Rank The Following Three Transitions In The Hydrogen Atom

Hey there, science fans! Ever stared up at the night sky and wondered about all that sparkly stuff? Well, today we're diving into something super cool, but way smaller than stars. We're talking about the teeny-tiny hydrogen atom. It’s basically the universe’s building block. And guess what? This little guy can do some pretty neat tricks. We're gonna rank its energy level jumps. Yep, it's like a tiny atomic olympics!
So, why is hydrogen so special? It's the simplest atom. Just one proton and one electron. That's it! No complicated inner workings. This makes it the perfect playground for quantum mechanics. It’s the OG of atoms. The one that started it all. And its electron? It doesn't just chill in one spot. It can be in different energy levels. Think of it like a little elevator. It can go up or down.
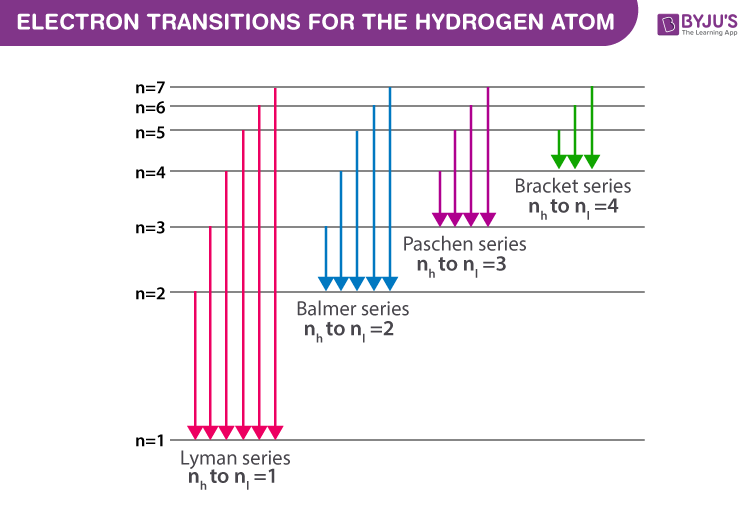
These elevator rides are called transitions. And they’re a big deal. They’re how atoms interact with light. When an electron jumps up, it absorbs energy. Usually in the form of a photon, which is basically a tiny packet of light. When it falls back down, it emits energy. Also as a photon. So, the next time you see a rainbow or a neon sign, you’re witnessing these atomic jumps in action! Pretty wild, right?
Today, we’ve got three specific transitions in the spotlight. We’re gonna rank them. From least to most… well, let’s just say exciting. It’s not about which one is "better." It's about the energy involved. The drama! The spectacle! Get ready for some atomic showmanship.
The Contenders: Our Atomic Elevator Guests
First up, we have the transition from the ground state to the first excited state. This is the n=1 to n=2 jump. The most basic of the basic. It’s like the warm-up act. The opening band. It’s important, but maybe not the headliner.
Next, we’ve got a leap to the second excited state. So, that’s from the ground state (n=1) all the way up to n=3. This is a bigger jump. Requires more oomph. More energy. It’s like going from the first floor to the third floor. A decent climb.
And finally, the grand finale! The transition from the ground state (n=1) to the third excited state. That’s n=1 to n=4. This is a serious elevator ride. A big climb. A real workout for our electron.
Ranking The Transitions: The Atomic Showdown!
Alright, let’s get down to business. We’re ranking these transitions based on the energy required for the electron to make the jump. The more energy, the higher the rank in our fun little competition.
Third Place: The Gentle Ascent (n=1 to n=2)
This is our bronze medalist. The transition from the ground state to the first excited state. It requires the least amount of energy of our three contenders. It’s like a gentle nudge. A polite request for an energy boost. This jump is responsible for absorbing and emitting ultraviolet light. Yep, the stuff that can give you a sunburn. So, even the smallest jump has a tangible effect!

This transition is super important in astronomy. It’s one of the primary ways we detect hydrogen in space. When a star is emitting UV light, and it hits a hydrogen atom, that electron gets excited. Boom! We see it. It’s like a cosmic Morse code. A whisper across the vastness of space.
It's the fundamental step. The first hurdle. Without this, the electron would just be chilling in its cozy ground state. Forever. So, it’s definitely not insignificant. It’s just… the beginning of the journey.
Second Place: The Energetic Climb (n=1 to n=3)
Taking home the silver medal is the transition from the ground state to the second excited state. This requires a significant amount more energy than the n=1 to n=2 jump. It’s like going from climbing a small hill to a moderately sized one. Still doable, but you’ll feel it!
This transition involves the absorption and emission of visible light. Specifically, this jump is responsible for one of the famous lines in the hydrogen spectrum: the Balmer alpha line. This is the bright red light you see in some specialized lamps and, famously, in nebulae. That’s right, some of the most beautiful cosmic colors are thanks to this specific energy jump!

Think about it: a simple electron jumping up two energy levels and painting the universe with vibrant red. It’s like a tiny artist with a limited palette, but oh-so-impactful. The energy here is higher, the photon emitted is more energetic. It’s a step up in the atomic world!
First Place: The Grand Leap (n=1 to n=4)
And the gold medal goes to… the transition from the ground state to the third excited state! This is our heavyweight champion. It requires the most energy of the three. This electron is making a serious leap. It's like going from the bottom of a skyscraper to the top floor. Wowza!
This jump involves even higher energy photons. When it falls back down, it can emit in different ways, but the initial excitation is the most demanding. It’s also responsible for visible light, but with even more energy than the previous jump. It’s a more energetic portion of the visible spectrum.
Imagine the electron being really motivated. It needs a serious jolt to get up there. This transition is a testament to the power of energy transfer in the atomic realm. It’s the most dramatic of our three contenders, requiring the biggest "push" from a photon to get our electron into this higher orbit.
Why This Stuff is Actually Fun
So, there you have it! Our fun little ranking of hydrogen atom transitions. It’s not just abstract science. These jumps explain why things glow, why stars shine, and how we can even study the universe from afar. It’s the microscopic dance that creates the macroscopic wonders we see every day.
And the best part? Hydrogen is everywhere. These transitions are happening all the time, all around us. It’s a constant, silent symphony of energy. A reminder that even the simplest things can be incredibly complex and beautiful.
So next time you look at the sky, or even just a flickering light bulb, remember the humble hydrogen atom and its energetic elevator rides. It’s a tiny world with huge implications. And that, my friends, is pretty darn cool. Science can be quirky, it can be dramatic, and it can explain the magic of the universe. What’s not to love?
