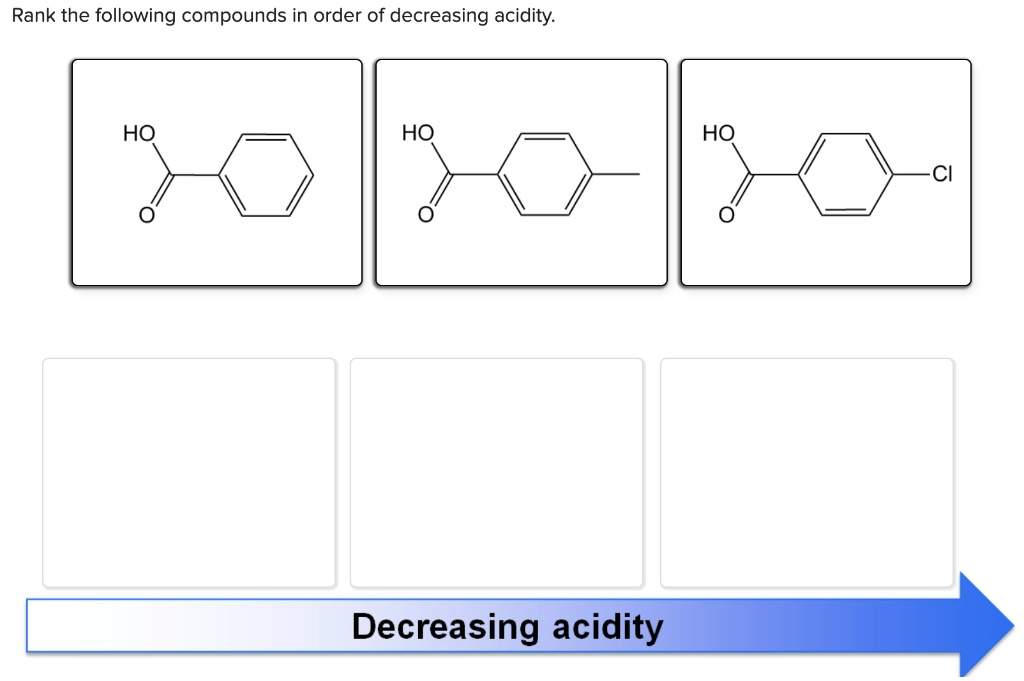
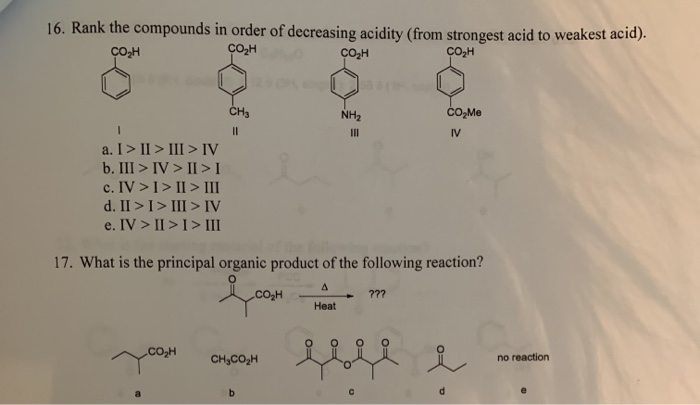
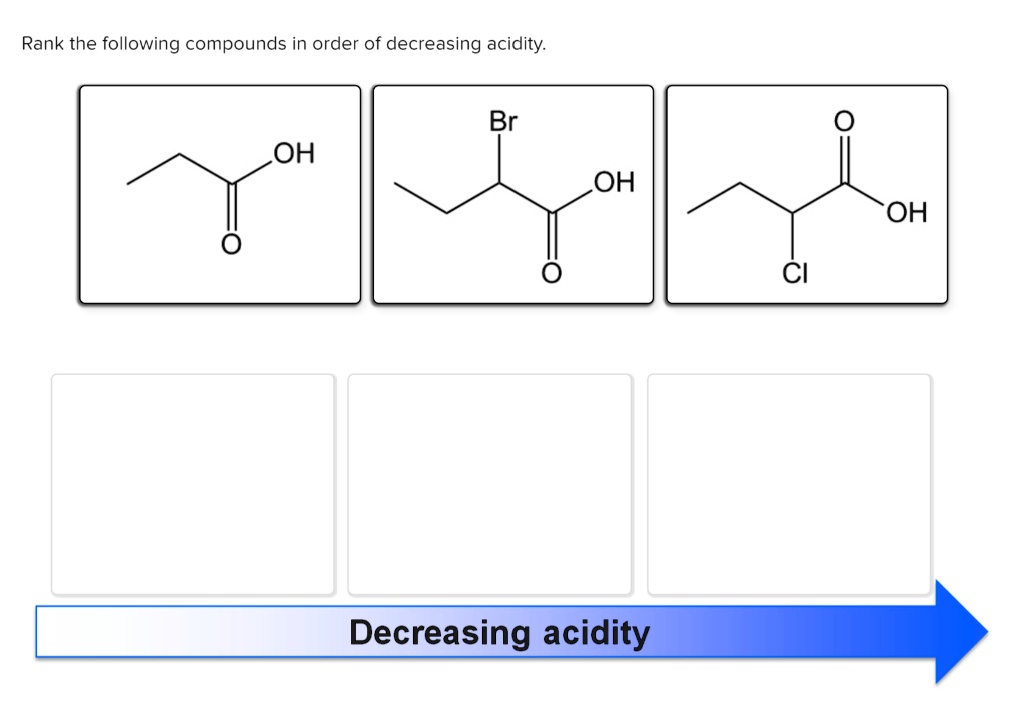
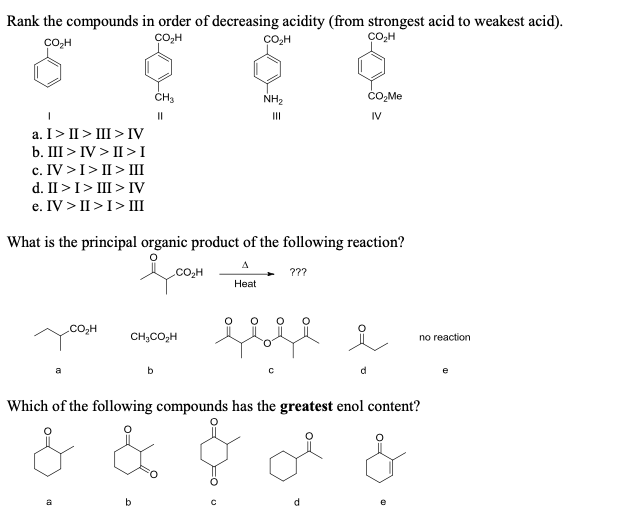
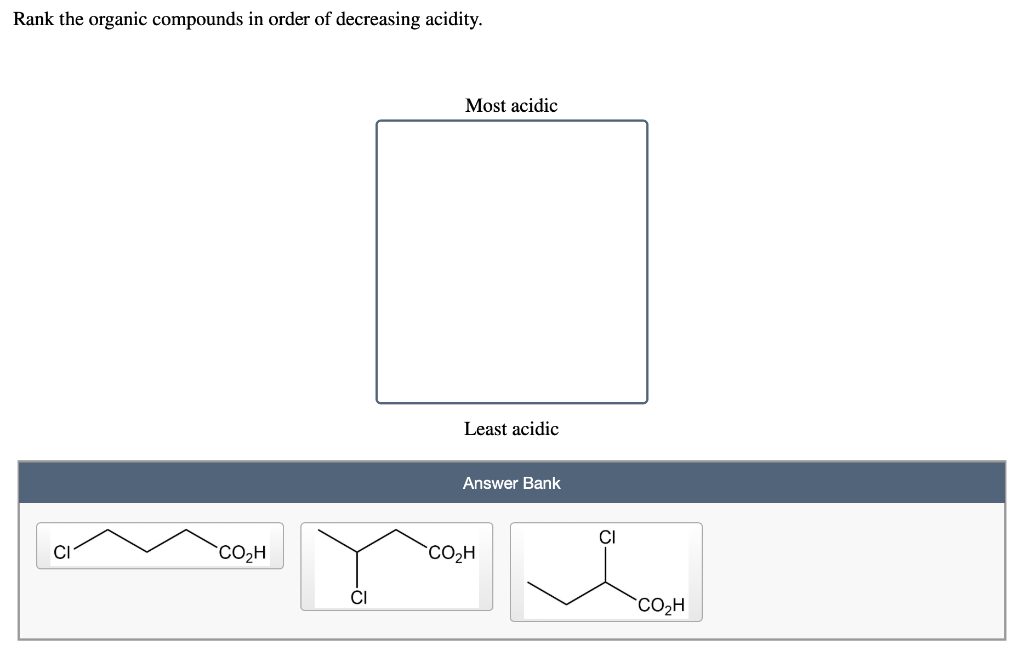
Rank The Organic Compounds In Order Of Decreasing Acidity.
Hey there, fellow seekers of that sweet, balanced life! Ever find yourself staring at a perfectly brewed cup of coffee, wondering about the subtle science behind that invigorating bitterness? Or maybe you’ve been experimenting in the kitchen, a little bit of lemon juice here, a dash of vinegar there, and you’ve started to notice that some ingredients just have more zing than others. Well, pull up a comfy chair and let’s dive into a topic that might sound a tad academic but is surprisingly relevant to our everyday adventures: organic compounds and their acidity. Think of it as a backstage pass to understanding why some things pucker your lips and others are as smooth as a jazz solo.
Now, don't let the fancy word "organic compounds" throw you off. We're talking about the building blocks of life, the stuff that makes up everything from the apple in your lunchbox to the very air you breathe. And within this vast, molecular universe, there's a concept called acidity, which essentially measures how readily a compound is willing to give up a little proton – a hydrogen atom minus its electron. It’s like a generosity score for molecules!
So, how do we sort these generous molecules? We rank them! It’s like putting together a playlist of your favorite songs, but instead of BPM, we're looking at acid strength. This isn't just some sterile lab concept, either. Understanding this order can actually inform your choices, from choosing the best marinade for your tofu to appreciating the tangy kick in your favorite kombucha.
The Great Acidity Showdown: Who’s Who?
Let's start at the top, with the undisputed heavyweights of acidity. Think of these as the rockstars of the proton-donating world. At the pinnacle, we have our carboxylic acids. These guys are like the generous philanthropists of the molecular scene. Their chemical structure, featuring a carboxyl group (-COOH), makes them super eager to release that proton. They're the backbone of so many of our beloved sour experiences.
Take citric acid, for instance. This is the superstar in lemons, oranges, and limes. It’s not just about the pucker-power; citric acid plays a vital role in our metabolism, helping us convert food into energy. Pretty cool, right? It’s the reason your lemonade is so delightfully tart. And then there's acetic acid, the main component of vinegar. That distinct sharp smell and taste? That’s acetic acid saying hello. It’s a staple in kitchens worldwide, from salad dressings to pickling. Imagine a world without the zing of vinegar – a rather bland existence, wouldn't you say?
Next in line, still strong but perhaps a tad less flamboyant, are the sulfonic acids. These are often found in synthetic compounds and detergents, but they are incredibly strong acids. They’re the silent workhorses, often used in industrial processes. Think of them as the highly efficient, no-nonsense producers who keep things running smoothly behind the scenes.
Then we move down the ladder to the phenols. These compounds have a hydroxyl group (-OH) attached to an aromatic ring (think benzene, like in the structure of many fuels and plastics, but in this context, it’s part of a molecule with a -OH group). They're less acidic than carboxylic acids but still possess a noticeable tang. Phenol itself is an interesting one; it’s used in disinfectants and in the production of plastics, but in a diluted form, it has a slightly medicinal scent and taste. You might encounter derivatives of phenols in some natural extracts, contributing to subtle flavors and aromas.
Moving further, we encounter alcohols. Ah, alcohols! The lifeblood of celebrations and sometimes, the cause of regrettable decisions. But chemically speaking, simple alcohols like ethanol (the stuff in your wine and beer) and methanol (found in fuels and some industrial solvents – definitely not for drinking!) are significantly weaker acids than phenols or carboxylic acids. They're like the laid-back friends at the party, much less eager to share their protons. Their acidity is so low that in everyday terms, we often don't even perceive them as acidic. It’s the imbalance of acidity and alkalinity that affects our bodies, not the subtle acidity of ethanol on its own.
Finally, at the very bottom of our acidity ranking for commonly discussed organic compounds, we have water. Yes, even water, the universal solvent, has a tiny bit of acidity. But it’s incredibly weak. Think of it as the zen master of the group, barely willing to part with anything. Its self-ionization (forming H+ and OH-) is minimal, which is precisely why it’s so neutral and essential for life.
The Science Behind the Zing: Why the Difference?
So, what gives these molecules their varying degrees of proton-giving eagerness? It all comes down to stability. When an acid donates a proton, it forms what's called a conjugate base. The more stable this conjugate base is, the more willing the original acid is to let go of its proton. It’s like a happy breakup – if both parties can move on and thrive independently, the separation is easier.
In carboxylic acids, the negative charge on the conjugate base is spread out over multiple atoms, making it very stable. This is thanks to a phenomenon called resonance. Think of it like sharing the load; if a burden is spread out, it's easier to bear. This resonance stabilization is a big reason why carboxylic acids are such strong contenders in the acidity game.
For sulfonic acids, similar electron-withdrawing effects stabilize the conjugate base even further, making them exceptionally strong. Phenols have some resonance stabilization, but it’s not as pronounced as in carboxylic acids, hence their moderate acidity.
Alcohols, on the other hand, don't have the same resonance stabilization for their conjugate bases. The negative charge is more localized, making it less happy to exist. This makes alcohols much weaker acids. Water, as we mentioned, is the epitome of minimal charge distribution, making it the least acidic among our contenders.
Practical Magic: Acidity in Your Everyday
Understanding this hierarchy isn't just for lab coats and beakers. It can actually enhance your daily life and make you a more informed consumer and cook.
Culinary Adventures: Ever wondered why lemon juice tenderizes meat so effectively? It’s the citric acid at work, breaking down proteins. This is why it’s a key ingredient in marinades, especially for tougher cuts. Similarly, the acetic acid in vinegar is a fantastic preservative and adds that essential tang to dressings and pickling brines. Knowing that carboxylic acids are generally stronger can help you understand why a splash of lemon might provide a more intense sour note than, say, a splash of wine (which contains a mix of acids, but often weaker ones than pure citric or acetic). If you’re making a glaze and want a gentle sweetness with a hint of tang, you might reach for a milder acid or use less of a stronger one.
Skincare Secrets: Many skincare products leverage the properties of organic acids. Alpha-hydroxy acids (AHAs), a type of carboxylic acid found in fruits (like glycolic acid from sugar cane and lactic acid from sour milk), are popular exfoliants. They gently dissolve the bonds between dead skin cells, revealing brighter, smoother skin. Understanding that these are indeed acids helps you appreciate why you might experience a slight tingling sensation and why it’s important to follow product instructions carefully. They're designed to be used in controlled, diluted amounts.
Home Hacks: Need to descale your coffee maker? That build-up of mineral deposits is often tackled with vinegar (acetic acid). Its acidity helps to break down those stubborn mineral layers. For tougher jobs, or in commercial settings, stronger acids might be employed, but for home use, the moderate strength of acetic acid is often just right – effective without being overly harsh.
The pH Scale: A Familiar Friend
You've probably seen it: the pH scale, ranging from 0 (very acidic) to 14 (very alkaline), with 7 being neutral. This scale is directly related to the concentration of hydrogen ions (H+) in a solution. The more readily a compound releases H+ (our proton), the lower its pH, and the more acidic it is. So, that sour lemon juice has a low pH, while baking soda (an alkali) has a high pH.
Our bodies are finely tuned ecosystems that thrive within a very narrow pH range, particularly our blood. This is where those weak organic acids in our diet, like those found in fruits and vegetables, are generally well-managed by our bodies' buffering systems. However, consuming excessively acidic foods or drinks can put a strain on these systems. It's all about balance, a concept we often strive for in our lifestyles, and it holds true at the molecular level too!
Fun Facts and Quirky Connections
Did you know that formic acid, a simple carboxylic acid, is what ants use to defend themselves? That burning sensation when an ant bites or stings is its way of saying, "Back off!" It's a primitive but effective defense mechanism.
And what about the smell of rain? While rain itself is slightly acidic due to dissolved carbon dioxide, the earthy smell after rain, called petrichor, is largely due to compounds called geosmins released by soil bacteria. However, the slight acidity of the rain can play a role in releasing these compounds from the soil.
In the world of wine making, the precise acidity of the grapes, primarily from tartaric acid (another carboxylic acid), is crucial for flavor, balance, and preservation. Winemakers carefully monitor and adjust acidity to achieve the desired taste profile. It's a testament to how these molecular properties translate into sensory experiences we cherish.
Think about your favorite sourdough bread. The tangy flavor comes from the lactic and acetic acids produced by the wild yeast and bacteria in the starter. It's a delicious example of controlled fermentation and the power of organic acids.
A Little Reflection: The Symphony of Balance
As we wrap up this exploration, it’s fascinating to see how the seemingly abstract world of organic chemistry mirrors our broader pursuit of balance and well-being. We strive for balanced diets, balanced work-life schedules, and balanced emotional states. Similarly, in the molecular realm, the strength of an acid is all about its willingness to achieve a state of balance through proton donation and the stability of its resulting form.
From the tartness of a grapefruit that invigorates your morning to the gentle cleaning power of vinegar, these organic compounds and their inherent acidity are woven into the fabric of our daily lives. They're not just chemical formulas; they're the flavor enhancers, the cleaning agents, the biological facilitators, and the subtle influences that shape our experiences. So next time you enjoy a zesty salad dressing or appreciate the complex flavors in a dish, take a moment to acknowledge the unsung heroes – the organic compounds and their remarkable journey up and down the acidity scale. It’s a symphony of balance, playing out in every bite, every cleaning spray, and every breath we take.
