Rank The Following Electron-pair Geometries By Increasing Steric Number.
Okay, confession time. When I hear "electron-pair geometry," my brain immediately conjures up images of tiny, invisible balls bumping into each other in a very organized, yet slightly chaotic, dance. It’s like a microscopic rave, and we’re trying to figure out the dance floor layout. It’s not exactly the stuff of blockbuster movies, but hey, someone’s gotta do it, right?
So, we’re talking about these electron pairs. They’re not exactly fond of each other, these guys. Think of it like trying to cram a bunch of teenagers into a tiny car for a road trip. They’re going to spread out as much as humanly possible, right? They want their personal space, and who can blame them?
This is where the concept of steric number waltzes in. It’s basically a fancy way of counting how many "things" are hanging around the central atom. These "things" are our electron pairs, whether they’re busy bonding or just chilling as lone pairs. More things mean more bumping, more nudging, and ultimately, a bigger space required.
Let’s dive into this little popularity contest of geometries. We’re going to rank them, from the most chill and spacious to the oh-my-goodness-get-outta-my-face! It’s a journey, folks, a beautiful, electron-bumping journey.
The Smallest Crowd
First up, we have the grand champion of personal space. This geometry is so laid-back, it practically invented the concept of "social distancing" before it was cool. It's the introvert's dream molecule. Only a select few are allowed in this inner circle.
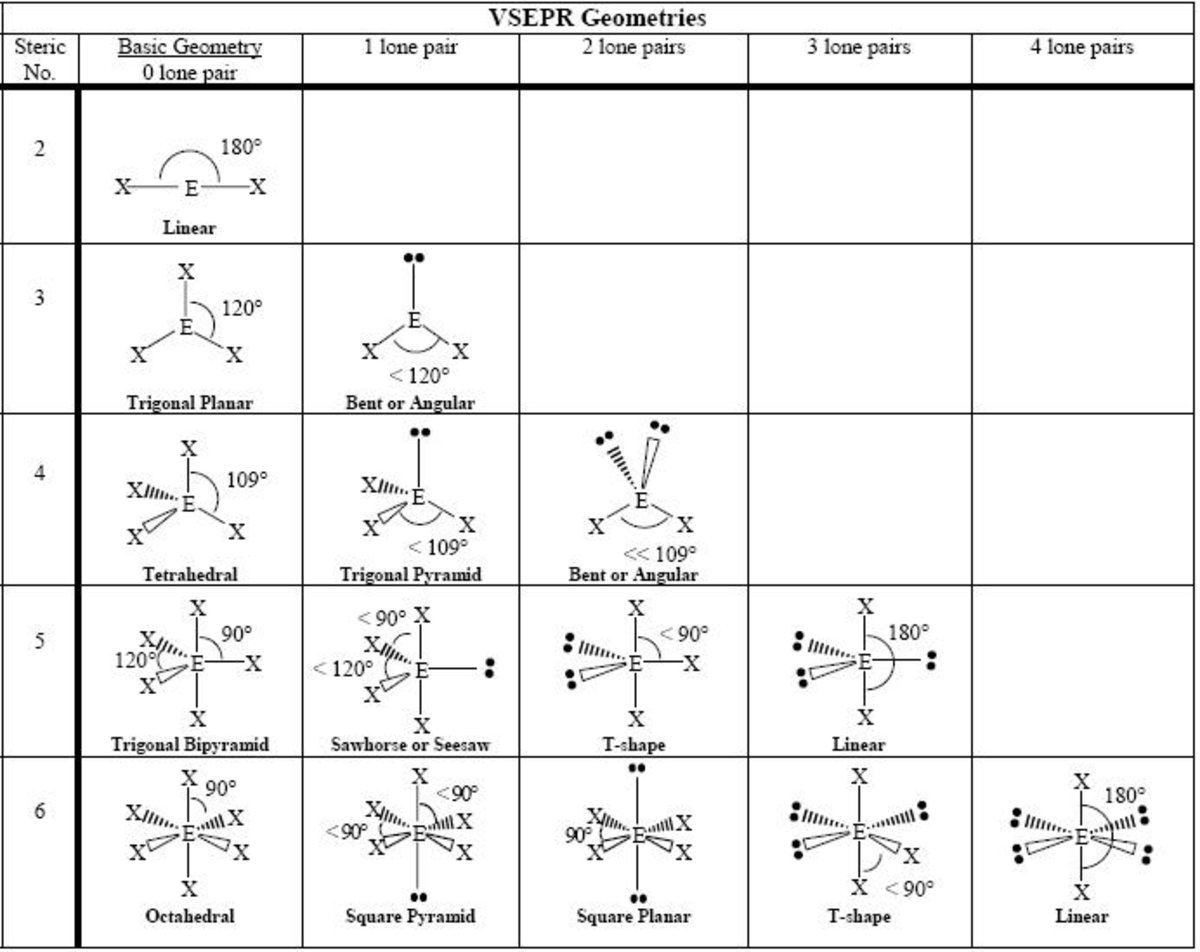
With a steric number of just two, this geometry is all about keeping it simple. Think of a straight line. Nothing too fancy, just two things saying, "Hey, let's not be too close, but not too far either." It’s the perfect amount of separation.
This is the domain of linear geometry. Imagine two balloons tied together with a short string. They’re not going to hug, but they're definitely not going to ignore each other. They've got just enough room to not drive each other completely bonkers.
It’s a simple arrangement, and honestly, I kind of respect it. In a world that’s always trying to cram more in, there’s something refreshing about an arrangement that says, "Nah, two is plenty." It’s like the minimalist aesthetic of molecular structures. Less is more, and in this case, it’s also a whole lot less bumping.

The Slightly More Social Bunch
Moving up the ladder, we encounter a geometry that’s a bit more adventurous. It’s like the group of friends who decided to go on a slightly longer road trip, but they’re still smart enough to get a bigger car. They’re not exactly packed in, but there’s a bit more interaction happening.
Here, our steric number jumps to three. We've got three electron pairs all vying for their slice of the pie. They're not exactly best buddies, but they're trying to make it work without any major drama. It’s a delicate balance.
This is where trigonal planar geometry shines. Imagine a three-leaf clover. Each leaf is a precious electron pair, and they’re all fanned out as much as possible on a flat surface. It’s like a tiny, molecular frisbee.
The beauty of this arrangement is its flatness. It's all happening on one plane, like a perfectly organized picnic blanket. No one is trying to climb over anyone else. It’s efficient, it’s organized, and it’s still got a decent amount of elbow room.
I have to admit, there's a certain elegance to this. It's not overly complicated, but it’s definitely more engaging than the lonely two. It’s like graduating from a single to a double room – a definite upgrade in terms of comfort and options.

The Happy Triangles
Now we're getting into the territory where things start to get a little more three-dimensional. Think of it as upgrading from a flat map to a globe. Things are no longer confined to a single plane, and the electron pairs are really starting to spread their wings.
Our steric number here is a solid four. Four electron pairs, all looking at each other and thinking, "Okay, how do we avoid a full-on molecular mosh pit?" This requires some serious spatial reasoning.
This is the realm of tetrahedral geometry. If you’ve ever seen a pyramid with a flat base and a point at the top, you’re on the right track. But in this case, it’s like a pyramid balanced on its point, with everything fanning outwards.
The key here is that the electron pairs are not on the same level. They’re angled in a way that maximizes their distance from each other in three dimensions. It’s like they’re all trying to reach for the sky, but also lean away from their neighbors simultaneously. It’s a sophisticated ballet.
This geometry feels like a good compromise. It’s not too cramped, and it’s not so spread out that it's unstable. It’s like the perfectly balanced seating arrangement at a family dinner – everyone has enough space, and no one is directly across from the person they secretly dislike.

The Group Hug (Kind Of)
As we ascend, the need for space becomes even more pronounced. Imagine a party where the room is getting crowded. People start to shift, to find corners, to create little pockets of breathing room. This next geometry is the molecular equivalent of finding that sweet spot.
Here, our steric number is five. Five electron pairs, all trying to find their unique spot. This is where things start to get a bit more complex, and frankly, a bit more interesting.
Welcome to trigonal bipyramidal geometry. Now, this one sounds fancy, and it is! Imagine two pyramids stuck together at their bases. Or think of an hourglass with a central point. It’s a shape with two distinct regions of crowding.
You've got some electron pairs that are in the "equatorial" positions, kind of like around the middle of the hourglass. Then you have others in the "axial" positions, sticking up and down. It’s a clever way to distribute five things without them all being in each other’s faces. It's a carefully orchestrated, multi-level arrangement.
I kind of love the ingenuity here. It’s like finding a way to fit five people into a booth that was really only designed for four. You make it work by having some people lean in and others spread out. It’s a testament to molecular problem-solving.
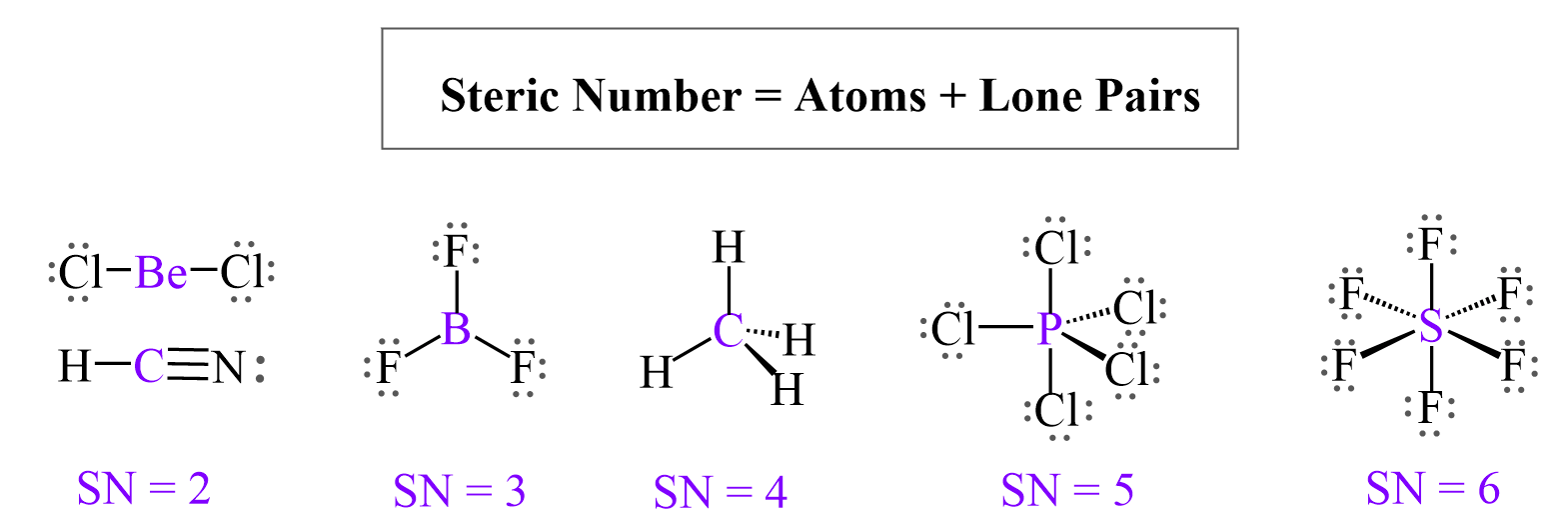
The Ultimate Space Invaders
Finally, we reach the pinnacle of electron-pair congestion. This is the geometry where you’ve invited everyone to the party, and the room is definitely feeling it. There’s a lot of shuffling, a lot of polite (or not-so-polite) nudging.
With a steric number of six, we have the most crowded situation we're looking at today. Six electron pairs are trying to coexist, and they're going to do everything in their power to not be too close.
This is the domain of octahedral geometry. Picture a pyramid with a square base, but then imagine another identical pyramid flipped upside down and stuck on top. It looks like two square pyramids joined at their bases. It’s a very symmetrical, and very spread-out, arrangement.
In this geometry, all the positions are actually equivalent. Unlike trigonal bipyramidal, where there’s a clear distinction, in octahedral, every electron pair has the same kind of spacing from its neighbors. It’s like a perfectly fair game of musical chairs, where everyone gets a decent spot.
This geometry is the ultimate testament to electron repulsion. It’s saying, "We need all this space, and we’re going to get it, no matter what!" It’s a bit extreme, but honestly, I admire the commitment to personal space on such a grand scale. It’s the molecular equivalent of booking out an entire convention center for your birthday.
So, there you have it. The ranking, from the most spatially relaxed to the most determined to avoid a molecular traffic jam. It’s a journey through the world of electron-pair geometries, proving that even at the atomic level, personal space is a big deal. And honestly, I’m here for it. It's a reminder that sometimes, the simplest arrangements are the most elegant, and sometimes, you just need a whole lot of room.
