most comfortable sweater. You’re feeling so good, so warm and snug, that the thought of getting up to grab more snacks seems like a monumental effort. Fluoride is wearing its ultimate cozy sweater in acetonitrile.
Now, consider the larger halide ions, like iodide (I-). These guys are like bowling balls. The negative charge is spread out over a much larger area. Acetonitrile molecules can still surround them, but they can't get quite as close or as tightly packed. The solvation shell around iodide is much weaker, more diffuse. It's like getting a gentle pat on the shoulder instead of a full-on bear hug.
So, the iodide ion is much less solvated. It's not as cocooned, not as comfortable. This means its negative charge is more exposed, more readily available to interact with other molecules. It’s like being at that same party, but you’re wearing a slightly less comfortable shirt. You’re not going to stand around all day; you’re more likely to get up and mingle, maybe even go get that snack for someone else.
The Ranking Revealed: A Surprising Order
Because of this solvation effect in acetonitrile, the order of nucleophilicity is flipped on its head compared to what you might expect if you just looked at size. The largest, least solvated ion is the most nucleophilic. It’s like the underdog story of the chemical world!
So, in acetonitrile, the order of nucleophilicity, from weakest to strongest, goes like this:
- Fluoride (F-): The queen of cozy. So tightly solvated, she’s practically napping. Weakest nucleophile.
- Chloride (Cl-): A bit more comfortable than iodide, but still feeling the acetonitrile snuggle.
- Bromide (Br-): Getting less cuddled, starting to feel a bit more energetic.
- Iodide (I-): The most free spirit! Least solvated, most eager to share. Strongest nucleophile.
Isn’t that wild? You’d think the tiny, energetic one would be the most eager, but it’s the spread-out, less hugged one that’s ready to party. It’s like the difference between someone who’s so comfortable on their couch they’ll never leave, and someone who’s a bit restless and always looking for the next adventure. Iodide is the adventurer.
Why This Matters in the Real World (Sort Of!)
Okay, so you might be thinking, "That's cool, but how does this relate to my actual life, beyond understanding why my dog loves a good belly rub more than a stern lecture?" Well, this principle, the influence of solvent on nucleophilicity, is fundamental to countless chemical processes.
Take organic synthesis, the art of building complex molecules. When chemists are trying to create new drugs, plastics, or fragrances, they often rely on nucleophilic substitution reactions. This is where a nucleophile comes in and replaces another atom or group on a molecule. Knowing which halide ion will be the most effective nucleophile in a given solvent, like acetonitrile, allows them to design reactions for maximum efficiency and yield.
Imagine you’re baking a cake and you need to add chocolate chips. You have a whole bag of chips, but some are stuck together, and some are loose. You want the loose ones to fall into the batter easily, right? Iodide is like the perfectly separated chocolate chips, ready to go. Fluoride is like a clump of chips that you’d have to break apart first.

This understanding also pops up in biochemistry. Many biological processes involve molecules interacting with each other, and the principles of nucleophilicity and solvation play a role. While the biological environment isn’t exactly acetonitrile, the core idea that the surrounding medium can dramatically affect reactivity holds true.
And even in environmental chemistry, understanding how ions behave and react is crucial. For instance, when pollutants interact with different solvents or water, their reactivity can change, influencing how quickly they break down or move through the environment. It’s like knowing if a particular type of mud will cling to your shoes easily or slide right off.
A Little More About Acetonitrile's Role
Let's give acetonitrile a little more credit. It’s not just some passive bystander. It’s an active participant in this electron-sharing drama. Its polar nature, with the partially positive hydrogen atoms on its methyl group (CH3) and the partially negative nitrogen atom, creates a specific environment.
The nitrogen atom of acetonitrile, being electronegative, is good at interacting with positive centers. The hydrogens on the methyl group, while not "acidic" like in water, can still form weak attractive forces, especially with anions.
The key here is that acetonitrile is better at solvating cations than anions. This means it’s pretty good at surrounding and stabilizing positive ions. When it comes to anions, especially the smaller ones like fluoride, it can still do a job, but it’s like trying to hold onto a tiny, super-charged balloon. It grips it tightly. For the larger, more diffuse anions like iodide, the grip is looser. The acetonitrile molecules are still there, like an entourage, but they’re not suffocating the ion.
When the Solvent Changes, Everything Changes!
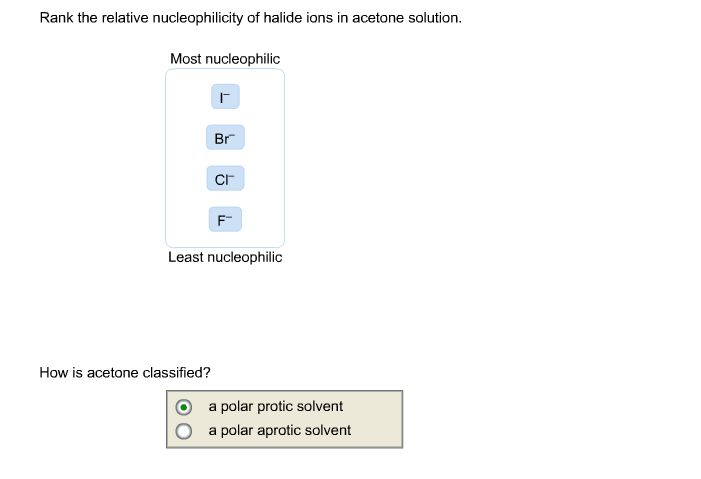
It's crucial to remember that this ranking of halide nucleophilicity is specific to acetonitrile. If you were to put these halide ions in a different solvent, say water, the story would change dramatically!
Water is a polar protic solvent. It has those "acidic" hydrogens. Water molecules can hydrogen bond very strongly with anions. And guess which anion is the best at forming strong hydrogen bonds? That's right, fluoride (F-). It’s like it’s made for it. In water, fluoride gets an even tighter, more robust solvation shell, making it an even weaker nucleophile.
In protic solvents like water, the nucleophilicity trend for halides actually reverses. Fluoride becomes the weakest, and iodide remains the strongest, but for different reasons related to hydrogen bonding strength. So, the solvent is like the director of a play – it can completely change the performance of the actors!
This is why chemists are so meticulous about choosing the right solvent. It's not just about dissolving the reactants; it's about creating the precise environment needed for the reaction to proceed as desired. It's like choosing the right music for a dance – the same dancer can perform very differently depending on the rhythm and tempo.
In Conclusion: The Comforts of Acetonitrile
So, there you have it. In the slightly peculiar, yet incredibly important, world of acetonitrile solutions, the halide ions put on a show where the biggest, least restrained performer steals the spotlight. Fluoride, the smallest and seemingly most potent, gets a bit too comfortable, wrapped up in the solvent’s embrace, and becomes a rather sluggish nucleophile. Chloride and bromide are somewhere in the middle, and iodide, the largest and least tightly held, is the undisputed champion of electron-sharing in this particular setting.
It's a reminder that appearances can be deceiving, and that context is everything. Just like a shy person might blossom in a supportive environment, or a bold personality might be reined in by strict rules, the reactivity of these ions is profoundly shaped by their surroundings. And the humble solvent, acetonitrile, plays the role of the genial host, influencing all the interactions at the chemical party. Keep this in mind the next time you're marveling at how efficiently a chemical reaction proceeds – there's a whole world of subtle interactions happening, all thanks to the right solvent and the willingness of some ions to be a little more adventurous than others.
