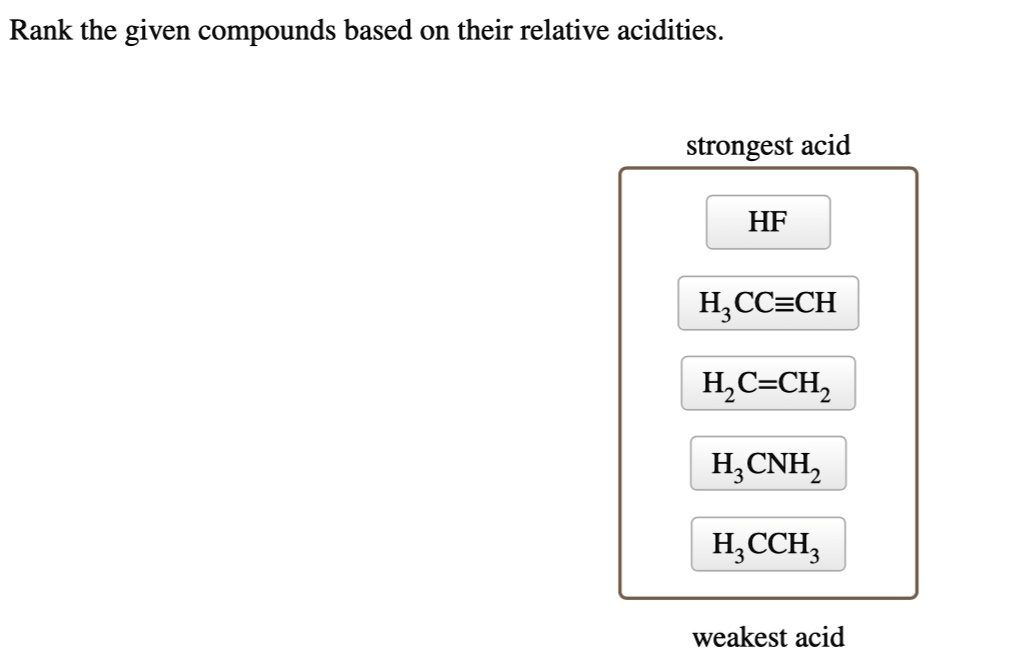
Rank The Given Compounds Based On Their Relative Acidities
Ever wondered why some kitchen staples are more "bitey" than others? Or why your car battery fluid needs a whole lot more caution than your morning coffee? It all comes down to a concept called acidity, and understanding it can be surprisingly fun and incredibly useful! Think of acidity as a chemical "oomph" – how readily a substance can donate a proton (a tiny positively charged particle). The more eager a compound is to give away that proton, the stronger its acidity. This isn't just for science geeks in labs; knowing about acidity helps us understand everything from how our bodies work to why certain cleaners are better for specific jobs.
So, how do we compare these "proton-donating champions"? That's where ranking acidity comes in. It's like a friendly competition where compounds line up to see who's the most acidic. We use a scale, often the pH scale, where lower numbers mean stronger acids. But beyond just a number, we can also think about the inherent properties of the molecules themselves. Factors like how stable the leftover part of the molecule is after it gives away its proton play a huge role.
Imagine a molecule saying, "Here, take this proton!" The stronger the acid, the more enthusiastically it's willing to part with it. This willingness is influenced by several things. One of the big players is electronegativity. Think of electronegativity as a molecule's "greed" for electrons. If a part of the molecule is very electronegative, it can pull electron density away, making the proton it's holding onto a little more exposed and easier to donate. It’s like a weak fencepost that’s easily knocked over.
Another crucial factor is the stability of the conjugate base. When an acid donates a proton, it leaves behind a conjugate base. If this conjugate base is really good at handling the extra negative charge (or the leftover bit), then the original acid was happy to give up its proton in the first place. This stability can come from spreading that negative charge out over a larger area (think of a party where everyone shares the punchbowl, so no one gets overloaded) or through resonance, where electrons can move around and delocalize the charge. Resonance structures are like a molecule's ability to shift its "weight" of negative charge, making it more comfortable.
Let's dive into ranking some compounds. We'll consider a few common types of molecules, and by looking at their structures and the principles we just discussed, we can put them in order from most acidic to least acidic. It’s like figuring out who’s the fastest runner on the track – you observe their build, their training, and their past performance.

Ranking the Compounds: The Challenge!
We're going to rank the following compounds based on their relative acidities:
- Water (H₂O)
- Ethanol (CH₃CH₂OH)
- Acetic Acid (CH₃COOH)
- Methanol (CH₃OH)
Let's break down why they'd line up the way they do. First, consider the electronegativity of the atoms directly attached to the hydrogen that can be donated. In acetic acid, the hydrogen is attached to an oxygen in a carboxyl group (-COOH). This oxygen is quite electronegative. In water, ethanol, and methanol, the hydrogen is also attached to oxygen, but the surrounding molecular environment plays a role.
Now, let's think about conjugate base stability. When acetic acid loses its proton, it forms the acetate ion (CH₃COO⁻). This ion is quite stable because the negative charge can be delocalized through resonance between the two oxygen atoms. It’s like the negative charge has two homes it can comfortably reside in, spreading out the "burden."
Resonance is a key player in stabilizing negative charges!
For water, losing a proton gives the hydroxide ion (OH⁻). For ethanol and methanol, losing a proton gives ethoxide (CH₃CH₂O⁻) and methoxide (CH₃O⁻) ions, respectively. These alkoxide ions are less stable than the acetate ion because the negative charge is localized on a single oxygen atom, and the surrounding alkyl groups (the CH₃CH₂- or CH₃-) are slightly electron-donating, which can actually destabilize a negative charge. Imagine trying to balance a heavy weight on a small point versus spreading it out on a larger surface – the latter is much more stable!
Comparing methanol and ethanol, the difference is subtle. The ethyl group in ethanol is slightly larger and more electron-donating than the methyl group in methanol. This means it might slightly destabilize the ethoxide ion compared to the methoxide ion, making methanol a very slightly stronger acid than ethanol. However, this difference is often minor.

So, where does water fit in? The hydroxide ion is more stable than the alkoxide ions because there are no electron-donating alkyl groups to destabilize the negative charge. Water is generally considered a weaker acid than simple alcohols in many contexts, but its acidity is still significant.
Putting it all together, the order of acidity from strongest to weakest would be:
- Acetic Acid (CH₃COOH): Due to excellent resonance stabilization of its conjugate base.
- Water (H₂O): The hydroxide ion is reasonably stable.
- Methanol (CH₃OH): The methoxide ion is less stable than hydroxide.
- Ethanol (CH₃CH₂OH): The ethoxide ion is slightly less stable than methoxide due to the slightly more electron-donating ethyl group.
This ranking demonstrates how subtle changes in molecular structure can lead to significant differences in acidity. It’s a fascinating peek into the world of chemical behavior and a great example of how understanding fundamental principles can help us predict and explain the properties of everyday substances!
