Rank The Following Five Elements By Ionization Energy.
Okay, so picture this: we're about to dive into a little game, a fun little science challenge that might sound a bit technical at first, but trust me, it's way more engaging than you think! We're going to rank five elements based on something called ionization energy. Sounds fancy, right? But it's actually a super cool concept that tells us something neat about these fundamental building blocks of everything around us.
Think of ionization energy like a game of "pulling teeth" for atoms. We're talking about how much oomph it takes to yank an electron away from an atom. Electrons are those tiny, zippy things that orbit the center of an atom. They're kind of like the little kids in the atom's family, and sometimes, you need a bit of effort to get one to leave home and go off on its own.
Now, why is this a game worth playing? Because it reveals a hidden order in the universe! It’s like a secret handshake that atoms give each other, showing us how they interact and behave. And the elements we're going to rank? They're like a quirky cast of characters, each with their own personality when it comes to letting go of an electron.
We've got:
- Lithium (Li)
- Beryllium (Be)
- Boron (B)
- Carbon (C)
- Nitrogen (N)
These are all pretty chill elements, sitting next to each other on the periodic table. You know, that giant chart that looks like a super-organized spreadsheet for all the elements? These guys are like neighbors in a very neat row. And because they're neighbors, their ionization energies have some really interesting patterns.
So, what makes this particular ranking so special? It’s not just about numbers; it's about understanding the why behind the numbers. It’s about noticing the subtle shifts in an atom’s structure that make it harder or easier to pull away an electron. It’s like figuring out which of your friends is the most stubborn when you ask them to lend you something, and which one is the most willing to share. Every element has its own unique level of "clinginess" to its electrons.

Let’s get our detective hats on. We're going to arrange these five elements from the one that’s easiest to steal an electron from (lowest ionization energy) to the one that’s the hardest (highest ionization energy).
Imagine you're at a carnival, and each element is a game. Some games are super easy to win a prize from. Others require a lot more skill and effort. We're ranking these elements by how "easy" or "hard" they are to win an electron from!
This is where the fun really kicks in. As we move across the periodic table, things start to change. The number of protons in the center of the atom increases, and that has a big impact. Protons are like the super-strong magnets in the atom's nucleus, holding onto those electrons. More protons mean a stronger pull!

But it's not just about the protons. The electrons themselves are also doing their own dance. They’re arranged in different "shells" or "energy levels," and their positioning matters. Some electrons are closer to the nucleus, feeling its strong pull, while others are further away, a bit more shielded and therefore a bit easier to snatch.
So, when we rank Lithium, Beryllium, Boron, Carbon, and Nitrogen, we're essentially observing how these factors – the number of protons and the arrangement of electrons – play out in a mini-drama. It’s a beautiful illustration of the rules that govern the atomic world.
Why is this entertaining? Because it’s a puzzle! It’s a bit like a treasure hunt where the treasure is understanding the fundamental nature of matter. You start with a question – "How do these elements compare?" – and through a bit of scientific reasoning, you uncover a clear, logical answer. There's a satisfying click when you figure it out, like solving a really good riddle.

What makes it special is that it’s not just some abstract concept confined to a textbook. This understanding of ionization energy affects everything. It explains why certain elements form specific types of bonds, how they react with other elements, and ultimately, why the world is the way it is. From the metals in your phone to the gases in the air, ionization energy is playing a role!
So, here it is, the grand ranking! Prepare for a little surprise, because sometimes nature likes to throw a curveball. Let's see who makes it to the top (or bottom, depending on how you look at it!).
We're ranking them by increasing ionization energy, meaning we start with the element that needs the least energy to lose an electron and end with the one that needs the most.
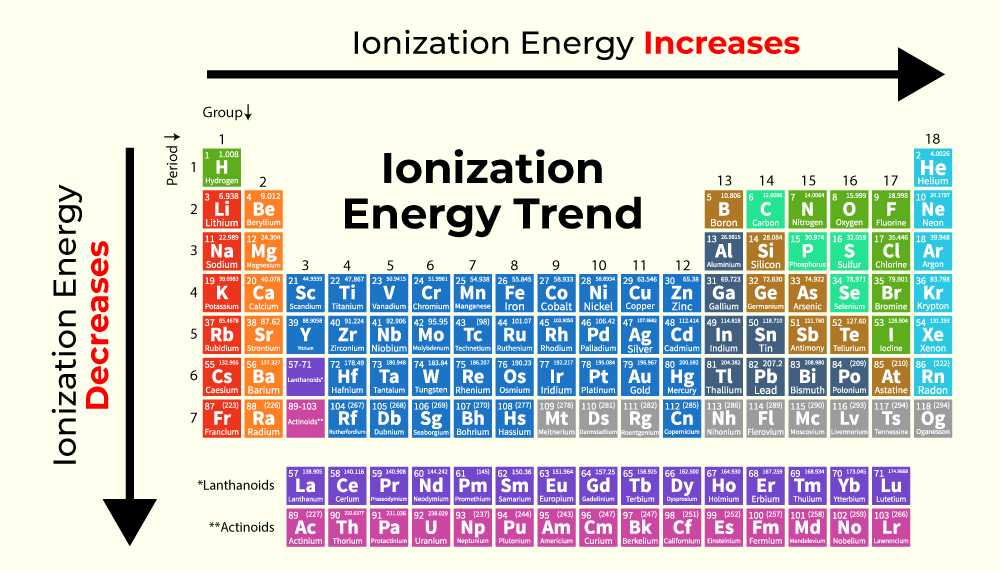
And the order is:
- Lithium (Li)
- Boron (B)
- Beryllium (Be)
- Carbon (C)
- Nitrogen (N)
Pretty neat, huh? You might notice a slight wobble in the pattern, particularly with Beryllium and Boron. That’s part of the charm! It shows that the world of atoms isn’t always a perfectly straight line. There are nuances and fascinating exceptions that make studying them so much fun. It’s a constant reminder that the universe is full of delightful complexities.
This little ranking is just a tiny peek into the vast and wonderful world of chemistry. It’s a fun way to start appreciating the subtle differences between elements and how these differences lead to the incredible diversity of materials we see and use every day. It’s like learning a few basic words in a new language; once you know them, you can start to understand whole sentences and even stories. So, are you curious to learn more about these atomic personalities? There's a whole universe of science waiting to be explored!
