Rank The Following Orbitals In Terms Of Their Energies.
Ever found yourself staring at a cloudy sky, wondering about the grand cosmic dance happening far above? Or perhaps you've been captivated by those mesmerizing images of swirling galaxies and nebulae? Well, for many of us, there's a certain thrill in understanding the fundamental building blocks of the universe, and that often leads us to the fascinating world of atomic orbitals. It's a bit like solving a cosmic puzzle, and one of the most satisfying pieces to fit is understanding the energy levels of these tiny electron homes. You might not think it, but this seemingly academic pursuit has some surprisingly practical applications and can even be a fun intellectual exercise.
So, why do we bother ranking atomic orbitals by their energies? It's not just for trivia nights (though it makes for great conversation!). Understanding orbital energies is absolutely crucial in fields like chemistry and physics. It helps us predict how atoms will interact with each other to form molecules, why certain chemical reactions happen, and why materials have the properties they do. Think about it: the colors of fireworks? That's all thanks to electrons jumping between different energy levels. The way your smartphone screen works? It relies on the electronic properties of materials, which are dictated by orbital energies. Even the development of new medicines and advanced materials hinges on this fundamental knowledge.
You might be wondering how this relates to your everyday life. Beyond the indirect influence on technology and materials, understanding orbital energies can foster a deeper appreciation for the natural world. When you look at a leaf, you can imagine the complex interplay of electrons within its atoms, all governed by these energy rules. It's a glimpse into the hidden, elegant order of the universe. For those who enjoy problem-solving and logic, ranking orbitals can be a very rewarding mental workout. It's like a brain teaser, requiring you to recall and apply a set of rules to arrive at the correct order.
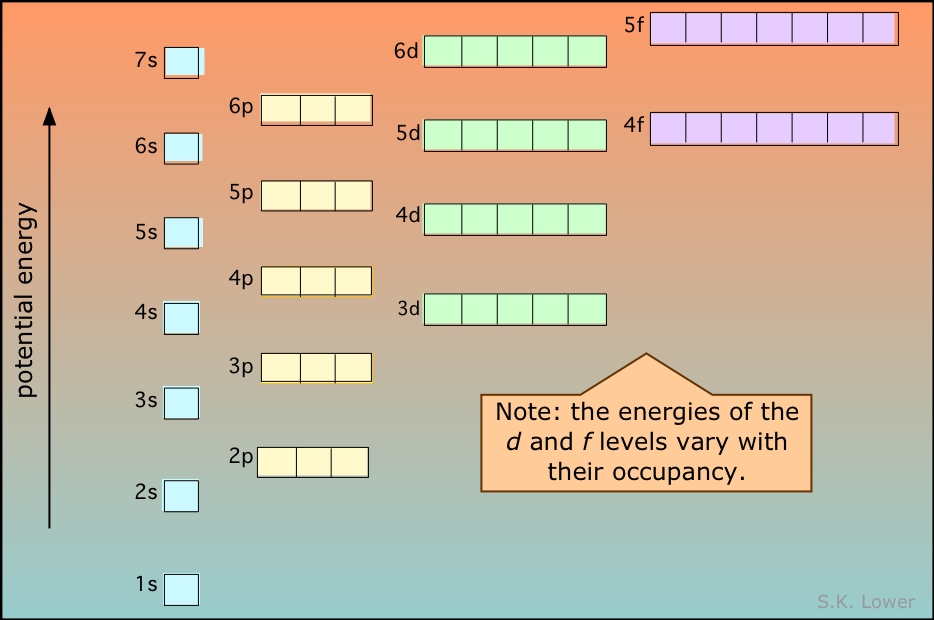
Now, let's get to the fun part: ranking these orbitals. For a general audience, we can simplify it a bit. Imagine electrons living in different "neighborhoods" around the atom's nucleus, each with a different energy cost to occupy. Generally, the closer an electron is to the nucleus, the lower its energy. We have different shapes of these neighborhoods, called s, p, d, and f orbitals. The s orbitals are spherical and are generally the lowest in energy. Then come the p orbitals, which have dumbbell shapes and are slightly higher in energy. Following those are the d orbitals, with more complex shapes and higher energies, and finally, the f orbitals, which are the most complex and highest in energy for a given shell.

However, things get a bit more interesting when we consider different electron shells (the main energy levels). The order isn't always a simple 1s, 2s, 2p, 3s, 3p, 3d... because sometimes a higher shell's orbital can have a lower energy than a lower shell's orbital! For instance, the 4s orbital is actually lower in energy than the 3d orbitals. This is a key point in understanding electron configurations. The general rule of thumb to remember this complex dance is often referred to as the Aufbau principle, which basically says electrons fill orbitals starting from the lowest energy level available. Think of it like filling up seats on a bus – you fill the front seats first!
To enjoy this activity more effectively, try visualizing it. Many online resources offer interactive diagrams that show the shapes and energy levels. Don't be afraid to sketch it out yourself! Drawing the shapes and noting the energy order can really help solidify the concept. And remember, it’s okay to be a bit curious and ask "why?" The more you explore, the more you'll appreciate the elegant simplicity and profound implications of these tiny, energetic electron homes.
