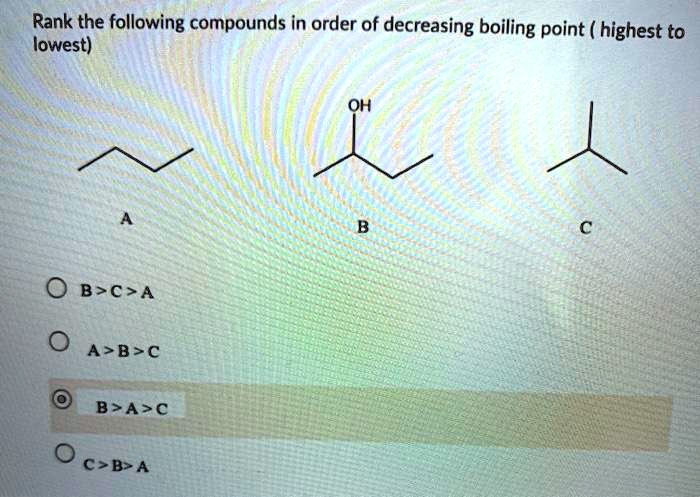
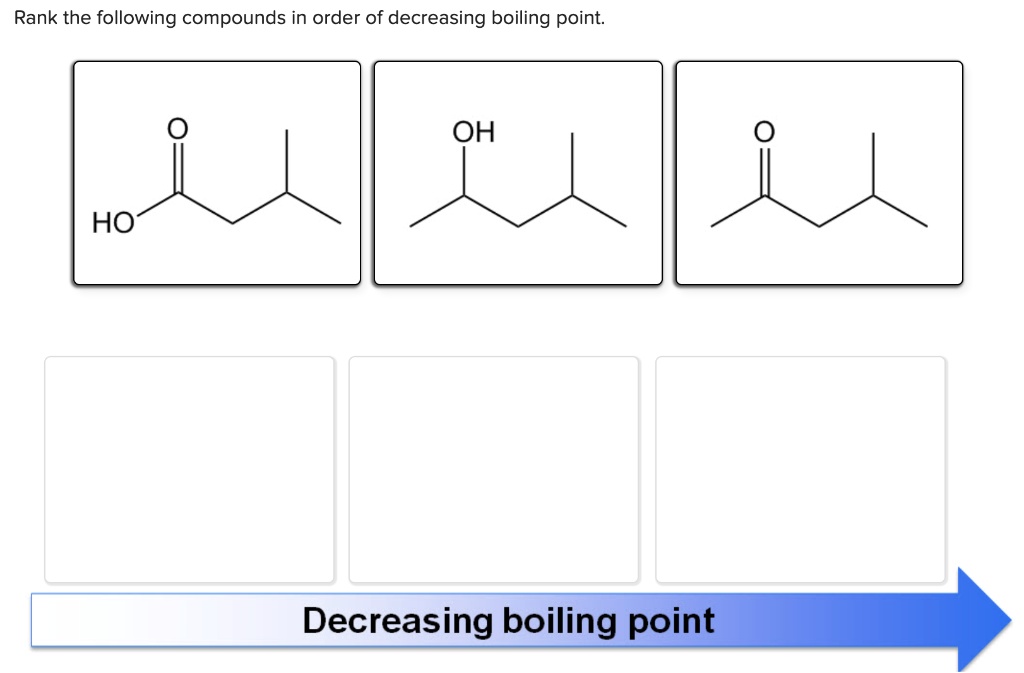
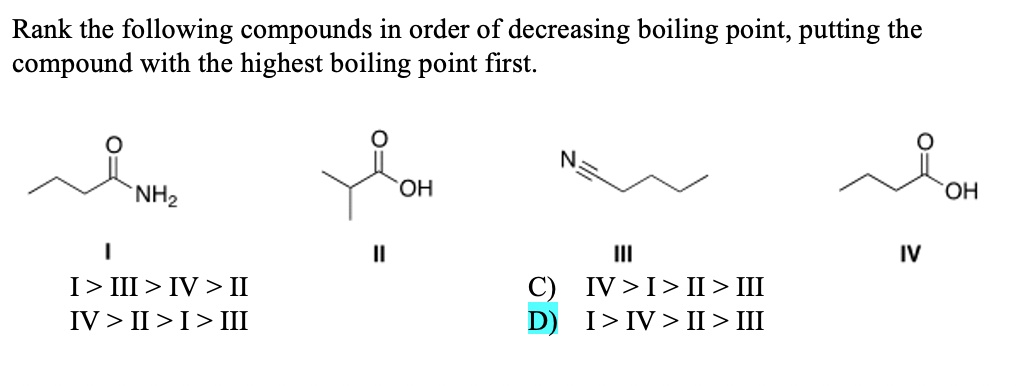
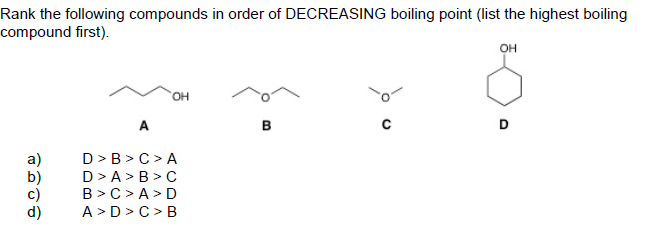
Rank The Following Compounds In Decreasing Order Of Boiling Point
So, picture this: my buddy, let’s call him Steve, is trying to impress this new person he’s seeing. He decides he’s going to whip up this fancy pasta dish, you know, the kind with the really specific olive oil and a sauce that takes hours. He’s feeling all suave, got the apron on, the whole nine yards. The recipe calls for him to boil water, obviously. And Steve, being Steve, decides he’s going to use the biggest pot he has. Like, ridiculously big. He pours in what feels like a gallon of water. The stove burner is going full blast, but this water… it’s just… simmering. Barely even bubbling. He’s tapping his foot, checking his watch, getting genuinely frustrated. Meanwhile, I’m over there, watching him struggle, and I’m thinking, “Dude, maybe it’s not just about the heat, it’s about… well, what’s in the pot, and how much of it there is.”
And that, my friends, is kind of how I felt when I was staring at this list of compounds. We’re talking about ranking them by their boiling points. Now, you might be thinking, “Boiling point? Isn’t that just when stuff gets hot enough to turn into a gas?” And yeah, that’s the super basic answer. But like Steve’s water situation, there’s a whole lot more going on under the surface (or, you know, in the liquid). It’s not just about jamming more heat into it; it’s about how those little molecules are holding onto each other. Some are practically begging to break free, while others are in a serious group hug, needing a serious shove to let go.
So, the compounds we’re looking at today are:
- Methane (CH₄)
- Ethane (C₂H₆)
- Propane (C₃H₈)
- Butane (C₄H₁₀)
- Pentane (C₅H₁₂)
See that pattern? They’re all hydrocarbons. Specifically, they’re alkanes. That means they’re just made of carbon and hydrogen atoms, linked together in a chain. Kinda like a little molecular train. And the cool thing is, we can predict a lot about their physical properties just by looking at how big these trains are.
Our mission, should we choose to accept it (and we totally have to, because it’s right there in front of us!), is to rank these guys from the highest boiling point to the lowest boiling point. Think of it like a competition: who needs the most energy (heat) to go from being a liquid to a gas? Who’s the most reluctant to leave their buddies?
Let’s dive in, shall we? Grab a comfy seat, maybe a beverage of choice. We’re going on a little chemistry adventure.
The Molecular Hug: Why Some Things Boil Easier Than Others
Okay, before we get into the nitty-gritty of methane and butane, let’s talk about the why. Why do some liquids boil at higher temperatures than others? It all boils down to the forces between the molecules, or as the fancy folks call them, intermolecular forces. You can think of these like invisible hands that molecules use to hold onto each other.
For these specific alkanes, the main intermolecular force at play is called the London dispersion force (or van der Waals forces, if you want to get even more technical). These forces arise from temporary fluctuations in electron distribution around the molecules. Basically, at any given moment, the electrons in a molecule might be a little more on one side than the other, creating a temporary, weak positive and negative charge. This little jiggle can then induce a similar jiggle in a neighboring molecule, creating a temporary attraction.
Now, here’s the kicker: the strength of these London dispersion forces depends on how many electrons a molecule has and how large and spread out its electron cloud is. Bigger molecules, with more electrons, have stronger London dispersion forces. It’s like trying to pull apart a small group of people versus a massive, interconnected crowd. The crowd is going to be a lot harder to disperse.
So, more electrons = stronger London dispersion forces = more energy needed to overcome those forces = a higher boiling point.
Think of it like this: imagine you’re trying to get your pet cat off your lap. If your cat is a tiny kitten, you can probably just lift it off easily. But if your cat is a huge Maine Coon, that’s going to take a bit more effort! The bigger, heavier cat has more… well, catness to resist being moved, just like bigger molecules have more intermolecular forces.
Another factor, though less significant for these particular alkanes which are all nonpolar, is molecular shape. More spread-out molecules can have more surface area for these interactions, leading to stronger forces. But for our alkanes, the primary driver is size and the resulting number of electrons.
So, in our list of methane, ethane, propane, butane, and pentane, we’re essentially looking at a series of molecules that are getting progressively bigger and carrying more electrons as we move down the list. This means their intermolecular forces will get progressively stronger.
The Contenders: Let’s Meet the Molecules
Here they are, in all their simple, hydrocarbon glory:
Methane (CH₄)
This is the smallest one. One carbon atom, four hydrogen atoms. It’s like the baby of the group. Its molecular weight is low, meaning it has the fewest electrons. Because of this, the London dispersion forces between methane molecules are extremely weak. It’s the least likely to want to stick to its neighbors.
Ethane (C₂H₆)
Next up, we have ethane. Two carbon atoms and six hydrogen atoms. It’s a step up in size from methane. It has more electrons, so the London dispersion forces are a bit stronger than in methane. It’s like the toddler of the group – a bit more established, but still pretty easy to manage.
Propane (C₃H₈)
Propane. Three carbon atoms and eight hydrogen atoms. We’re building up the chain here. More electrons means stronger intermolecular forces than ethane. This is the teenager – definitely more substance, a bit more resistant to being pushed around.
Butane (C₄H₁₀)
Butane. Four carbon atoms and ten hydrogen atoms. It’s getting substantial! More electrons, stronger London dispersion forces. This is your adult friend who is firmly planted in their ways. It takes more to convince them to change their state.
Pentane (C₅H₁₂)
And finally, pentane. Five carbon atoms and twelve hydrogen atoms. This is the big kahuna of our little group. It has the most electrons, and therefore, the strongest London dispersion forces. This molecule is giving its neighbors a really good hug. It’s going to take the most energy to pry them apart.
The Verdict: Ranking Them From Strongest Hug to Weakest
Alright, drumroll please! Based on what we’ve discussed about molecular size and intermolecular forces, we can now confidently rank these compounds in decreasing order of boiling point. Remember, decreasing order means starting with the highest boiling point and going down to the lowest.
The Top Contender (Highest Boiling Point): Pentane (C₅H₁₂)
Pentane is our winner! With its longer carbon chain, it has the most electrons and the strongest London dispersion forces. These molecules are really clinging to each other, so it takes a significant amount of heat to give them enough energy to break free and become a gas. Think of it as trying to get a whole bunch of grown-ups to let go of each other – takes some serious persuasion (and heat!).
Second Place: Butane (C₄H₁₀)
Butane comes in next. It’s a bit smaller than pentane, so its London dispersion forces are a little weaker. It still needs a good amount of heat to boil, but not quite as much as pentane. It’s like getting a group of older teenagers to release their grip – still strong, but a tad less formidable.
Third Place: Propane (C₃H₈)
Propane is our bronze medalist. It’s smaller still, with weaker intermolecular forces. It’ll boil at a lower temperature than butane. Think of this as a group of regular adults. They’re holding on, but you can probably convince them to loosen up with moderate effort.
Fourth Place: Ethane (C₂H₆)
Ethane takes fourth place. It’s smaller than propane, meaning its London dispersion forces are weaker. It requires less heat to become a gas. This is like a bunch of younger adults – still have some strength, but not as much as the older ones.
The Underdog (Lowest Boiling Point): Methane (CH₄)
And the compound with the lowest boiling point is, you guessed it, methane! This tiny molecule has the fewest electrons, resulting in the weakest London dispersion forces. Methane molecules barely interact with each other. It takes very little energy to get them to switch from liquid to gas. It’s like trying to get a group of small children to let go of each other – they might be holding hands for a second, but a gentle nudge is all it takes! Methane is actually a gas at room temperature and pressure, which is a direct consequence of its very low boiling point.
The Full Ranking (Decreasing Order of Boiling Point):
- Pentane (C₅H₁₂) - Highest Boiling Point
- Butane (C₄H₁₀)
- Propane (C₃H₈)
- Ethane (C₂H₆)
- Methane (CH₄) - Lowest Boiling Point
Pretty neat, right? It’s all about those intermolecular forces, and for these alkanes, size really matters. The bigger they are, the more they stick together, and the more heat it takes to make them boil.
So, next time you’re watching water boil, or maybe even some cooking oil, you can impress your friends (or just yourself!) by thinking about the invisible forces at play. It’s not just about the heat; it’s about how those little molecules are dancing with each other.
And remember Steve and his giant pot of water? If he were trying to boil pentane instead of water, he’d need a lot more than just a big pot and a strong burner. But that’s a story for another day! For now, I hope this little dive into the world of boiling points has been as fun and enlightening for you as it was for me.
