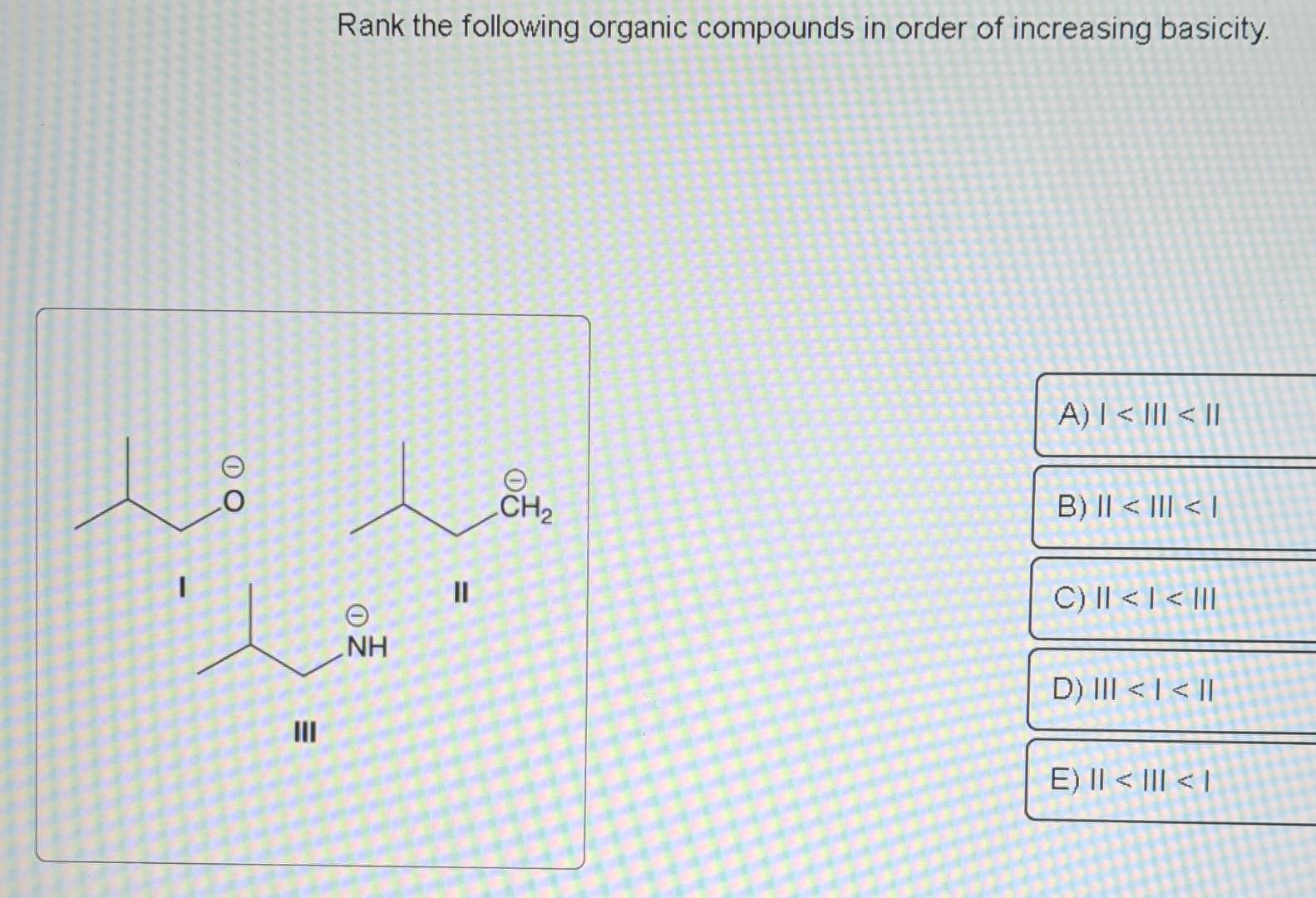
Rank The Following Organic Compounds In Order Of Increasing Basicity.
Ah, the thrill of a good ranking! Whether it's sorting your favorite pizza toppings, deciding who gets the last slice of cake, or, in our case, ordering a lineup of organic compounds, there's something undeniably satisfying about putting things in their proper place. It's a little like solving a puzzle, and for those of us who dabble in the fascinating world of chemistry, this particular puzzle – ranking basicity – is a delightful intellectual exercise. It taps into that innate human desire for order and understanding, offering a sneak peek into the hidden behaviors of molecules.
But why bother with basicity, you might ask? Isn't that just for lab coats and beakers? Far from it! Understanding basicity is surprisingly relevant to our everyday lives. It's the principle behind why your antacid neutralizes stomach acid, why certain cleaning products work, and even how your body's own systems regulate pH. Think of it as the secret sauce that allows for countless chemical reactions to occur, from the mundane to the life-saving. When a substance is basic, it means it has a tendency to accept protons (tiny positively charged particles), effectively counteracting acidity. This fundamental interaction is at play everywhere!
Common examples abound. Your humble baking soda (sodium bicarbonate) is a base, used in everything from baking to cleaning. Ammonia, a familiar scent in many household cleaners, is also a base. Even the proteins in your body rely on the basic properties of certain amino acids to perform their vital functions. When we talk about ranking basicity, we're essentially predicting which molecule will be the strongest proton acceptor. It's a way to quantitatively compare their chemical personalities.
So, how do we tackle the task of ranking the following organic compounds in order of increasing basicity? It's a journey that involves considering factors like electron density and stability of the conjugate acid. Generally, more electron-rich atoms are better at attracting protons, making the molecule more basic. Conversely, if the species formed after accepting a proton is very stable, the original molecule is also more likely to act as a base. It’s a bit like choosing who to borrow money from – you'd rather borrow from someone who can easily afford it and won't be too bothered by it!
To enjoy this ranking process even more effectively, try visualizing the structures. Draw them out, identify the potential sites for protonation, and think about what happens to the electrons. Online resources and chemistry textbooks are your best friends here, offering diagrams and explanations. Don't be afraid to consult them! The more you practice, the more intuitive it becomes. Think of it as learning a new language, and with each ranking, you're becoming more fluent in the language of chemistry. So, grab your metaphorical ranking hat, and let's dive into the exciting world of molecular basicty!
