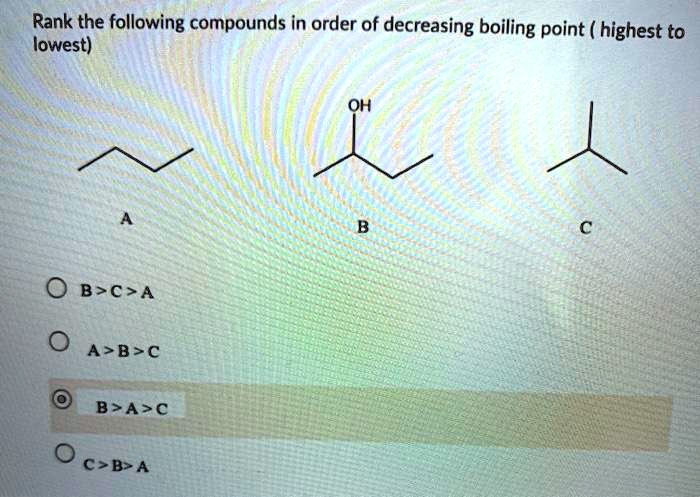
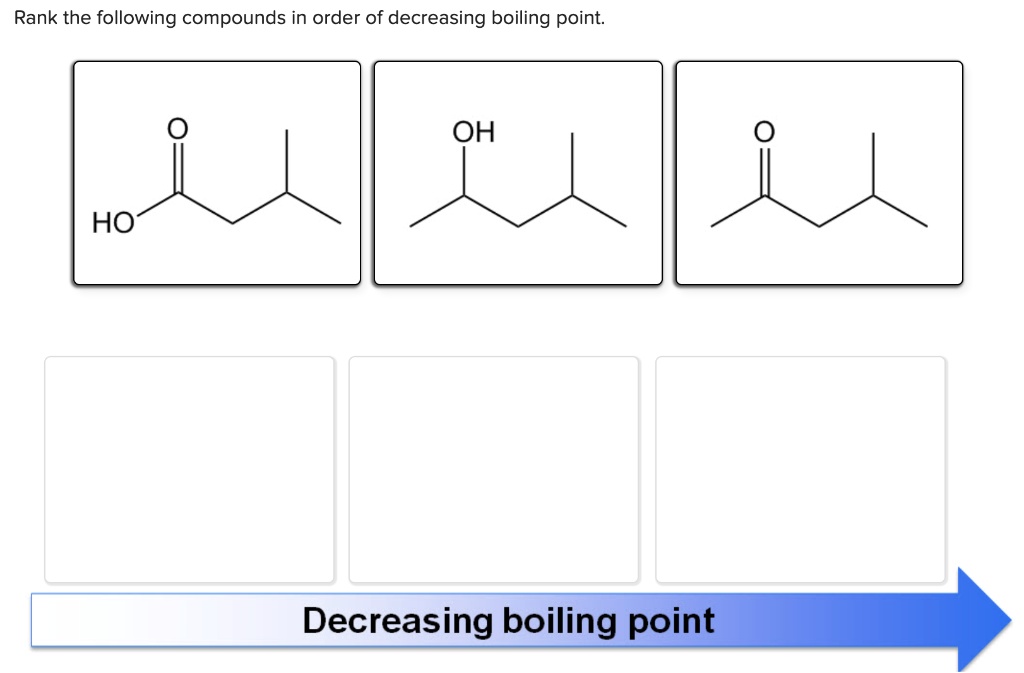
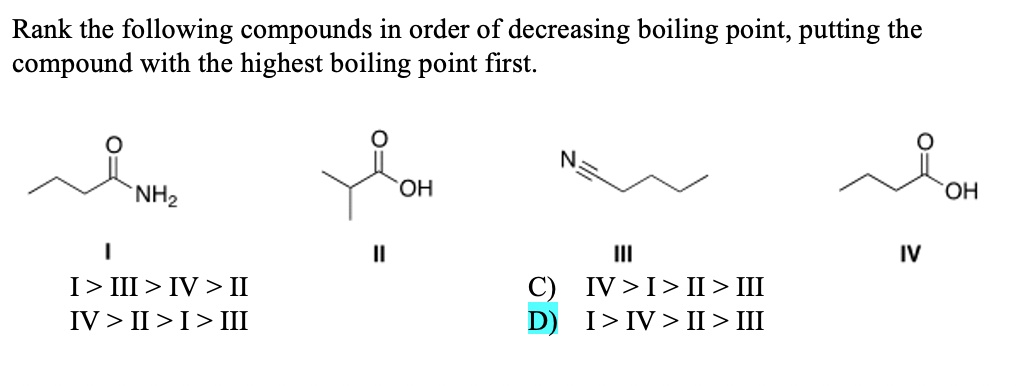
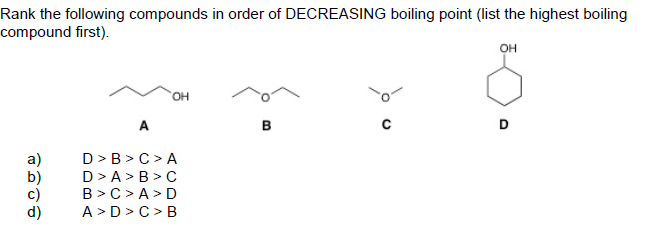
Rank The Following Compounds In Order Of Decreasing Boiling Point
Hey there, science adventurer! Ever wonder what makes water boil and coffee perk? It’s all about boiling points. And today, we're gonna dive into a super cool challenge: ranking some compounds by how much heat they can take before they go from liquid to gas. Think of it like a molecular showdown! Who’s the toughest? Who’s a total wimp? Let’s find out!
We’ve got a lineup of molecules that are pretty common, but their boiling points are anything but ordinary. We’re talking about:
- Methane (CH4)
- Water (H2O)
- Sodium Chloride (NaCl)
- Ethanol (C2H5OH)
Get ready to have your mind blown. This isn't just about numbers; it's about the invisible forces holding these tiny things together. It’s like a cosmic dance party where some molecules are holding hands really tight, and others are just casually waving hello.
The Challenge: Who’s the Hottest Mess?
Our mission, should we choose to accept it (and we totally should!), is to order these guys from highest boiling point to lowest. So, who needs the most oomph to turn into a gas? Who’s easily swayed by a little heat?
Let’s break down what a boiling point actually is. Basically, it’s the temperature where a liquid’s vapor pressure equals the surrounding atmospheric pressure. Sounds fancy, right? But what it really means is: when the liquid molecules have enough energy to escape into the air, poof, they’re a gas! And how much energy they need depends on how much they like hanging out together in their liquid form.
The Players and Their Quirks
Before we start ranking, let’s get to know our contestants. They’re all fascinating in their own ways!
Methane (CH4): The Tiny Titan (or not?)
First up, we have methane. This is the main ingredient in natural gas. You know, the stuff that makes your stove work and, uh, can cause some interesting atmospheric phenomena. It’s a super simple molecule, just one carbon atom hugging four hydrogen atoms.
Think of methane as the shy, introverted kid at the party. It doesn't really have any friends to hold onto. The forces between methane molecules are really, really weak. Like, a gentle breeze could probably get them to float away from each other. This means it doesn't take much heat to get methane to turn into a gas. It’s practically gaseous already!
Fun fact: Methane is a potent greenhouse gas. So while it’s great for cooking, it’s not so great for the planet. Also, ever heard of swamp gas? Yep, that’s often methane!
Water (H2O): The Classic Cool Kid
Ah, water. The universal solvent. The essence of life. It’s the stuff we drink, swim in, and cry over. Water is made of one oxygen atom and two hydrogen atoms, and it's got a special trick up its sleeve: hydrogen bonding.
Imagine water molecules are like little magnets. The oxygen end is a bit negative, and the hydrogen ends are a bit positive. They’re attracted to each other. So, when water is in liquid form, these molecules are all holding hands, forming little networks. It takes a good amount of energy (heat!) to break these sticky hydrogen bonds and let the molecules escape. That's why water boils at a respectable 100°C (212°F).
Quirky detail: Ice floats! This is thanks to the extensive hydrogen bonding in the solid state, making it less dense than liquid water. Pretty neat, huh? Without this, lakes would freeze solid from the bottom up, and life as we know it might not exist. Thanks, water!
Ethanol (C2H5OH): The Party Animal
Now, let’s talk about ethanol. This is the alcohol in your favorite (or not so favorite) beverages. It's got a bit more going on than water. It has a carbon chain (that's the C2H5 part) and then an -OH group, just like water has.
Ethanol can also form hydrogen bonds, just like water. But, it's also got this "hydrophobic" (water-fearing) carbon tail. This tail doesn't really want to interact with other ethanol molecules that strongly. So, while it has hydrogen bonding, it's not as strong as in pure water. Think of it as having some friends to hold hands with, but also some who are a bit more independent.
This means ethanol needs less energy to boil than water. It’s more eager to become a gas. You can also say it’s more volatile, which is a fancy word for "likes to evaporate easily." And yes, that's why alcohol on your skin feels cold as it evaporates – it's taking heat away!
Sodium Chloride (NaCl): The Ultimate Hugger
Finally, we have sodium chloride, better known as table salt! This stuff is what makes our food delicious. But when we're talking about boiling points, it’s in a league of its own. Sodium chloride isn't made of molecules in the same way the others are. It's an ionic compound.
Imagine sodium (Na+) and chloride (Cl-) as super-charged particles, one positively charged and one negatively charged. They are intensely attracted to each other, forming a crystal lattice. This attraction, called an ionic bond, is way, way stronger than the intermolecular forces in methane, water, or ethanol. It's like a giant, super-glued embrace that’s really hard to break!
To get sodium chloride to boil, you need an incredible amount of heat. We’re talking temperatures hotter than a volcano’s surface. It’s a tough cookie, this salt!
The Grand Ranking: Boiling Point Showdown!
Alright, time for the main event! Let's line them up from the highest boiling point to the lowest. Who wins the gold medal for needing the most heat?
1. Sodium Chloride (NaCl): The Unmovable Mountain
Seriously, salt needs a mind-boggling 1465 °C (2669 °F) to boil. It’s so high, it’s almost theoretical for most everyday situations. You’d need industrial furnaces to even think about boiling salt.
Why so high? Those ionic bonds are the ultimate clingy friends. They just refuse to let go. It’s the strongest attraction we've got in our list.
2. Water (H2O): The Strong Hugger
Our beloved water comes in next with a boiling point of 100 °C (212 °F). It needs significant heat, but nothing compared to salt. Those hydrogen bonds are strong, but they are breakable with enough kitchen-stove power.
It’s amazing that life thrives at these temperatures. Imagine if our blood boiled at room temperature! Definitely not a fun party.
3. Ethanol (C2H5OH): The Energetic Entertainer
Ethanol is next, boiling at about 78.37 °C (173.07 °F). It's got some hydrogen bonding going on, but its non-polar carbon chain makes it a little less inclined to stick together than water. It’s more ready to hit the gas phase.
This lower boiling point is why you can smell alcohol evaporating so easily. It’s already eager to join the air party!
4. Methane (CH4): The Faint Whisper
And at the very bottom, needing the least amount of heat, is methane, with a boiling point of -161.5 °C (-258.7 °F). Yes, you read that right. It’s negative! This means at room temperature, methane is already a gas. It’s practically boiling before you even turn on the heat!
This is because methane molecules are like ships passing in the night. They have very weak intermolecular forces (Van der Waals forces, if you want to get technical). They barely acknowledge each other's existence.
The Takeaway: It’s All About the Bonds!
So there you have it! The order of decreasing boiling points is: Sodium Chloride > Water > Ethanol > Methane.
It all comes down to how tightly the particles are held together. Ionic compounds like salt have super strong bonds. Polar molecules like water have strong hydrogen bonds. Less polar molecules like ethanol have weaker attractions. And non-polar molecules like methane have the weakest forces of all.
Isn't science fun? It’s like unlocking little mysteries about the world around us. Next time you boil water for your tea, you can impress your friends with your newfound knowledge of molecular dance parties. Stay curious!
