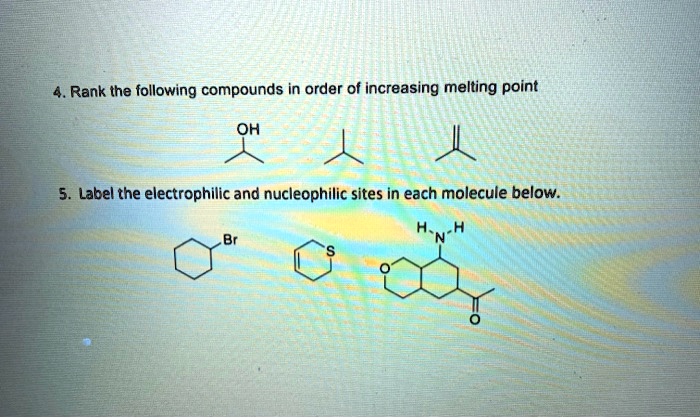
Rank The Following Compounds In Order Of Increasing Melting Point
Alright, science fans, and even you folks who just clicked because you saw the word "compounds" and thought it sounded fancy! Today, we're diving into the wonderfully weird world of melting points. Forget your complicated formulas and dense textbooks. We're talking about how easily (or not so easily) some stuff turns from solid rock to sloshy liquid. Think of it like trying to convince your teenager to clean their room. Some things just resist change!
So, I've got a little lineup of compounds here. And my mission, should I choose to accept it (and I did, because free internet points!), is to rank them. From the ones that practically melt if you just look at them funny, to the ones that could probably survive a dragon's fiery breath. This is my completely unofficial, slightly biased, and definitely fun-focused ranking. Buckle up, buttercups!
First up, we have the absolute melt-master. The champ of going liquid. The king of… well, becoming runny. I'm talking about sodium chloride. You know, the stuff you put on your fries? Yeah, that’s the one. It's like the most chill substance on the planet. Give it a little nudge, maybe a warm breeze, and poof – it's a puddle. It’s so easygoing, it’s practically begging to become a liquid. It’s the friend who’s always up for anything, no questions asked.
Next on our melt-ability scale, we've got something a bit more… stoic. Meet water. H₂O, folks. Our lifeblood. The stuff that makes puddles and polar bears. Water's got a decent melting point. It's not a wimp like some others we'll get to, but it’s definitely not a superhero either. It’s like your average adult. Needs a bit of coaxing, a good solid zero degrees Celsius, to get it moving from ice to water. It’s got its boundaries, but it’s not entirely inflexible.
Now, things are starting to get interesting. We're moving into the territory of the stubborn. These compounds are like, "Nah, I'm good as a solid, thanks." Our next contender is aluminum oxide. This stuff is tough. You ever see a really shiny, hard ceramic mug? There's a good chance aluminum oxide is involved. It’s like that one friend who is always impeccably put together, even on a Monday morning. It takes a serious amount of heat to convince this one to even think about melting. It’s got a serious case of "staying put."
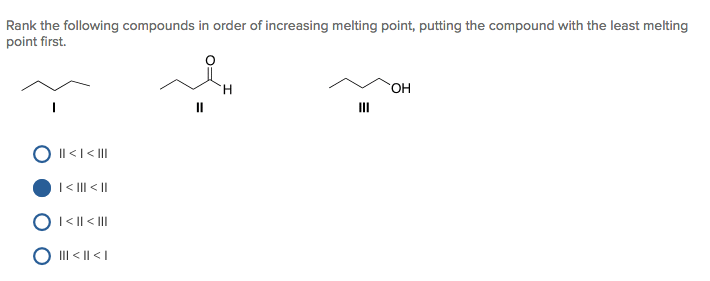
We're getting closer to the ultimate melt-resisters now. This next one is practically a fortress. Introducing silicon dioxide. You know this guy as sand. Yes, plain old sand! Think about a beach on a scorching hot day. The sand is there, doing its sandy thing. It's not exactly turning into a molten lava flow, is it? It’s got a serious backbone. It’s the grumpy old man who sits on his porch and watches the world go by, totally unfazed by the chaos. It’s like, "You can try all you want, but I'm staying solid."
Honestly, sometimes I think silicon dioxide just enjoys being solid. It’s like its personal mission statement.
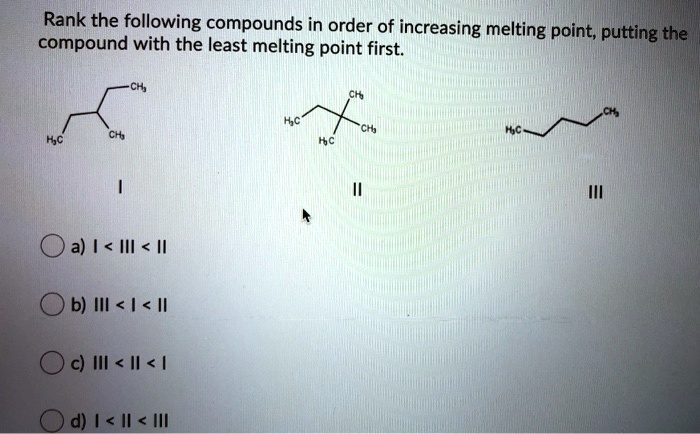
SOLVED: Rank the following compounds in order of increasing melting
And finally, the grand champion of not melting. The heavyweight titleholder of staying solid. We're talking about tungsten. This metal is so unbelievably stubborn, it’s almost admirable. It’s used in things that need to withstand insane temperatures, like the filaments in old light bulbs. Those things got hot, but tungsten just shrugged it off. It’s the ultimate "nope" to melting. It’s like the person who refuses to take a vacation. Ever. It’s committed. It's the rock of Gibraltar in the melting point world. It’s the one that makes you think, "Seriously, how hot do you need to get this thing?"
So, there you have it. My totally scientific (wink wink) ranking from easiest to melt to practically impossible: sodium chloride, then water, followed by the ever-so-stubborn aluminum oxide, the sandy powerhouse silicon dioxide, and finally, the undisputed, unyielding king of solids, tungsten. It’s a journey from "oh, is that getting a bit warm?" to "better call the fire department… and maybe a rocket ship." Who knew something as simple as melting could be so… dramatic? It’s all about who’s willing to get a little bit (or a whole lot) hotter. And some are just built tougher than others, I guess. Kind of like us, right?