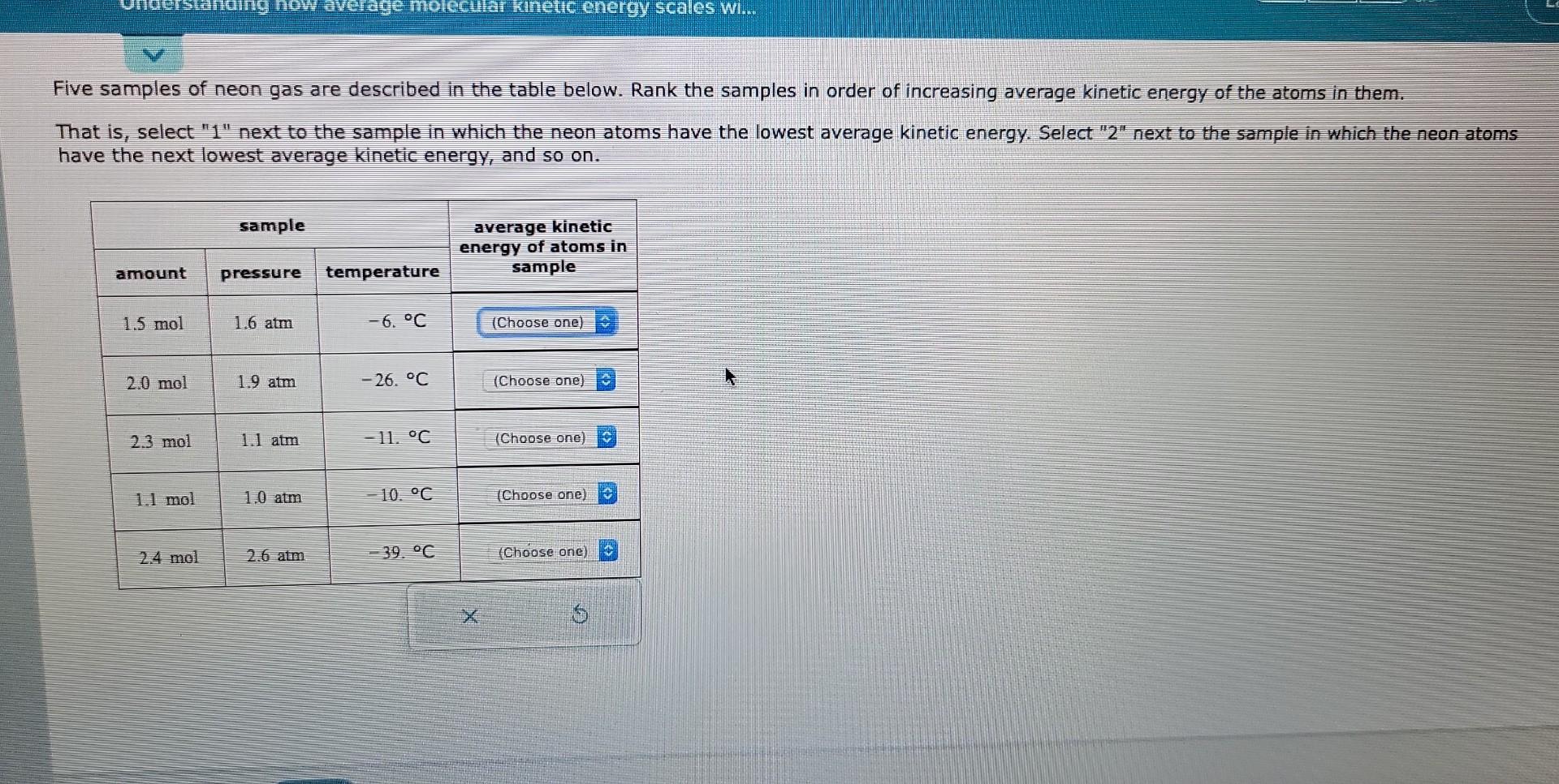
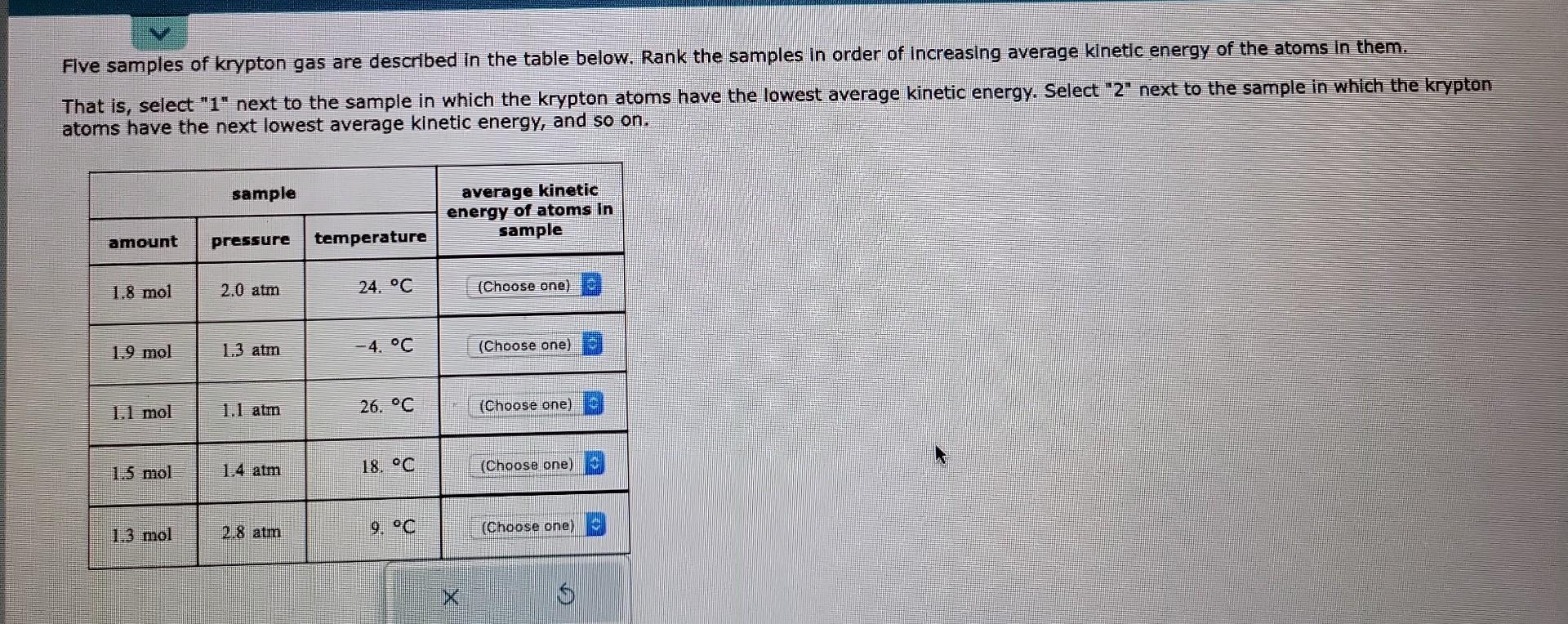
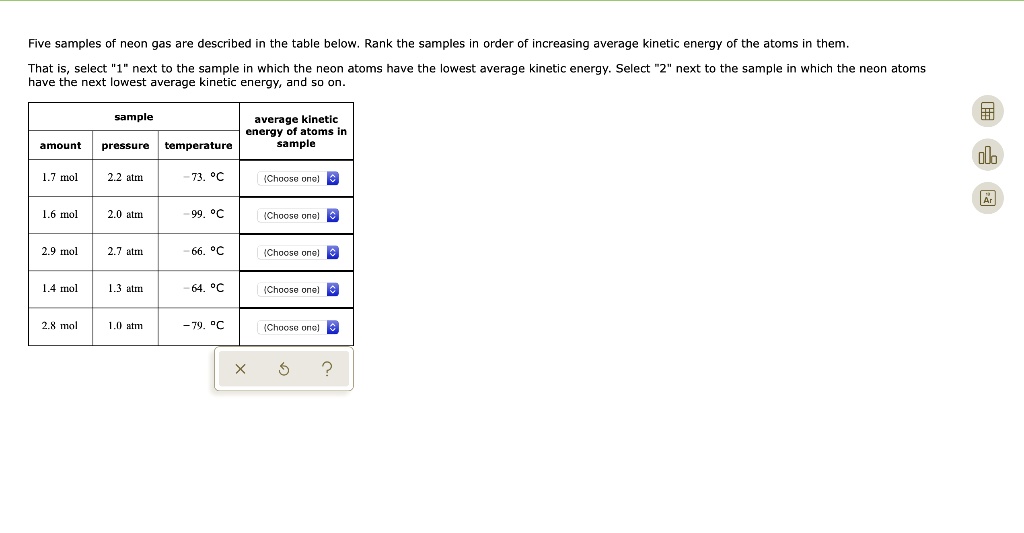
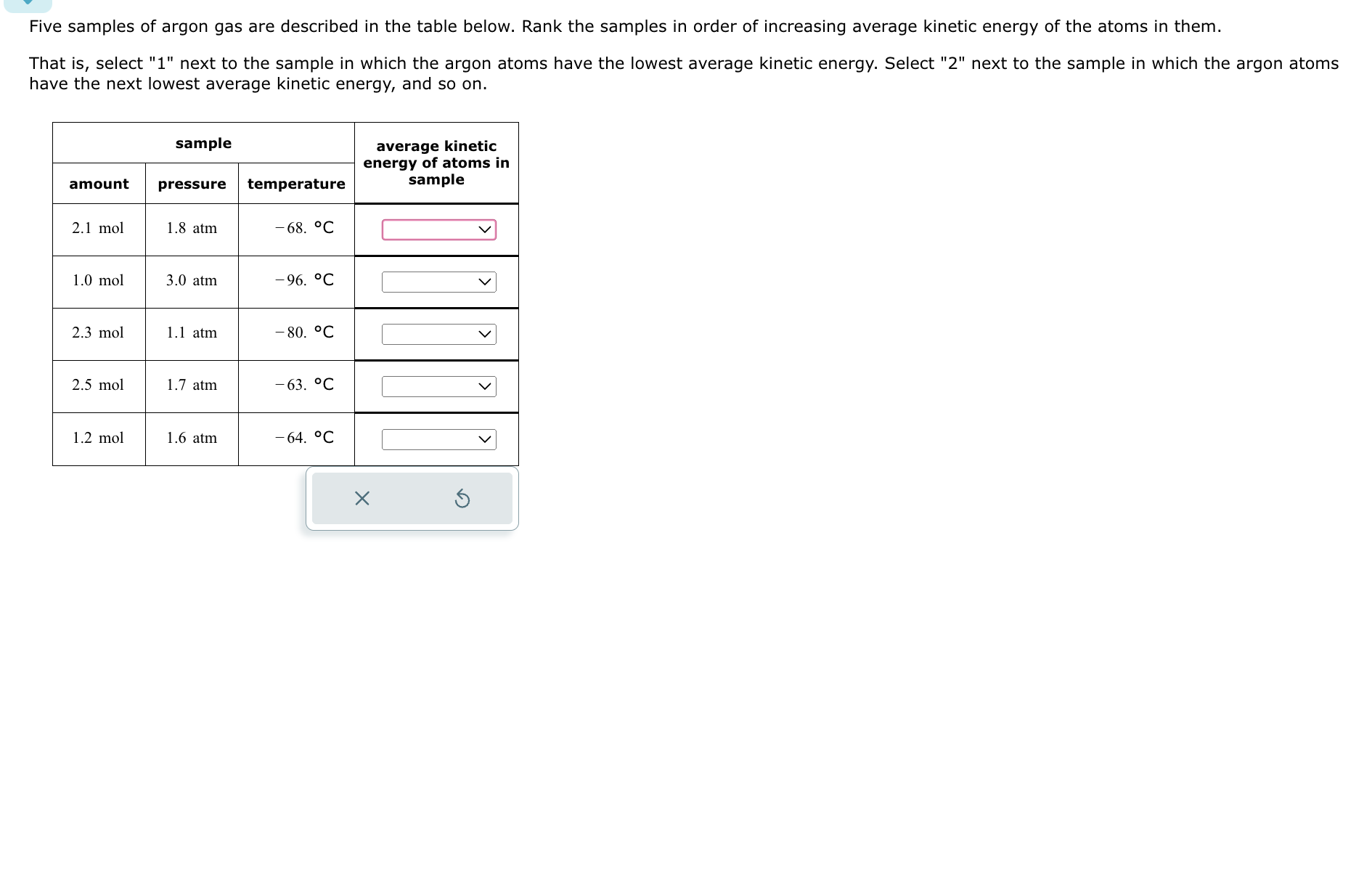
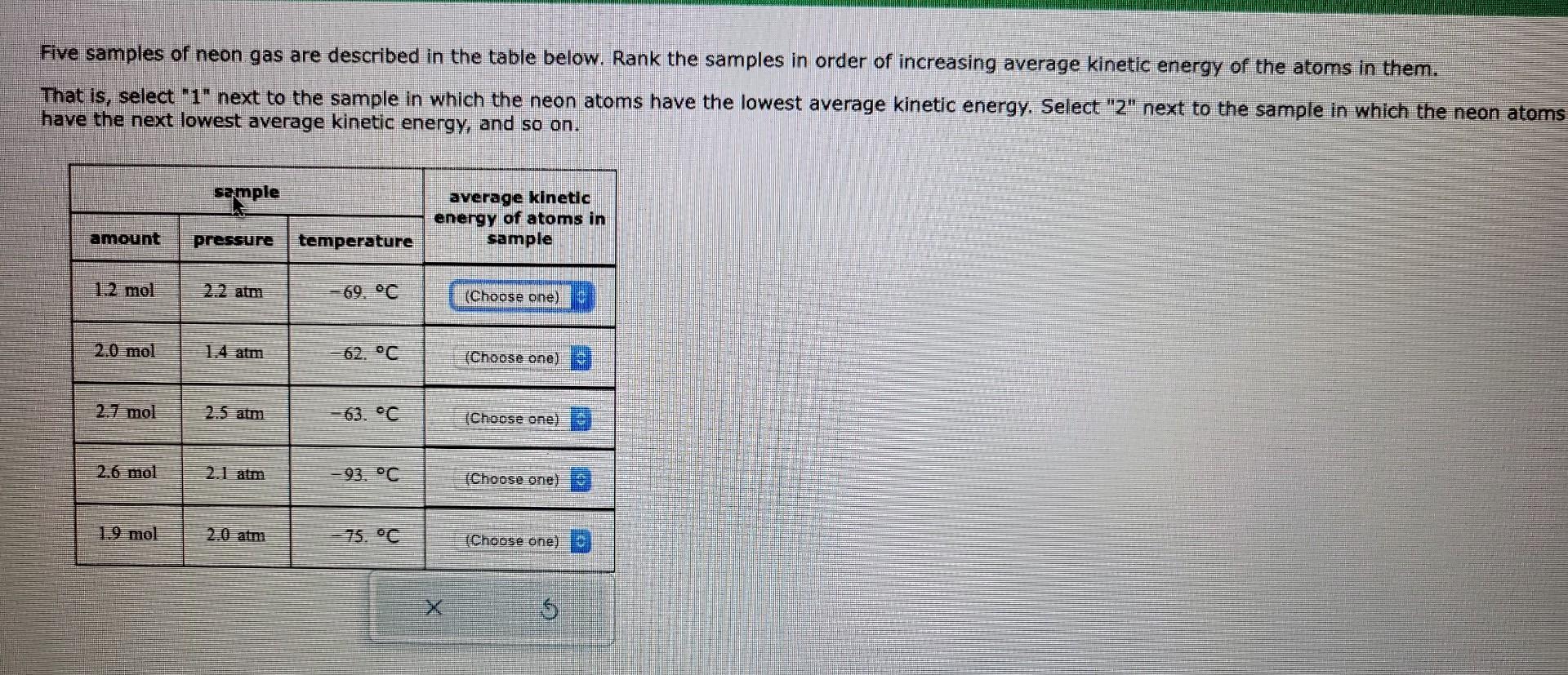
Rank The Gases In Order Of Increasing Average Kinetic Energy
Hey there! Grab your coffee, let's chat about something a little… bouncy. Ever wonder what makes gases… well, gaseous? It's all about their tiny particles zipping around, right? And how fast they zip around? That’s the magic word: kinetic energy. Think of it as their get-up-and-go energy. So, if we were to line up different gases, from the chillest dudes to the hyperactive ones, what would that even look like?
We're talking about ranking these gaseous critters by their average kinetic energy. Pretty neat, huh? It's like a race, but instead of speed, we're judging their overall wiggle factor. And the funny thing is, it’s not about the type of gas, really. Nope! It's all about the temperature. Mind. Blown. Seriously, at the same temperature, every single gas molecule, whether it's a hulking Helium or a tiny Hydrogen, is rocking the same average kinetic energy. Wild, right?
But wait, you might be thinking, "If they all have the same average kinetic energy, how can we rank them?" Ah, my friend, that’s where the devil (or the angel, depending on your perspective!) is in the details. Kinetic energy, for our little gas buddies, is defined by this formula: KE = 1/2 * mv². See that 'm'? That's mass. And the 'v'? That's velocity. So, even if the KE is the same, if the mass changes, the velocity has to adjust. It’s like a seesaw, but with atoms!
So, if we're talking about increasing average kinetic energy, and we know temperature is the kingpin, then what’s the deal? Well, it’s a trick question if you’re thinking about different types of gases at the same temperature. At the same temperature, they're all tied for first place! But let's imagine we're comparing them at different temperatures. That's when the ranking game gets real. The higher the temperature, the higher the average kinetic energy. It’s as simple as that. Think of it like this: you’re at a party. The room temperature is 70 degrees Fahrenheit. Everyone’s kind of milling about, you know? Now crank that thermostat up to 90 degrees. Suddenly, everyone’s doing the Macarena! More energy, more movement. Same party, just hotter!
But let's get playful with this. Let's pretend for a second that we're not at the same temperature. Let's imagine we have a bunch of gases chilling at different temps. Which one's gonna be the most energetic? The one that's been sitting under a heat lamp, obviously! So, to rank them in order of increasing average kinetic energy, we’d simply be ordering them by their temperature, from coldest to hottest. Easy peasy, lemon squeezy. We’re talking about things getting more jiggly, more zoomy.
Okay, so you’ve got your gases, maybe some Nitrogen (N₂), some Oxygen (O₂), some Helium (He), and maybe even some super heavy Xenon (Xe). Let's say we put them all in separate, equally warm rooms. Like, identical 25°C rooms. At this point, they're all having the same amount of fun, on average. Their average kinetic energies are neck and neck. It’s a tie! They’re all vibrating and tumbling and bumping into each other with the same gusto.
But here’s the sneaky part, the bit that makes the physics nerds (like me, sometimes!) giggle. While their average kinetic energy is the same, their individual speeds are not. Remember that mass thing? Lightweight gases, like Helium (He), have to move way faster to have the same kinetic energy as a heavier gas, like Nitrogen (N₂). It's like a little pixie trying to keep up with a sumo wrestler doing the same speed-walk. The pixie's gotta scoot!
So, let's reframe this. If we're talking about different gases, but we're fixing the temperature, then the ranking isn't about the kinetic energy itself (which is constant). It's about how that kinetic energy is distributed amongst the particles, and specifically, how the velocity is affected by mass. So, if we were to somehow "rank" them by how fast they're moving on average (which isn't the same as ranking their kinetic energy), then it would be the other way around!
Let’s get back to the prompt though, because it's all about increasing average kinetic energy. And for that, my friends, temperature is king. There’s no other way around it. So, if you’re comparing gases, and you want to order them from least energetic to most energetic, you’re just ordering them by their temperature. It’s like lining up kids after they’ve had different amounts of sugar. The one who chugged the entire soda bottle is gonna be way higher on the "wiggle energy" scale than the one who just had a tiny sip.
So, imagine you have these hypothetical scenarios. You have a beaker of Nitrogen gas at 100 Kelvin. Then you have another beaker of Oxygen gas at 200 Kelvin. And finally, a third beaker of Helium gas at 150 Kelvin. Which one has the highest average kinetic energy? The Oxygen, because it's at the highest temperature! Which has the least? The Nitrogen, at the lowest temperature.
Therefore, if we were to rank these three gases (Nitrogen, Oxygen, and Helium) in order of increasing average kinetic energy, it would simply be by their temperatures: Nitrogen (100 K) < Helium (150 K) < Oxygen (200 K). See? It’s not about the gas itself, but the environmental conditions it's chilling in. Kind of puts things in perspective, doesn't it?
Now, what if the question was phrased a little differently, and we were talking about the average speed of the molecules at the same temperature? That’s where the mass comes in with a vengeance. At the same temperature, remember, KE is constant. So, 1/2 * m₁v₁² = 1/2 * m₂v₂². If m₁ is smaller than m₂, then v₁ has to be larger than v₂. So, in that scenario, for gases like Helium and Hydrogen (the lightest champs), they'd be zipping around like tiny, invisible race cars, while Xenon (the heavyweight contender) would be… well, cruising. They’d be faster but have the same bounce. It’s a subtle, but important, distinction.
But for the question at hand – increasing average kinetic energy – it’s all about the heat. More heat, more jiggles, more wiggles, more kinetic energy. It’s the universal law of gas party stamina. Think of it like this: a frozen solid? Practically zero kinetic energy. Boiling water? Off the charts! Gases exist in that wonderfully energetic middle ground, and their average kinetic energy is a direct reflection of how much of that "party heat" they've absorbed.
So, to be super clear, if you see a question asking you to rank gases by their average kinetic energy, and the temperatures are different, just look at the temperatures. The coldest gas has the least kinetic energy, and the hottest gas has the most. It’s like a temperature-fueled race, and the finish line is pure, unadulterated molecular motion. Whoosh!
Let’s consider some common gases. We’ve got Hydrogen (H₂), Helium (He), Nitrogen (N₂), Oxygen (O₂), and Argon (Ar). Let’s say we’re comparing them at three different temperatures: 100 K, 250 K, and 400 K. If we pick one temperature, say 250 K, then all of these gases have the same average kinetic energy. Again, tie! But if we’re ranking them across these temperatures? Then it’s all about which one is getting the warmest treatment.
So, if we’re ranking these gases in order of increasing average kinetic energy, and we're implicitly assuming they are at different, increasing temperatures, then the order would be whatever gas is at the lowest temperature first, then the next lowest, and so on. It’s not about the specific gas molecule’s weight. It’s about the overall thermal energy of the sample. Like a whole bunch of tiny dancers, their collective energy is determined by how much of a "beat" they're getting from the music – and the music’s volume is the temperature.
Think about it this way: If you have a tiny little ant and a big ol' elephant, and you give them both the exact same amount of energy to run a race, they’ll cover different distances, right? The ant’s gonna be a blur, the elephant’s gonna be… well, an elephant. But the energy they expended is the same. That’s the KE for you. However, if you just say "who's got more potential to zoom?", you'd probably say the ant, assuming they're both given the same opportunity to move. But here, we're talking about actual kinetic energy. And that, my friends, is tied to the thermal bath they're swimming in.
So, the simplest, most direct answer to ranking gases by increasing average kinetic energy is to simply rank them by their increasing temperatures. It’s the most fundamental principle at play. Everything else, like the mass, becomes a factor when you start talking about individual molecular speeds at a constant temperature. But for the average kinetic energy? Temperature reigns supreme. It's the ultimate determinant of how much internal "zoom" the gas has. So next time you're feeling a bit sluggish, just remember, your kinetic energy is directly related to your internal "temperature"! Maybe crank up the thermostat on your own life. 😉
Let's solidify this. If I give you a list like: Gas A at 50 K Gas B at 100 K Gas C at 25 K Then, the order of increasing average kinetic energy is: Gas C < Gas A < Gas B. Because 25 K is the coldest, 50 K is in the middle, and 100 K is the hottest. It’s that straightforward! No complex calculations needed, just a good old-fashioned look at the thermometer readings. It’s the universe’s way of saying, “More heat, more hustle!”
And you know what’s really cool? This applies to all gases. Whether it’s a noble gas like Neon, a diatomic molecule like Chlorine, or even something more complex. The average kinetic energy is a universal measure of how "excited" the particles are, and that excitement level is directly dictated by the temperature of their surroundings. So, it’s not about the type of gas, it’s about the degree of thermal celebration they're experiencing. Pretty awesome, when you think about it!
