Rank The Following Compounds In Order Of Increasing Boiling Point
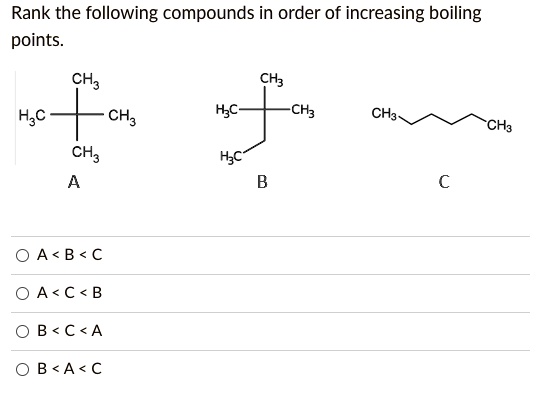
Get ready for a little scientific adventure! We're about to dive into something super cool: ranking compounds by their boiling points. Think of it like a friendly competition where molecules are jostling to see who can hang onto their liquid form the longest. It's all about the forces between them, and it's more fascinating than you might imagine.
Imagine you have a bunch of tiny dancers. Some dancers are really good at holding hands, while others are a bit more free-spirited. The "holding hands" ones need a lot more energy to break free and start their solo dance (boiling). That's essentially what we're exploring with these compounds.
So, what compounds are we talking about today? We've got a fun lineup! We're looking at Methane, Ethane, Propane, and Butane. These are all pretty common. You might have even encountered them without realizing it!
Think about your stove. If you have a gas stove, there's a good chance you're using Propane or a mix that includes it. Butane is often found in lighter fluid and camping stoves. And Methane? That's the main ingredient in natural gas, used to heat many homes.
It's amazing how these simple-looking molecules play such big roles in our everyday lives. And their boiling points? They tell a story about how they behave. It's like their own little personality test.
Let's get to the fun part: the ranking! We're going to arrange them from the lowest boiling point to the highest. This means the first one on our list will be the most eager to turn into a gas. It's the easiest one to get excited and let loose!
At the very bottom of our list, the champion of easy vaporization, is Methane. This little guy is the simplest of the bunch. It's got just one carbon atom and four hydrogen atoms attached.
Because Methane is so small and simple, the forces holding its molecules together are quite weak. Think of it as having just a gentle handshake between molecules. A tiny bit of heat is all it takes to break those bonds and send it off into the gaseous world.
Next up, we have Ethane. Now, Ethane is a step up. It has two carbon atoms and six hydrogen atoms. It's like Methane's slightly bigger sibling.

With an extra carbon atom, Ethane's molecules have a bit more surface area. This allows for slightly stronger attractions between them. It needs a little more effort, a bit more "oomph," to get these molecules to separate and boil.
So, Ethane's boiling point is higher than Methane's. It's a noticeable difference, showing how even a small change in molecular structure can have an impact. It’s like upgrading from a quick wave hello to a slightly longer hug.
Holding the third position is Propane. This is the molecule that makes so many of our outdoor cooking dreams come true! Propane has three carbon atoms and eight hydrogen atoms. It’s getting a bit more substantial now.
As we add more carbon atoms, the molecules become larger. This increased size means there are more opportunities for the molecules to interact with each other. The attractions between Propane molecules are stronger than those in Ethane.
To make Propane boil, we need to supply more energy. It's like going from that hug to a full-on group dance. The molecules are more entangled, and it takes a more energetic push to get them dancing solo.
And finally, at the top of our list, the most reluctant to boil, is Butane. This compound has four carbon atoms and ten hydrogen atoms. It’s the largest molecule in our current lineup.
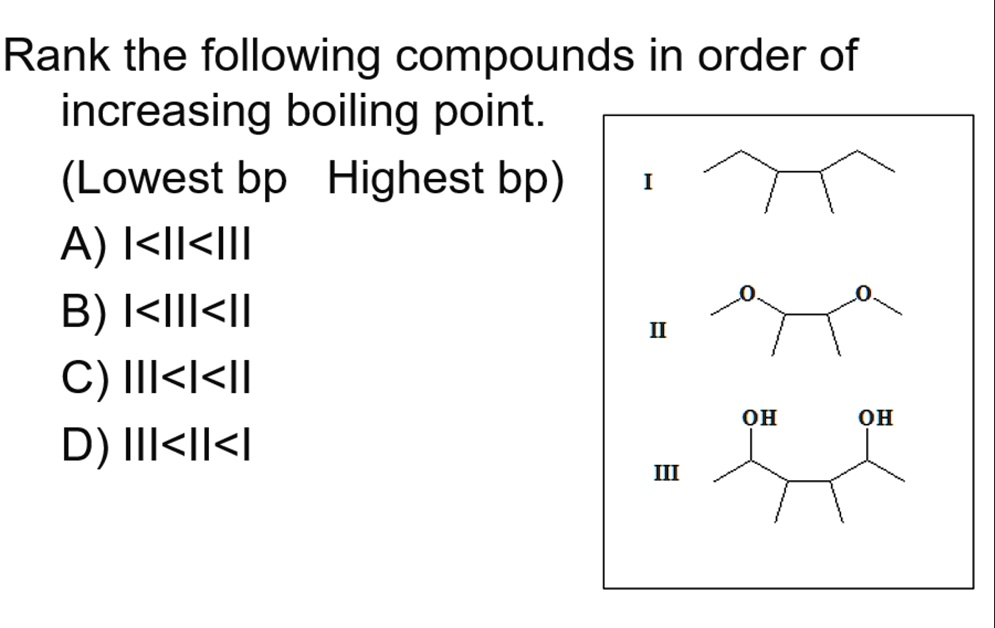
Butane molecules are bigger and have more electrons. This leads to stronger intermolecular forces, specifically London dispersion forces, which are a type of temporary attraction that arises from the movement of electrons. The bigger the molecule, generally, the stronger these temporary attractions can be.
Think of Butane as having a really strong grip on its neighbors. It needs a significant amount of heat to break free and become a gas. This is why it's often used in applications where a more stable, less volatile fuel is desired.
So, let's recap our exciting order! From lowest boiling point to highest, it's:
Methane (the easiest to boil)
Ethane
Propane
Butane (the hardest to boil)
Isn't that neat? We started with simple molecules and saw how a little bit of extra size and structure dramatically changed their boiling points. It’s a clear demonstration of a fundamental principle in chemistry.
This concept of intermolecular forces is what makes so many things in our world work the way they do. It's the reason water boils at a specific temperature, and why different liquids behave differently. It’s the invisible glue that holds matter together.
What makes this ranking so special is that it’s not just abstract numbers. These are real compounds with real-world applications. Understanding their boiling points helps us design fuels, create refrigerants, and even understand weather patterns.
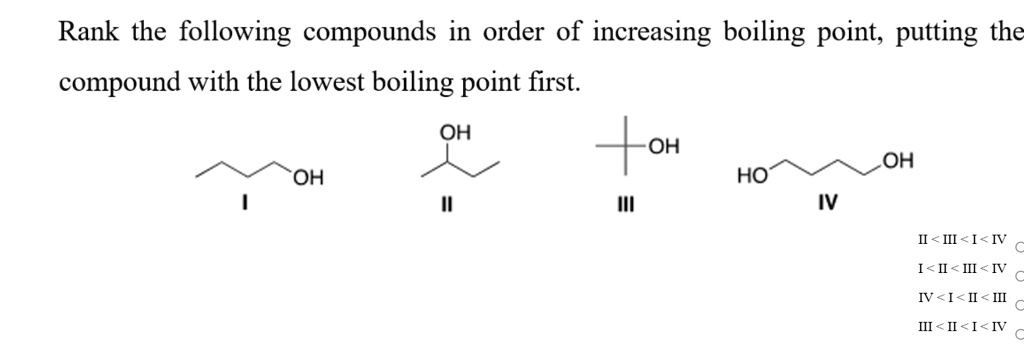
It’s like unlocking a secret code for how substances interact. The journey from Methane to Butane shows a clear trend. As the molecules get larger and have more electrons, their boiling points steadily increase. This is a direct consequence of stronger forces between the molecules.
The beauty of this is its simplicity, yet its profound implications. You don't need a fancy lab coat to appreciate it. Just a bit of curiosity and an open mind.
Imagine you're a chef. Knowing the boiling points of ingredients helps you cook them perfectly. Too low, and they won't cook. Too high, and they might burn. It’s all about finding that sweet spot, just like with these compounds.
This isn't just about these four alkanes, either. This principle applies to countless other substances. The same idea of stronger attractions meaning higher boiling points holds true across a vast range of molecules.
It’s a little like a puzzle. Each molecule has its own unique set of attractions. By understanding these attractions, we can predict how they will behave under different conditions, like when heated.
The names themselves, Methane, Ethane, Propane, Butane, sound a bit like a secret language. But once you understand the pattern, they become quite familiar. They’re the building blocks of many organic compounds.

The increase in boiling point as you go up the series is so consistent. It's a beautiful example of how nature follows predictable rules. Even at the molecular level, there's an underlying order.
Think about it: these are all gases at room temperature, but they can be liquefied and used as fuels. This transformation is directly linked to their boiling points. Understanding this helps us harness their power safely and effectively.
So, the next time you see a camping stove ignite or smell that familiar gas from your kitchen, you’ll have a little more insight into the science behind it. It’s the molecules themselves, dancing their way into different states of matter.
The progression from Methane to Butane is a fantastic introductory lesson. It's accessible, relatable, and truly demonstrates a core chemical concept in an engaging way. It makes you wonder what other secrets the world of molecules holds.
This is what makes science so captivating. It's not just about memorizing facts; it's about understanding the 'why' behind everything. And in this case, the 'why' is all about how strongly molecules hold onto each other.
It's the subtle differences that make the biggest impact. That slight increase in size, the extra atom here or there, can change a substance's boiling point significantly. It's a testament to the intricate design of the molecular world.
So, there you have it! Our little lineup of compounds, ranked from easiest to hardest to boil. It's a simple yet powerful illustration of a fundamental scientific principle. Keep that curiosity alive, and you'll find wonder in the most unexpected places!
