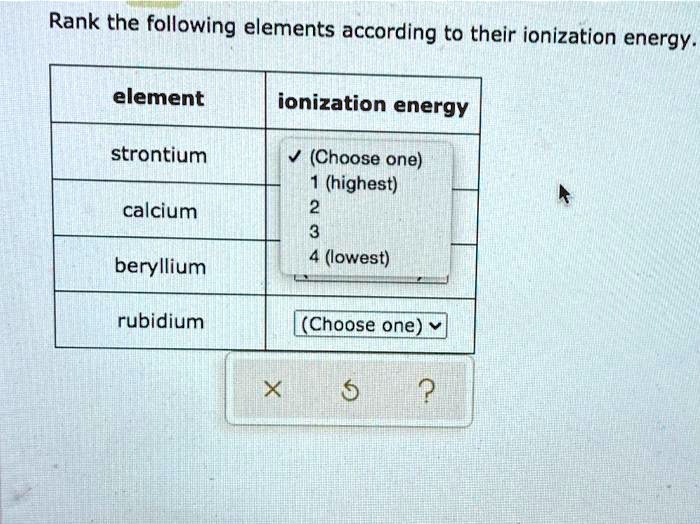
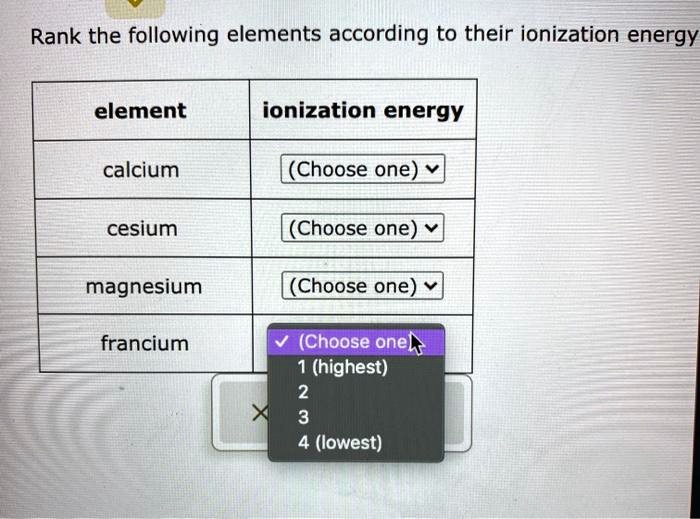
Rank The Following Elements According To Their Ionization Energy

Okay, so we're gonna play a little game today. It's all about ionization energy. Don't worry, it's not as scary as it sounds. Think of it like wrestling with electrons. Who's the toughest to get a piece of? Who gives up their electrons easily? It's a bit like figuring out who's the most dramatic in your friend group.
We've got a list of elements. Some are old friends, some are a bit more obscure. We need to rank them. It's not about who's the best element, or who's the most popular. It's purely about their ionization energy. So, let's get this party started!
First up, we have Lithium. Ah, Lithium. It's like that friend who's always a little too eager to share. Its ionization energy is pretty darn low. It's practically giving away its electrons. You don't have to twist its arm much at all.
Then we have Sodium. Sodium is just like Lithium, but, you know, more so. It’s like they’re competing to see who can be the most generous with their electrons. Seriously, it's like a free-for-all over there.
Now, let's talk about Potassium. Potassium. This one is in the same family as Lithium and Sodium. And guess what? It’s also super chill about letting go of an electron. It’s almost like a tradition in that family. “Here, take it! Don’t mind me!”
So, if we're ranking them from lowest ionization energy to highest, Lithium, Sodium, and Potassium are way down at the bottom. They're the ones who are easily bribed. A little nudge, and poof, an electron is gone.
Next on our imaginary podium, we have Beryllium. Beryllium is a bit more of a tough nut to crack. It’s not as eager as its alkali metal cousins. It holds onto its electrons a little tighter. It’s like it needs a little more convincing.
And then there's Magnesium. Magnesium is in the same group as Beryllium. It's also a bit reluctant, but maybe not quite as much as Beryllium. It's like Beryllium is the grumpy one, and Magnesium is just a little less grumpy.
Moving on, we encounter Calcium. Calcium is another one from that group. It’s similar to Magnesium. It’s in the same ballpark when it comes to clinging to its electrons. They’re all playing the same game, just with slightly different rules.
So, Beryllium, Magnesium, and Calcium. They're above Lithium, Sodium, and Potassium. They require a bit more effort to pry an electron away. They’re not giving them away for free, that’s for sure.
Now, things get a bit more interesting. We're going to throw in some elements that are a little more complicated. Think of them as the ones who have more defenses up.
Let's consider Aluminum. Aluminum is in the same row, or period, as Magnesium. But it’s in a different column. And this makes a difference. Aluminum is a bit more stingy with its electrons than Magnesium.
So, our ranking is starting to get a little more nuanced. Aluminum needs a bit more oomph than Magnesium to lose an electron. It’s like it’s got a better grip on its belongings.
And then we have Silicon. Silicon is right next to Aluminum in the same period. And Silicon is even more hesitant to part with its electrons. It’s like it’s saying, “Nope, not today, thank you very much.”
Think of it like this: Magnesium is like, “Okay, fine, here’s one.” Aluminum is like, “Hmm, let me think about it.” And Silicon is like, “Absolutely not, find another electron.”
Now, let’s introduce Phosphorus. Phosphorus is the next element over from Silicon. And you guessed it, it’s even more resistant. Phosphorus is a real holdout. It doesn't want to let go of any electrons without a serious fight.
So, in this little section, we've got Magnesium, Aluminum, Silicon, and Phosphorus. Their ionization energies are going up as we move across the period. It’s like a gradient of stubbornness.
But wait, there's more! We're going to throw in some elements that are really good at holding onto their electrons. These are the ones you have to wrestle with the most. They are the ultimate electron hoarders.
Let's bring in Sulfur. Sulfur is right after Phosphorus. And it’s a bit of a drama queen when it comes to its electrons. It's got that extra electron cloud thing going on, making it a bit more challenging.
Then we have Chlorine. Oh, Chlorine. This element is famously clingy. It really does not want to give up its electrons. It’s got a strong hold on them. It’s like it’s built a little electron fortress.

And finally, the grand champion of electron hoarding: Argon. Argon is a noble gas. And noble gases are, well, noble. They’re above all this electron-sharing nonsense. They’re perfectly content with their electron setup. They have no interest in making it easier for anyone to steal their electrons.
So, in our final ranking, from the easiest to get an electron from to the absolute hardest, we have a whole spectrum. It's a journey from "take it, it's yours!" to "absolutely not, not even for a million dollars!"
Our order, if we were to be super official about it, and you know, actually do the science, would look something like this, from lowest ionization energy to highest ionization energy:
Lithium, Sodium, Potassium (These guys are the easiest. Seriously, just ask nicely.)
Beryllium, Magnesium, Calcium (A little more effort required. They’re not as spontaneous.)
Aluminum (It’s starting to get a bit more serious.)
SOLVED: Rank the following elements according to their ionizationSilicon (Now we’re talking about a real standoff.)
Phosphorus (Getting feistier. It’s putting up a fight.)
Sulfur (It’s developing some serious defenses.)
Chlorine (This one is a master of electron retention. It’s a tough one.)
Argon (The ultimate winner. It doesn’t give up electrons. Period.)
So there you have it. A playful ranking of elements based on how much they don't want to give up an electron. It's all about that ionization energy hustle. Pretty entertaining, right? My unpopular opinion? Argon is just showing off. But hey, at least it’s consistent!

