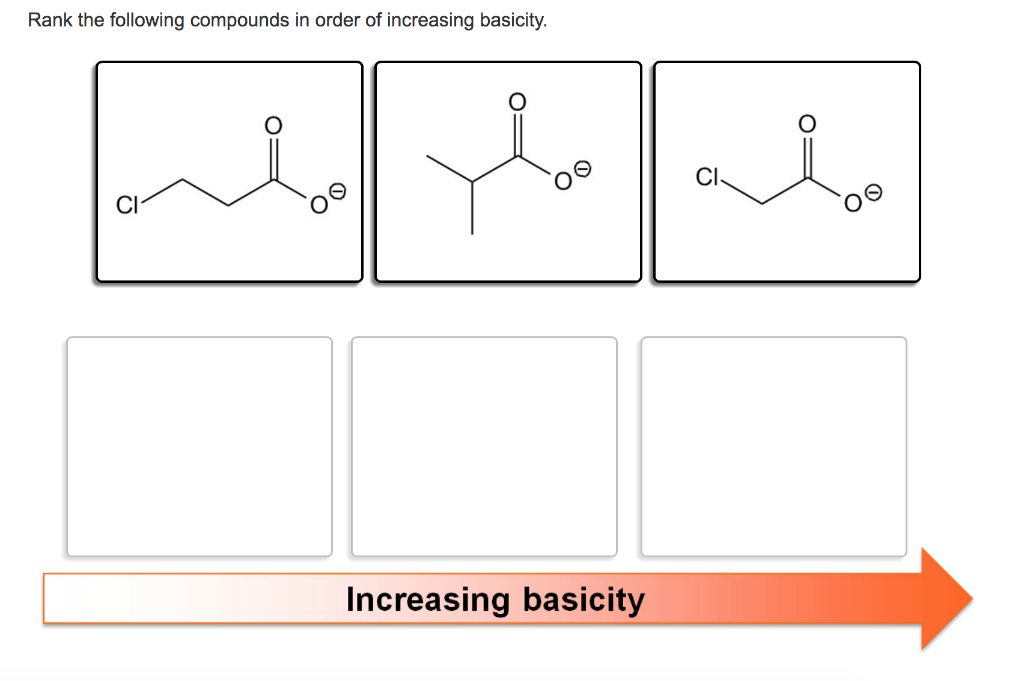
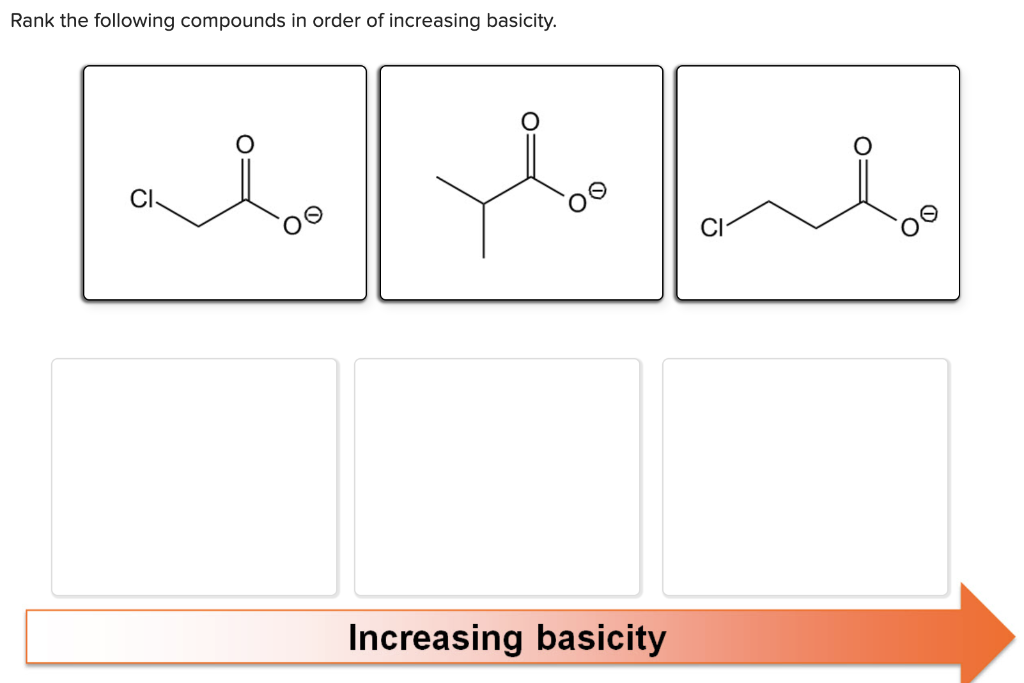
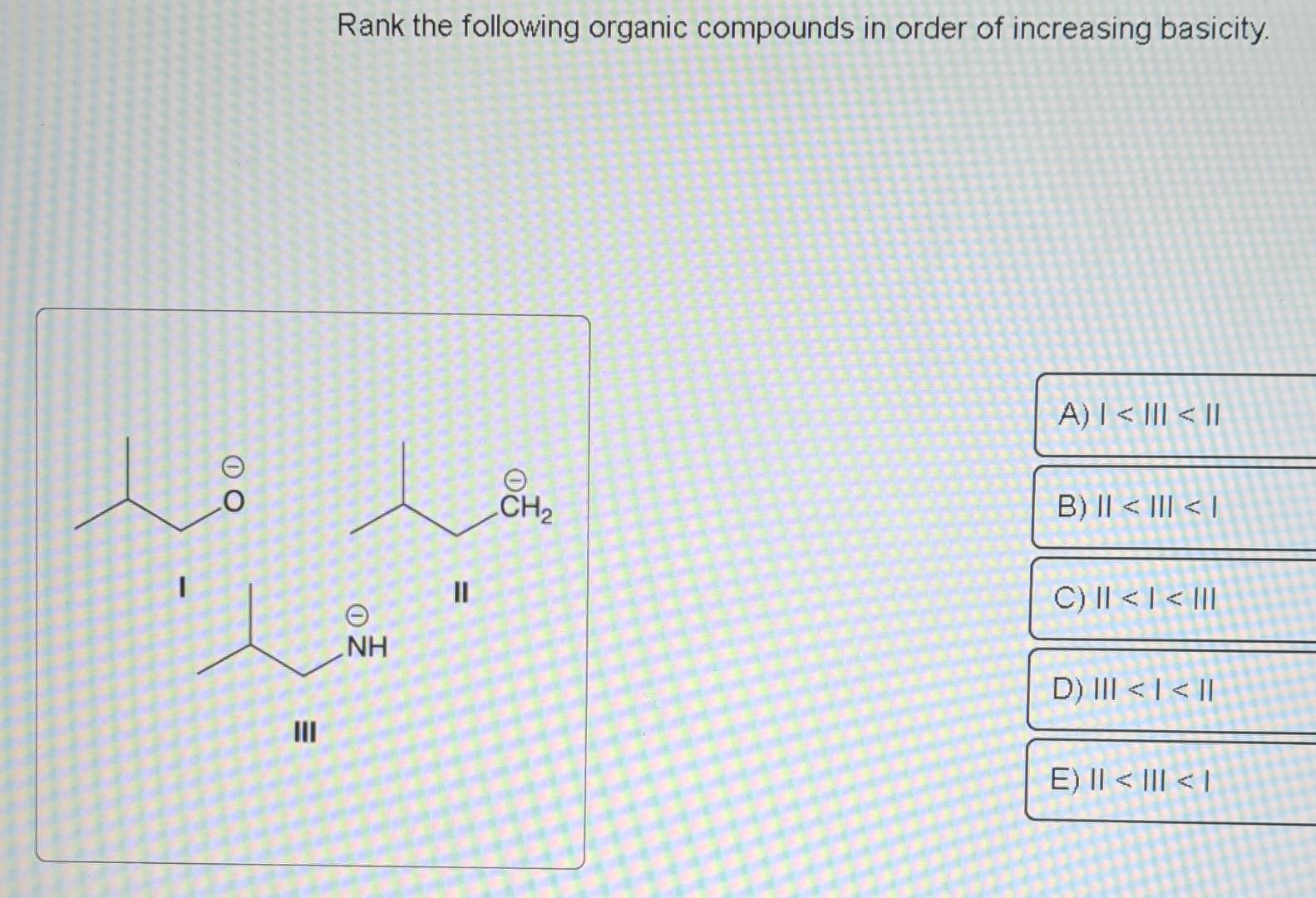
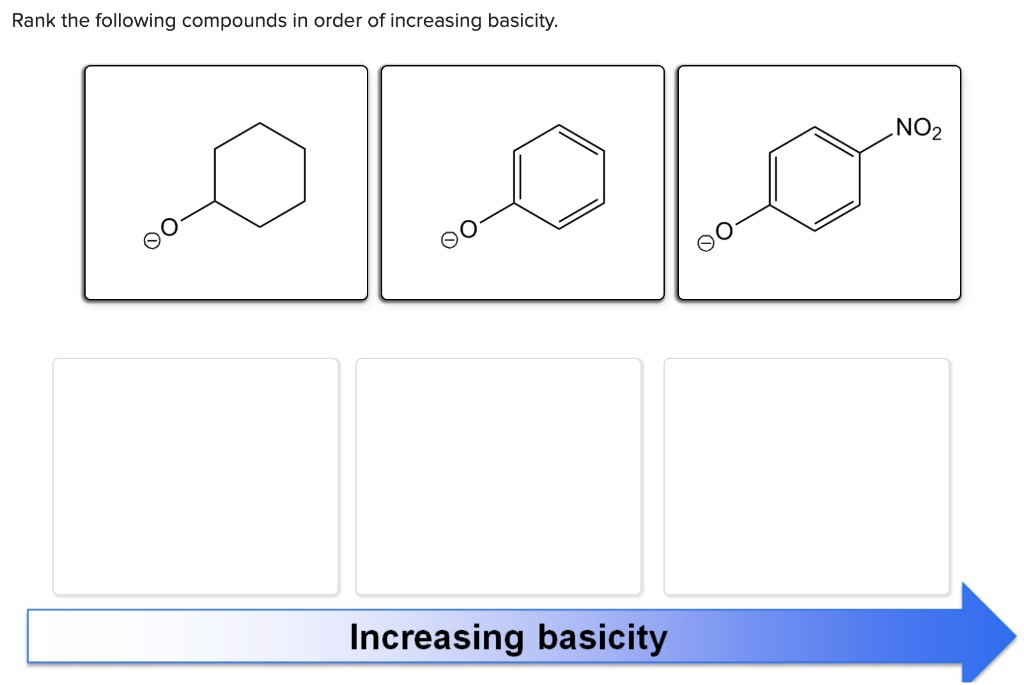
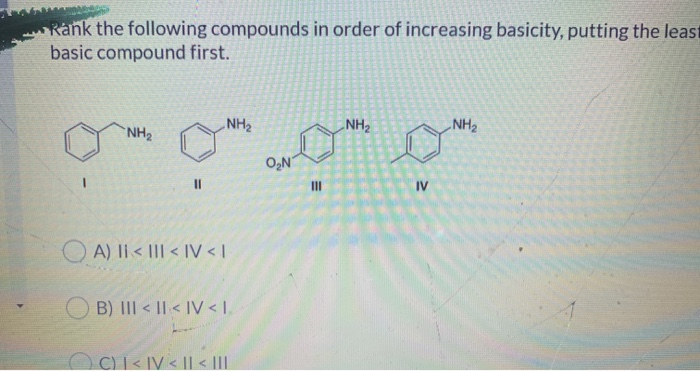
Rank The Following Compounds In Order Of Increasing Basicity
Hey there, chem curious cats! Ever wonder what makes some things more "clingy" than others, chemically speaking? Today, we're diving into the wild world of basicity. Think of it like a popularity contest for molecules. Who's the life of the party, ready to share their electron toys? We're gonna rank some compounds and figure out who's the ultimate electron-donor extraordinaire!
So, what exactly IS basicity? It's all about a molecule's ability to accept a proton. Protons are like the tiny, energetic toddlers of the chemical world, always wanting to latch onto something. Bases are the super chill parents who are happy to let these protons hang out for a bit. The more willing a molecule is to take on a proton, the stronger its base game is.
It's not just about being a grumpy scientist in a lab coat. Understanding basicity helps us explain everything from why your antacid actually works to how your body digests food. Pretty cool, right? It’s like having a secret code to understand the everyday magic happening around you.
Let's Meet Our Contestants!
We've got a lineup of molecules ready to duke it out for the title of "Most Basic." Think of them as contestants on a quirky game show. We've got some familiar faces and a few that might make you scratch your head. Don't worry, no pop quizzes here, just pure, unadulterated chemical fun!
The Contenders
First up, we have water (H₂O). Yeah, that stuff you drink and shower with. It's kind of the middle-of-the-roader in this competition. It can be acidic or basic, depending on its mood, which is kinda like that friend who goes with the flow.
Next, we've got ammonia (NH₃). This one's a classic. It's got that extra electron pair just begging to be shared. Think of it as the eager beaver of the group, always ready to lend a proton.
Then, there's hydroxide ion (OH⁻). This is like ammonia's slightly more intense sibling. It's already got a negative charge, making it super attractive to those protonic toddlers. It's practically a proton magnet!
And finally, we're throwing in methoxide ion (CH₃O⁻). This one's got a similar setup to hydroxide but with a little extra "oomph" from that carbon group attached. It's like hydroxide with a side of extra confidence.
The Ranking Reveal: Who's King of the Castle?
Alright, drumroll please! We're gonna rank these guys from least basic to most basic. Think of it as a slow build-up to the ultimate proton party. Get ready for some surprises!
The Underdogs (Least Basic)
Starting at the bottom, we have water. Why? Because while it can accept a proton, it's not exactly jumping for joy. It's more like, "Eh, sure, I guess I'll hold your proton for a sec." It's pretty stable on its own, so it doesn't have a burning desire to become something else by grabbing a proton.
Imagine water as the polite guest at a party. It’s perfectly pleasant, but it’s not the one starting the conga line. It’s got its own thing going on, and taking on another proton would kind of change its whole vibe. And honestly, who wants to mess with the vibe of perfectly good water?
The Rising Stars
Moving up the ladder, we find ammonia. Now we're talking! Ammonia has that lone pair of electrons just practically screaming, "Pick me! Pick me!" It's way more inclined to accept a proton than water. It's like the friendly neighbor who’s always happy to babysit.
Think of ammonia’s nitrogen atom as having a little pocket, just perfect for tucking a proton away. It’s not as desperate as some others, but it’s definitely more enthusiastic than water. It’s the friend who brings snacks to the party, you know?
The Powerhouses
Getting into the serious basicity zone, we have hydroxide ion. This is where things get really interesting. Hydroxide is already negative. That negative charge is like a giant neon sign saying, "Protons welcome! All aboard!" It's way, way more basic than ammonia.
Hydroxide is like the host of the party who’s already got confetti in their hair and is handing out party favors. It’s actively seeking out those proton toddlers. It’s got that extra electron density just itching to bond. You can almost hear it cheering, "More protons, please!"
The Reigning Champion (Most Basic)
And the winner is... methoxide ion! Boom! Methoxide is the undisputed champion of this particular showdown. It’s even more electron-rich and proton-hungry than hydroxide. Why? That little methyl group (CH₃) is like a tiny electrical booster shot, making the oxygen even more negative and therefore, even more eager to snatch a proton.
Methoxide is the life of the party, the one dancing on the tables, the one who brought the karaoke machine. It’s so good at accepting protons that it makes hydroxide look a bit reserved. It’s like the ultimate, super-charged proton-grabbing machine. It’s basically saying, "You want to give me a proton? Great! I'll take ten!"
The Quirky Details You Didn't Know You Needed
So, why is this whole "proton-grabbing" thing so important? Well, think about it. When you have heartburn, you take an antacid, right? That antacid is usually a base, and it works by neutralizing the stomach's excess acid. It's basically these basic compounds doing their job, taking those pesky protons away from the acid!
It's also crucial in reactions happening inside your cells. Your body is a finely tuned chemical orchestra, and basicity plays a huge role in keeping everything in harmony. Without it, our bodies wouldn't be able to do all the amazing things they do, from building muscles to thinking deep thoughts.
And get this: the weaker the base, the stronger its conjugate acid. It's like a chemical seesaw! So, water, our weakest base, has a pretty strong conjugate acid (hydronium ion). Methoxide, our strongest base, has a very weak conjugate acid. It’s a whole interconnected universe of chemical relationships!
Why It's Just Fun to Talk About
Honestly, isn't it kind of amazing that we can look at these tiny, invisible molecules and understand their personalities? They have preferences, they have tendencies, they're literally battling it out for protons! It’s like a microscopic soap opera.
And the best part? You don't need a fancy lab coat to appreciate it. Just by understanding these basic principles, you can start to see the chemical world in a whole new light. Next time you reach for an antacid or notice how something fizzes, you'll know there's a whole world of basicity at play!
So, there you have it! Our basicity contestants, ranked and ready. Remember, it's all about who's willing to share those electron toys and become the best proton-holding buddy. Keep your eyes peeled, and who knows what other chemical personalities you'll discover!
