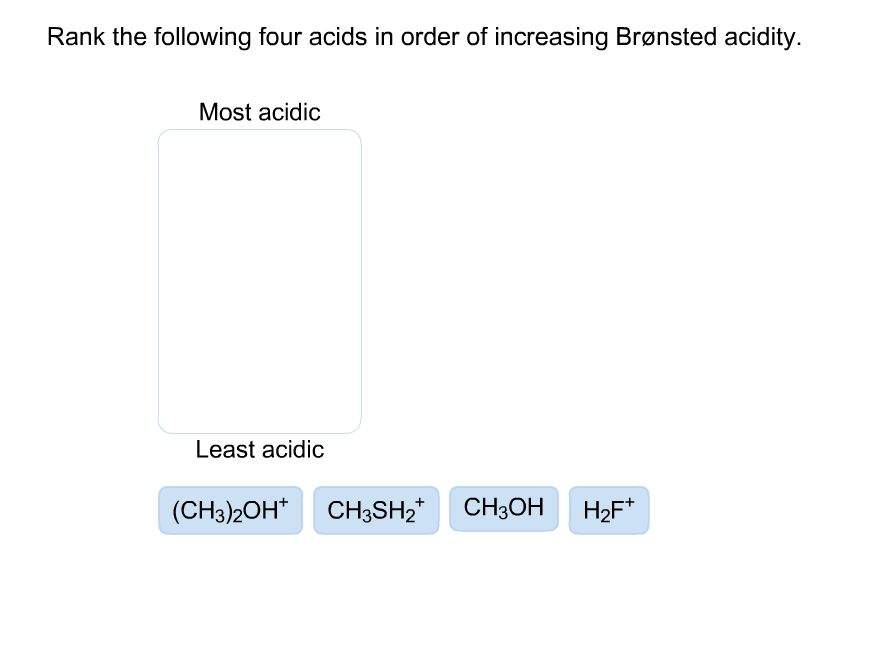
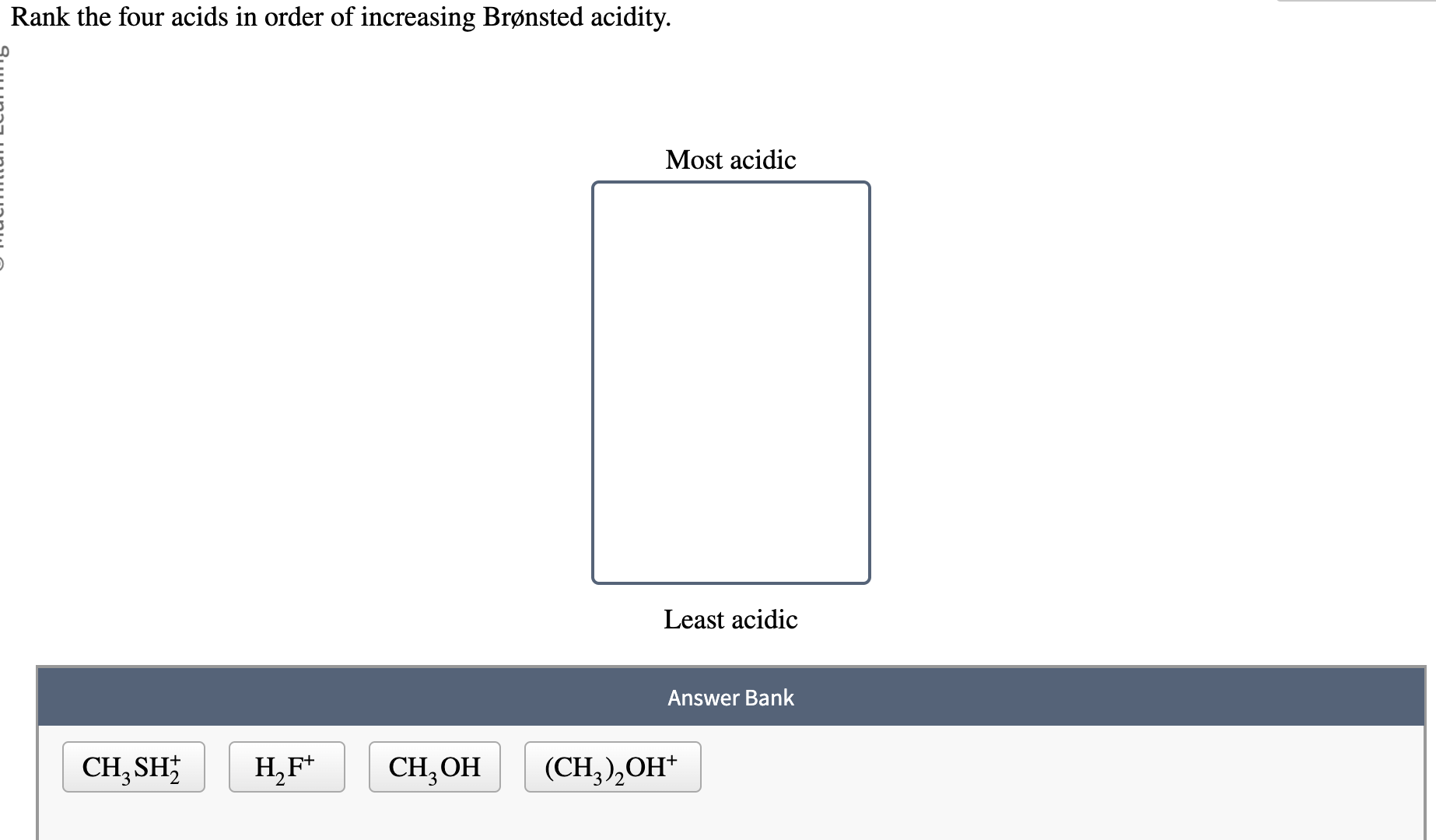
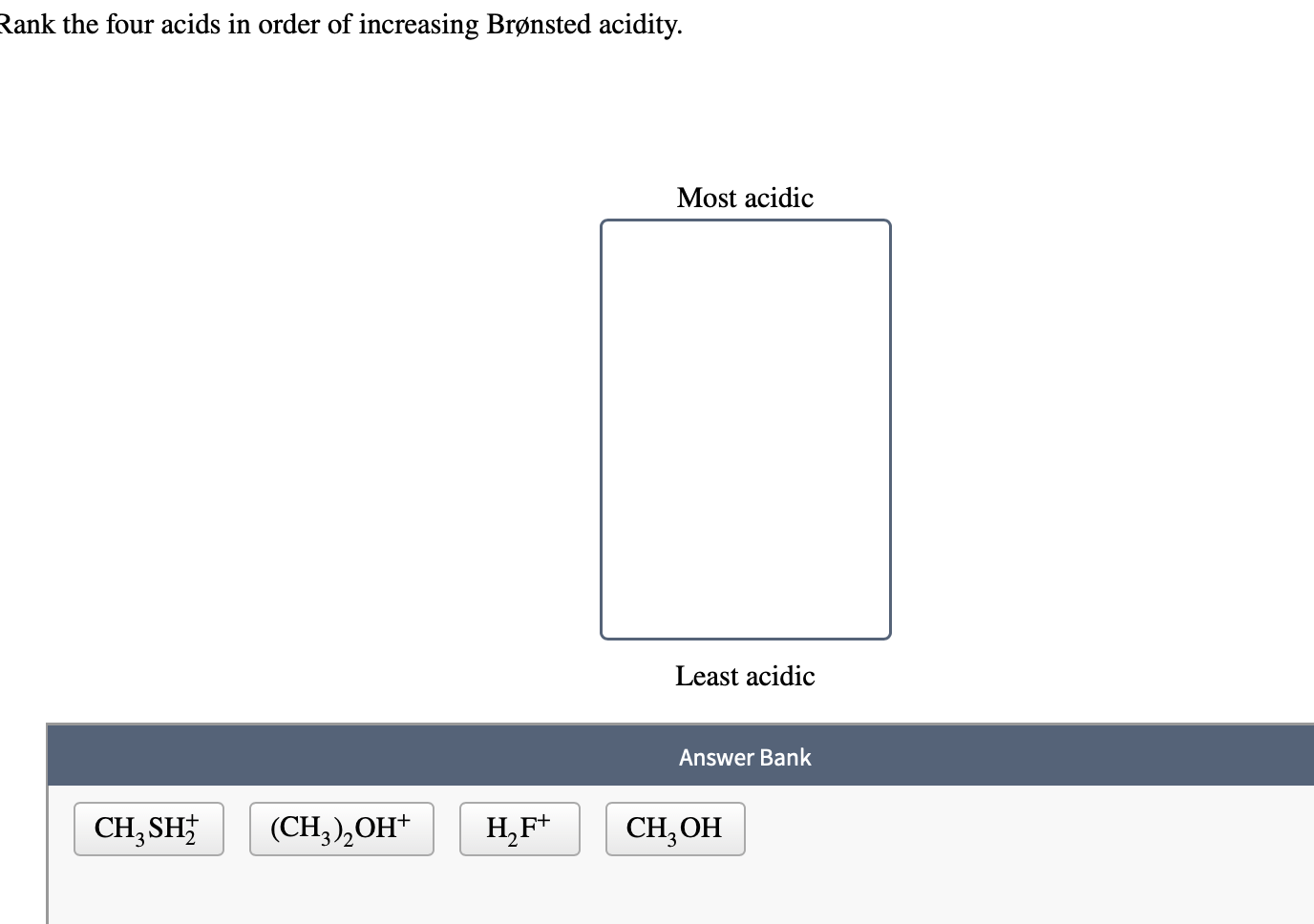
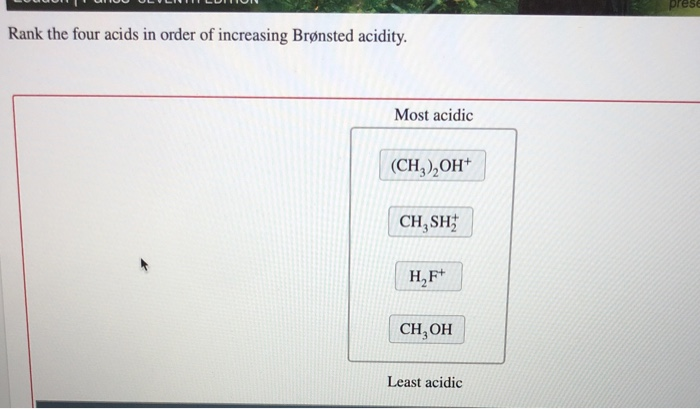
Rank The Four Acids In Order Of Increasing Brønsted Acidity.

So, picture this: I'm tinkering around in my kitchen, you know, the usual Saturday morning chaos, trying to whip up some fancy vinaigrette. I’ve got this collection of vinegars – balsamic, red wine, apple cider, even a fancy champagne one. I’m pouring, sniffing, tasting (don’t judge, it’s part of the process!), and I start thinking. They all taste… well, acidic, right? But some are definitely sharper, more biting than others. That lightbulb moment? It hit me like a splash of lemon juice right in the eye – these aren't all equally sour! And that, my friends, is how we tiptoe into the wonderfully weird world of Brønsted acidity.
Yeah, I know, "Brønsted acidity" sounds like something you’d find etched on an ancient scroll, not in your salad dressing. But stick with me! It’s actually a pretty cool concept that explains why some things are more eager to donate a proton (that's the 'H+' part of an acid, for those of you who skipped chemistry or just slept through it) than others.
So, let’s imagine we’ve got our four acids lined up. For the sake of this little adventure, let’s call them Acid A, Acid B, Acid C, and Acid D. We're going to try and figure out which one is the biggest proton-pusher, the one that’s practically begging to get rid of its H+. Think of it like a popularity contest for hydrogen ions. Who's the most desirable? Who's the least?
Now, why are we even bothering to rank them? Well, it’s not just about making better vinaigrettes (though that’s a noble pursuit!). Understanding acidity is super important in chemistry. It helps us predict how reactions will behave, design new materials, and even understand how our bodies work. Seriously, it’s everywhere!
Before we dive into the nitty-gritty of our mystery acids, let's have a quick refresher on what makes an acid an acid, Brønsted-style. A Brønsted acid is defined as a substance that can donate a proton (H+). Simple enough, right? It’s like a generous friend who’s always willing to lend you a bit of their energy (in this case, a proton).
The flip side of this coin is a Brønsted base, which is a substance that can accept a proton. So, our generous acid friend needs someone to receive that proton. It’s a give-and-take relationship, like a chemical tango!
Now, the strength of an acid isn't just about how sour it tastes. It's about how easily it can donate that proton. Some acids are super eager, practically throwing their protons at anything that’ll take them. These are our strong acids. Others are a bit more hesitant, they hold onto their protons pretty tightly. These are our weak acids.
And how do we quantify this eagerness? We look at something called the acid dissociation constant (Ka). Don't let the scary name fool you! A higher Ka value means the acid is more likely to dissociate (break apart) and donate its proton. Think of it as a score: the higher the score, the more "acidic" the acid is in terms of its ability to donate a proton.
Alternatively, you might see something called pKa. This is just the negative logarithm of Ka. So, the lower the pKa, the stronger the acid. It's like flipping the score around, so lower numbers are better. It can be a bit confusing, I know! It’s like they deliberately make these things complicated just to keep us on our toes. But once you get the hang of it, it makes perfect sense.
So, for our ranking exercise, we’re essentially looking for the acid with the lowest pKa (or the highest Ka) to be our strongest Brønsted acid.
Now, what factors influence this proton-donating prowess? It’s not just a random toss-up. Several things play a role:
First up, we have electronegativity. This is a fancy word for how much an atom likes to pull electrons towards itself. Atoms that are more electronegative will pull electrons away from the atom they're bonded to, weakening that bond and making it easier for a proton to escape.
Then there’s bond strength. A weaker bond between the hydrogen and the rest of the molecule means the hydrogen is more likely to be released as a proton. It’s like a frayed rope – easier to snap!
We also need to consider the stability of the conjugate base. When an acid donates a proton, what’s left behind is called its conjugate base. If this conjugate base is really stable (meaning it doesn't mind being left with that extra electron after the proton leaves), then the original acid was probably pretty eager to donate its proton in the first place. It’s like leaving a party: if you know you’re going to a much cooler party afterwards, you’re going to leave this one in a heartbeat!
And finally, there are resonance effects. This is where electron density can be spread out over multiple atoms in the conjugate base, making it more stable. Think of it like sharing the load. If one person has to carry all the groceries, they're going to be unhappy. But if you have a whole team to help, it's much easier!
Okay, enough theory! Let's get to our four mystery acids. For the sake of our example, and to make things a bit more concrete, let's pick some common ones that showcase these principles. Let's consider these four:
The Contenders:
1. Hydrochloric Acid (HCl)
2. Acetic Acid (CH3COOH)
3. Water (H2O)
4. Ammonia (NH3)
Now, you might be thinking, "Wait, is water an acid? And ammonia? Isn't ammonia a base?" Ah, my curious friend, you're touching on a very important point! Many substances can act as both acids and bases depending on the situation. They're like those chameleon-like characters in movies. Water is famously amphoteric, meaning it can be an acid or a base. Ammonia, while typically known as a base, can technically act as a very, very weak acid.
Let's look at their typical properties and their approximate pKa values (remember, lower pKa means stronger acid):
- Hydrochloric Acid (HCl): This is a classic strong acid. Its pKa is around -7. Yes, that’s a negative number! That’s how strong it is.
- Acetic Acid (CH3COOH): This is the acid in vinegar. It’s a typical weak acid. Its pKa is around 4.76.
- Water (H2O): Water's self-ionization constant (Kw) is 1.0 x 10-14 at 25°C, and its pKa is approximately 15.7. So, it's a pretty weak acid.
- Ammonia (NH3): As an acid, ammonia is extremely weak. Its pKa for the reaction NH3 + H2O ⇌ NH4+ + OH- is more commonly discussed in terms of its basicity. However, if we consider it acting as an acid (donating a proton from one of its H atoms), the pKa is estimated to be around 38. That’s a whoppingly high number!
So, with these pKa values in hand, let's put them in order of increasing Brønsted acidity. This means we're going from the weakest proton-donor to the strongest.
The Big Reveal: Ranking Our Acids
Drumroll, please… here are our four acids, ranked from weakest Brønsted acid to strongest:
1. Ammonia (NH3) - The Reluctant Proton Donor
With a pKa around 38, ammonia is by far the weakest Brønsted acid on our list. It holds onto its protons with the tenacity of a cat clinging to a curtain. For it to donate a proton, you'd need an incredibly strong base to coax it away. In most everyday scenarios, we consider ammonia a base, not an acid, and for good reason!
Why is it so weak? Well, look at the conjugate base that would form if it did donate a proton: the amide ion (NH2-). This ion is incredibly unstable and highly reactive because the negative charge is localized on the nitrogen atom, which isn't particularly good at accommodating it. It’s like asking someone to carry a ridiculously heavy weight all by themselves – they’re not going to be eager to pick it up!
2. Water (H2O) - The Middle-of-the-Road Molecule
Next up is water, with a pKa of about 15.7. Water is a much better proton donor than ammonia, but still significantly weaker than typical acids. This is why pure water has a very low conductivity – not many H+ ions are floating around.
When water acts as an acid, it forms the hydroxide ion (OH-). This ion is more stable than the amide ion because oxygen is more electronegative than nitrogen, meaning it can better handle that negative charge. However, it's still not an incredibly stable species on its own. Water is happy to act as an acid when it encounters a very strong base, but it prefers to act as a base when it encounters an acid.
It’s this dual nature that makes water so fascinating. It’s the universal solvent, and its ability to both donate and accept protons is key to so many chemical and biological processes. Think about it: even the slight acidity of water plays a role in how life functions!
3. Acetic Acid (CH3COOH) - The Familiar Vinegar Zing
Here we have acetic acid, with a pKa of around 4.76. This is where things start to feel more like what we traditionally think of as an "acid." It's strong enough to give vinegar its characteristic sour taste and to react with bases. It's a typical weak acid.
When acetic acid donates a proton, it forms the acetate ion (CH3COO-). This conjugate base is stabilized by resonance. You see, the negative charge isn't just stuck on one oxygen atom; it can be shared between the two oxygen atoms in the carboxylate group. This delocalization of charge makes the acetate ion much more stable than the hydroxide ion, meaning acetic acid is more willing to donate its proton.
This resonance stabilization is a big deal in organic chemistry. It’s what gives many carboxylic acids their acidic properties. So, the next time you taste vinegar, you can impress your friends (or just yourself) by thinking about the resonance-stabilized acetate ion!
4. Hydrochloric Acid (HCl) - The Reigning Champion of Proton Donation
And finally, the undisputed king of our little acid club: Hydrochloric Acid (HCl), with a pKa of about -7. This is a strong acid. When you put HCl in water, it practically dissociates 100% into H+ (which immediately forms hydronium ions, H3O+, in water) and Cl- ions.
Why is HCl so strong? Several factors contribute. First, chlorine is a highly electronegative atom. It pulls electron density away from the hydrogen, weakening the H-Cl bond. Second, the chloride ion (Cl-) is a very stable conjugate base. It's a large ion, and the negative charge is spread out over its electron cloud, making it very happy not to be bonded to the proton anymore. It doesn't have the same need to snatch a proton back as, say, a hydroxide ion might.
Think of it this way: if the conjugate base is really stable and doesn't want the proton back, the original acid is going to be very eager to give it away. HCl’s conjugate base, Cl-, is like a person who just won the lottery and is off to live on a tropical island – it has no intention of coming back!
Putting It All Together: The Order of Increasing Brønsted Acidity
So, to recap, from weakest Brønsted acid to strongest, our order is:
Ammonia (NH3) < Water (H2O) < Acetic Acid (CH3COOH) < Hydrochloric Acid (HCl)
This ordering is based on their relative abilities to donate a proton. The factors we discussed – electronegativity, bond strength, and especially conjugate base stability – are what drive these differences.
It’s a pretty neat illustration of how molecular structure and electron distribution dictate chemical behavior. It’s not just about memorizing facts; it’s about understanding the underlying principles that make these molecules behave the way they do.
And there you have it! A not-so-dry dive into Brønsted acidity, all thanks to a vinaigrette experiment and a healthy dose of curiosity. Next time you’re dealing with acids, whether it’s in the lab or in your kitchen, you’ll have a better appreciation for their relative strengths and why they behave the way they do. Chemistry is everywhere, and it’s often more interesting than you might think!
