Rank The Leaving Groups Below From Worst To Best.
Welcome, fellow adventurers, to a thrilling quest through the fascinating world of chemical reactions! Ever wondered what makes some reactions zip along with lightning speed while others seem to take an eternity? Today, we're diving into the nitty-gritty, exploring the unsung heroes (or villains, depending on your perspective!) that dictate the pace and outcome of countless chemical transformations: leaving groups. Forget dusty textbooks and intimidating equations; we're making this fun, accessible, and surprisingly useful.
Why Are Leaving Groups Such a Big Deal?
Think of a chemical reaction as a dance. For the dance to happen, someone needs to gracefully exit the floor to make space for new partners. In chemistry, these "exiting partners" are our leaving groups. They are atoms or groups of atoms that detach from a molecule during a reaction, taking a pair of electrons with them. The better a group is at leaving, the more likely and faster a reaction will occur. It's like a party – the more enthusiastic people are to leave when the night is over, the smoother the cleanup!
Understanding leaving groups is not just a niche interest for hardcore chemists; it's a fundamental concept that unlocks the secrets of organic chemistry. Why is this useful? Well, imagine you're trying to synthesize a new medicine, develop a more efficient industrial process, or even understand how your body digests food. In all these scenarios, predicting and controlling chemical reactions is key. And what controls many reactions? You guessed it: the quality of the leaving group!
The purpose of ranking leaving groups is to provide a clear hierarchy of their "goodness." A "good" leaving group is one that is stable on its own after it departs. This stability is usually related to how weak the bond was to the original molecule and how well the leaving group can accommodate the negative charge it acquires. We’re basically ranking them on their willingness to bail from the party!
The benefits of mastering this concept are immense. For students, it's a cornerstone of understanding reaction mechanisms, particularly in substitution and elimination reactions. For researchers, it's a tool for designing experiments and predicting product yields. For anyone curious about the molecular world, it's a satisfying piece of the puzzle that explains so much about why things happen the way they do. So, without further ado, let's get to the main event!
The Grand Ranking: From "Meh" to "Magnificent!"
We're going to rank these leaving groups from the absolute worst (the ones that hang on for dear life) to the absolute best (the ones that can't wait to get out). This ranking is based on their inherent stability and their ability to stabilize a negative charge.
Starting at the bottom, we have groups that are incredibly stable before they leave, which makes them reluctant to leave at all. Think of someone who is perfectly happy at home and has no desire to go out.
The Absolute Worst: -OH (Hydroxide) and -OR (Alkoxide)These guys are notoriously bad leaving groups. Why? Because when they leave, they form hydroxide ions (OH-) or alkoxide ions (OR-). These are strong bases and are quite unstable with a negative charge. They'd much rather stay attached! Reactions involving these as leaving groups are slow and often require harsh conditions or catalysts to force them to depart.
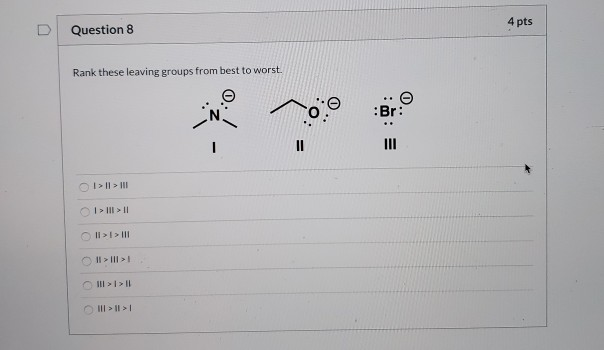
Solved 4 pts D Question 8 Rank these leaving groups from | Chegg.com
Moving up a notch, we have groups that are slightly better, but still not ideal. They're like the friend who might consider leaving the party if you really twist their arm.
Still Not Great: -NH2 (Amide) and -F (Fluoride)Amide ions (NH2-) are also strong bases and don't like to be on their own. Fluoride (F-) is a bit better than OH- because fluorine is more electronegative, but it's still a relatively strong base and a poor leaving group. The bond to fluorine is also quite strong.
Now we're getting into the territory of "okay, this might work." These are the groups that are starting to think about finding their own space.
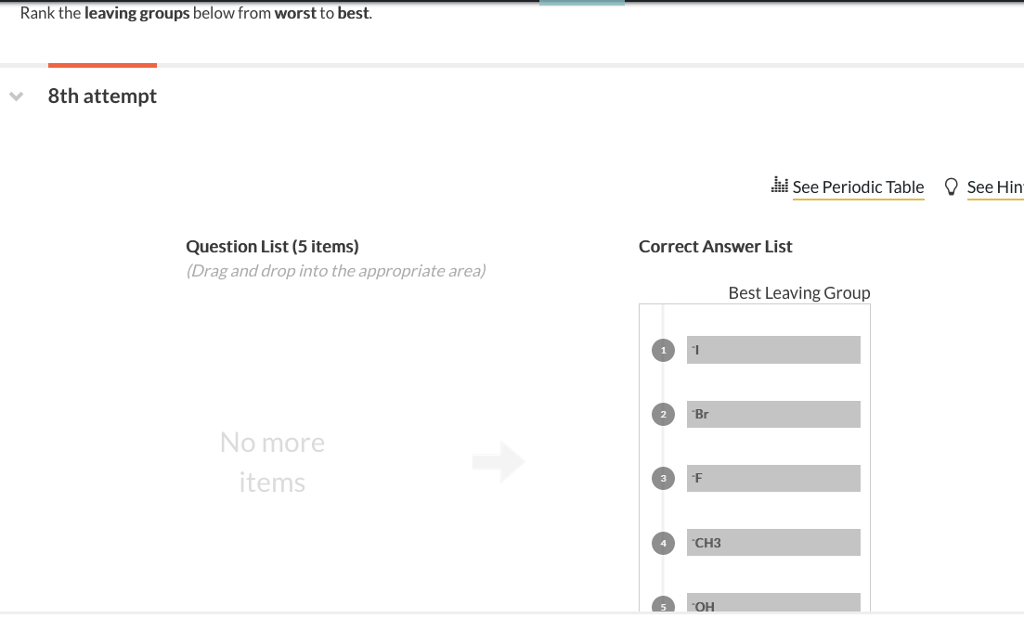
The Mediocre Crew: -Cl (Chloride), -Br (Bromide), and -I (Iodide)Ah, the halides! These are significantly better leaving groups. As you go down the halogen group (Cl, Br, I), their ability to leave improves. This is because larger atoms can spread out the negative charge more effectively, making them more stable. Iodide is the best among these three, followed by bromide, then chloride. They're like the reliable guests who will leave when it's getting late.
This next tier includes some groups that are quite good and facilitate reactions readily.
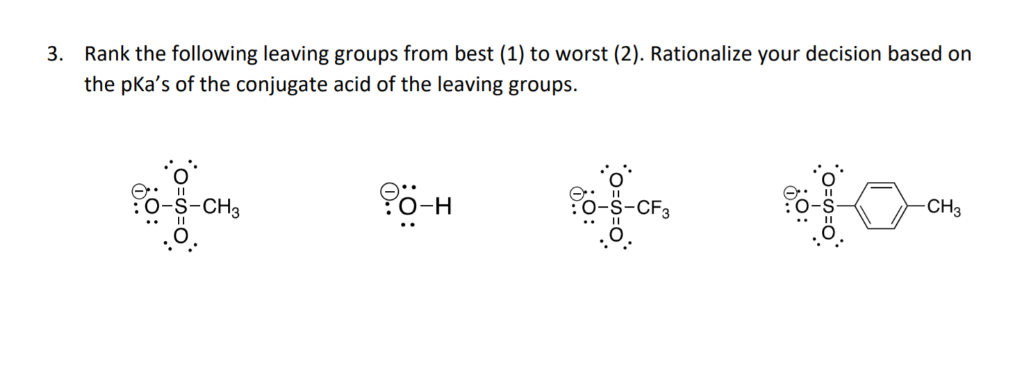
Getting Good: -S-R (Thiolate), -O-SO2-R (Sulfonates like Tosylate and Mesylate)Thiolates are good leaving groups because sulfur is larger than oxygen and can better stabilize a negative charge. But the real superstars here are the sulfonates. Groups like tosylate (-OTs) and mesylate (-OMs) are formed when alcohols are reacted with sulfonyl chlorides. The leaving group part is actually the entire sulfonate ester. The reason these are so fantastic is that the negative charge on the oxygen atoms in the sulfonate group is highly delocalized through resonance, making it incredibly stable. Think of them as the early birds who have their exit strategy planned out perfectly!
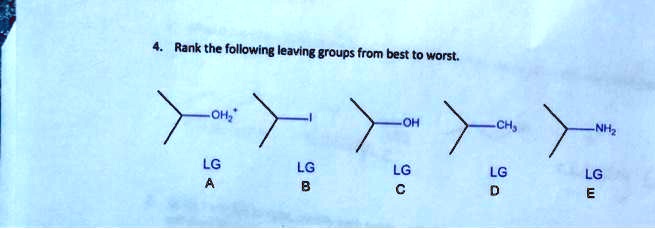
Solved Rank the following leaving groups from best to worst. | Chegg.com
And now, for the ultimate champions, the ones that are practically begging to leave and make a reaction happen!
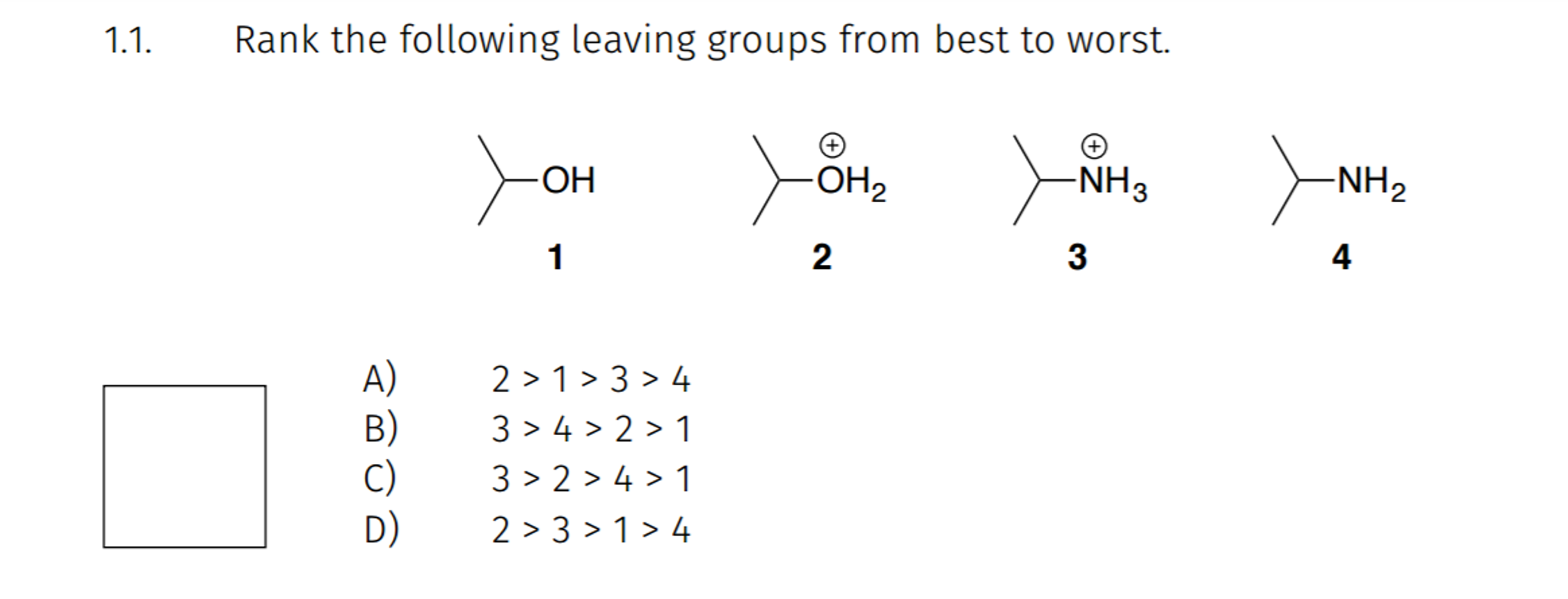
The Best of the Best: Water (H2O) and Nitrogen Gas (N2)When an alcohol is protonated (e.g., by an acid), the -OH group becomes -OH2+. This is an excellent leaving group because when it leaves, it forms a neutral, very stable molecule: water! Water is an incredibly stable entity and readily separates. Even better is nitrogen gas (N2). Certain compounds, like diazonium salts, can expel nitrogen gas. Since nitrogen gas is incredibly stable and has a triple bond, it's the ultimate leaving group. Reactions that produce nitrogen gas are often very fast and vigorous. These are the rockstars of leaving groups, exiting with a bang!
So there you have it! From the reluctant hydroxide to the jubilant nitrogen gas, ranking leaving groups gives us a powerful insight into the world of chemical reactivity. It’s a fun way to understand why some molecules are more dynamic than others and how chemists can manipulate these principles to create amazing things. Keep an eye out for these departing guests in your next chemical encounter!