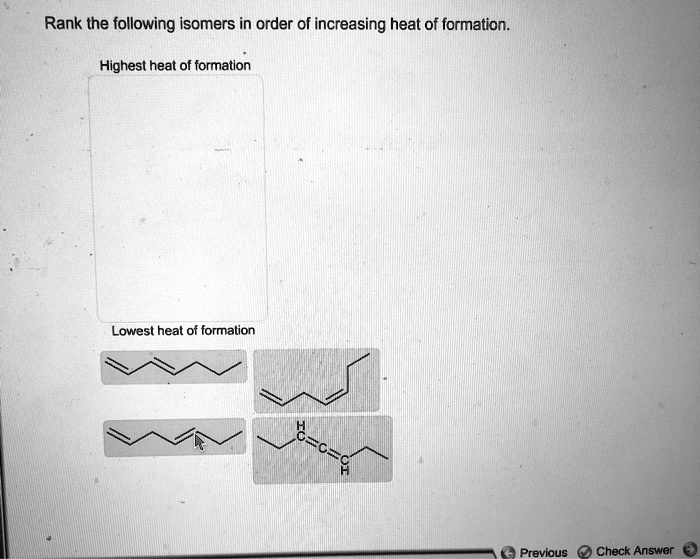
Rank The Isomers In Order Of Increasing Heat Of Formation.
Hey there, curious minds! Ever wonder why some things in life just feel more stable than others? Like why your favorite comfy couch stays put, but that wobbly chair at the diner always seems ready to give you a surprise adventure? Well, in the wonderful world of chemistry, we have a concept that's kind of like that, and it's called the heat of formation. Don't let the fancy name scare you off; it's actually pretty relatable, and understanding it can be surprisingly cool!
So, what exactly is heat of formation? Imagine you're building something, like a magnificent LEGO castle. You start with a pile of individual LEGO bricks. To get those bricks into a fancy castle, you have to put in a bit of effort, right? You're connecting them, stacking them, making them all neat and tidy. The heat of formation is a way of measuring how much energy (or heat) was either released or absorbed when a substance was created from its basic building blocks, its elemental components, in their most stable form. Think of it as the energy cost of "building" that molecule.
Now, here's where it gets interesting, especially when we talk about isomers. Isomers are like twins, or maybe even triplets or quadruplets, that have the same ingredients but are put together in a slightly different arrangement. They have the same number of atoms of each element, but those atoms are connected in a different pattern. It's like having the same set of LEGO bricks but building two completely different things. You might have a little car and a tiny house, all from the same box of bricks!
Let's take a common example: butane. You've probably heard of it – it's used in lighters and camping stoves. Butane has a chemical formula of C4H10. Now, there are actually two common ways these four carbon atoms and ten hydrogen atoms can be arranged. We have n-butane, which is like a straight line of carbon atoms with hydrogens attached. And then we have isobutane (or 2-methylpropane), which is like a central carbon atom with three other carbon atoms branching off it, and then hydrogens filling in the gaps.
These two molecules, n-butane and isobutane, are isomers. They have the same LEGO bricks, but they're built differently. And here's the kicker: because they're built differently, they have different heats of formation. This means that when they were "built" from their elemental components (carbon and hydrogen), different amounts of energy were involved.
Why Should You Even Care About This LEGO-Building Energy?
Okay, so it's about energy and building molecules. So what? Well, think about it this way: a substance with a lower heat of formation is generally more stable. It's like that perfectly balanced, sturdy chair you love. It doesn't want to fall apart or change easily. It's content with its arrangement.
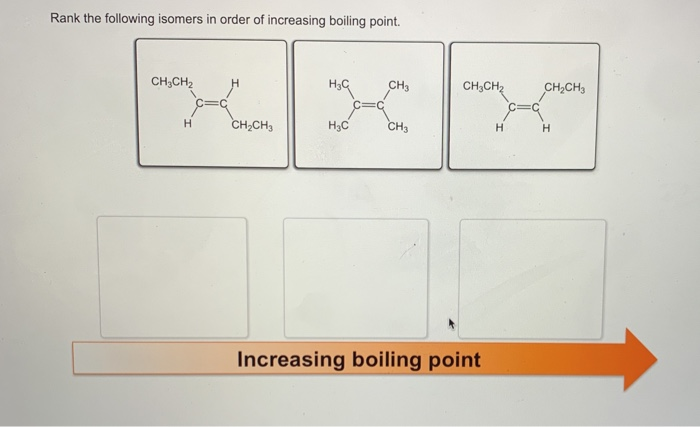
On the other hand, a substance with a higher heat of formation is a bit less stable. It's more like that wobbly chair. It's got a bit more "oomph" to it, a bit more potential energy ready to be released. This can mean it's more likely to react, to break apart, or to change into something else. It’s like having a tower of dominoes that’s just begging to be toppled.
So, when we rank isomers in order of increasing heat of formation, we're essentially ranking them from the most stable to the least stable. It's like organizing your LEGO creations from the ones you're sure will survive a mild earthquake to the ones that might spontaneously deconstruct if you breathe on them too hard.
Let's Talk Butane Twins: n-Butane vs. Isobutane
Let's go back to our butane friends. In general, when we look at these kinds of branched vs. straight-chain isomers, the more branched an isomer is, the more stable it tends to be. Think of it like packing things into a box. If you have a bunch of long, skinny items, they might be a bit awkward and less compact. But if you have some more compact, branched items, they can often fit together more snugly and efficiently.
For n-butane and isobutane, isobutane is the more branched one. It has that central carbon atom with three other carbons attached. This more compact, symmetrical arrangement often leads to stronger intermolecular forces (the little "stickiness" between molecules) and a more stable overall structure. Because it's more stable, it actually has a lower heat of formation.
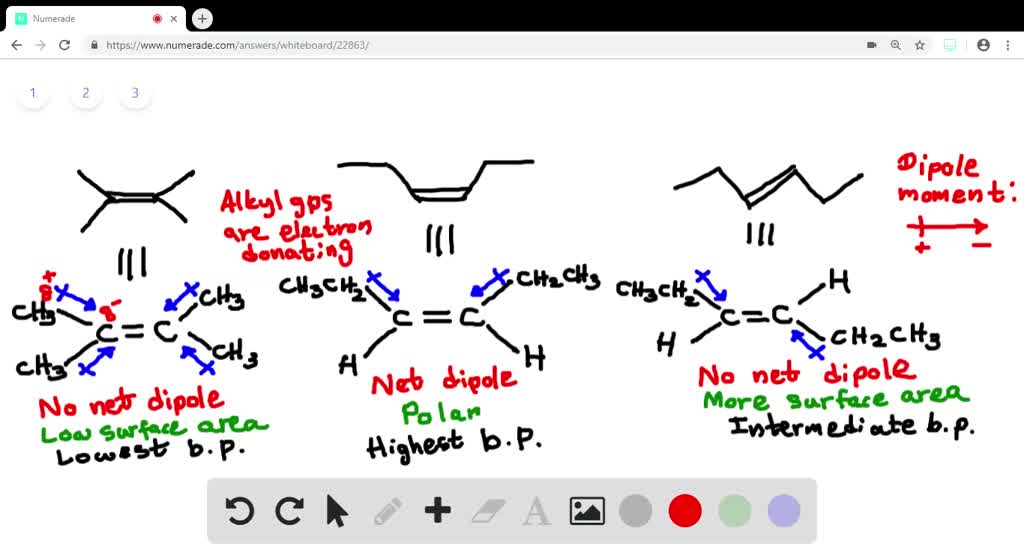
So, if we were to rank them, isobutane would come first in our order of increasing heat of formation. It’s the chill, laid-back twin, happy in its stable configuration. Then comes n-butane, which is a little less stable and has a slightly higher heat of formation.
This means that to create isobutane from its basic elements, you'd generally need to put in a bit less energy compared to creating n-butane. Or, if they were to break down, n-butane would release a bit more energy because it has more "stored tension" in its structure compared to isobutane.
Why Does This Matter in the Real World? (Besides Super Stable LEGOs)
This concept of heat of formation and stability is super important in many areas. In the world of fuels, for example, stability plays a big role in how efficiently and safely a fuel burns. Different isomers of the same fuel can have slightly different burning characteristics and energy outputs.
Think about gasoline. It's a complex mixture of hydrocarbons, many of which are isomers. The specific blend of these isomers affects the octane rating and how the fuel performs in your car's engine. A more stable isomer might burn more cleanly, while a less stable one might be more prone to knocking in the engine. So, chemists and engineers carefully choose which isomers to include to get the best performance!
Even in drug development, the shape and stability of molecules (which are influenced by their heat of formation) can determine how they interact with the body and whether they're effective medicines or potential toxins. A slightly different arrangement of atoms can make a molecule work wonders or do absolutely nothing, or even cause harm.
It's also crucial in understanding chemical reactions. If you know the heats of formation of the starting materials and the products, you can predict whether a reaction will release heat (exothermic) or absorb heat (endothermic). This is vital for controlling industrial processes and ensuring safety.
Putting It All Together: The Order of Increasing Heat of Formation
So, let's imagine we have a few isomers of a compound, and we want to line them up from least energetic (most stable) to most energetic (least stable). We're looking for the order of increasing heat of formation.
The isomer with the lowest heat of formation is the most stable. It's like the one that's perfectly content in its spot, not wanting to change. It required the least "effort" to build from its elemental parts.
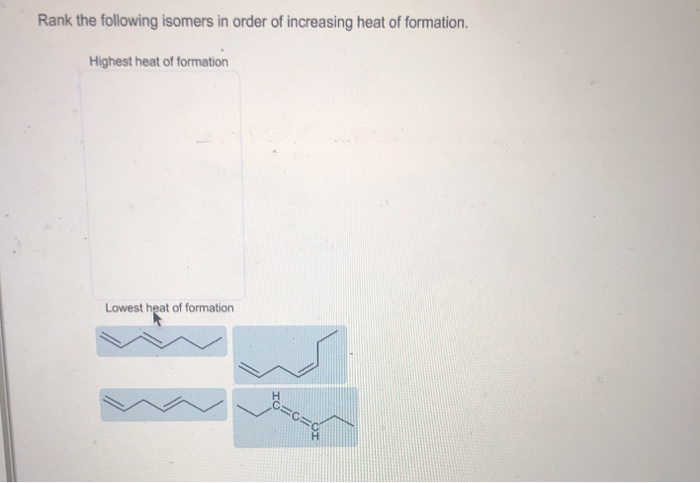
As we move up the list, each subsequent isomer will have a higher heat of formation. This means it's a bit less stable, a bit more prone to change, and required a bit more energy to be put together initially. It’s like moving from a perfectly stacked pile of blocks to one that’s starting to lean a little.
The isomer with the highest heat of formation is the least stable. This one is the most "energetic," the most eager to react or change. It's like that precarious Jenga tower that's just one wobble away from disaster!
So, the general rule we often see is that more branching leads to greater stability and a lower heat of formation. This is a handy little trick to remember when you encounter different isomers. Think of those incredibly branched molecules as the "chill vibes" of the chemical world – they're just happy to be where they are and don't need much fuss.
It's fascinating how these tiny differences in how atoms are arranged can lead to such big differences in how substances behave. So, the next time you're enjoying the power of a lighter or filling up your car, give a little nod to the heat of formation and the stability of those isomeric molecules. They're working hard, in their own unique ways, to make your everyday life happen!
