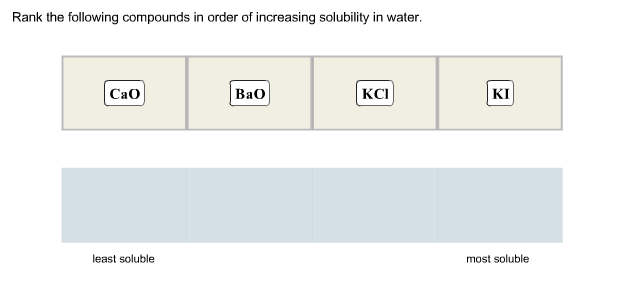
Rank The Following Compounds According To Increasing Solubility In Water

Alright, so we've all been there, right? You're in the kitchen, maybe attempting to whip up some fancy concoction that looks way cooler on Pinterest than it does in reality. Or perhaps you're just trying to make a decent cup of tea, a feat that, surprisingly, can sometimes feel like rocket science. And then it hits you: the age-old question, the one that might have kept even Sir Isaac Newton up at night (okay, maybe not that old, but you get the drift). We're talking about solubility, folks! Specifically, how well things like to chill out and mix with our good old pal, water.
Think of it like this: water is the ultimate party host. It's super welcoming and loves to invite all sorts of molecules to join the shindig. But, just like at any good party, some guests are naturally more social butterflies than others. Some just slurp up the hospitality and blend right in, while others are a bit more… standoffish. They'd rather cling to their friends or just stand awkwardly in the corner, pretending to check their phone.
Today, we're going to dive headfirst into a little solubility showdown. We've got a lineup of compounds, and we're going to rank them from "barely even bothered to show up" to "absolutely living their best life in the water." It's like grading your friends on how much they'll help you move. Some will be there bright and early with gloves and a smile, others will conveniently "forget" and send a text saying they're "stuck in traffic" (wink wink). Let's get this party started!
The Usual Suspects: Our Solubility All-Stars (and Benchwarmers)
So, who are our contenders in this aquatic arena? We've got a motley crew, ready to show us what they're made of. Imagine these guys as characters in a quirky indie film. We've got:
- Sodium Chloride (NaCl): This is your classic table salt. Think of it as the life of the party, the one who knows everyone and is immediately handing out hugs.
- Potassium Iodide (KI): A bit fancier than your everyday salt, this one is like the reliable friend who's always up for anything, but maybe with a slightly more refined taste.
- Magnesium Sulfate (MgSO₄): Epsom salt! This is the friend who's always trying to "help" you relax, like offering a soothing bath. A bit more chill than salt, but still pretty keen to get involved.
- Calcium Carbonate (CaCO₃): This is your chalk, your seashells, your antacid tablets. This guy is a bit of a loner, prefers to keep to himself. Think of him as that acquaintance you see at parties but never really talk to.
- Barium Sulfate (BaSO₄): This one is famously used in medical imaging – the "barium swallow." It's designed not to dissolve, so it can paint a clear picture. This is the friend who shows up to your party, takes one look around, and says, "Nah, I'm good," and heads straight back home.
Now, before we get all scientific on you, let's remember what makes something "dissolve." It's all about those sneaky little water molecules, H₂O. They're like tiny, energetic cheerleaders with a positive and a negative end. When you plop an ionic compound (like our salts) into water, these water molecules get super excited. They surround the individual ions – the positive bits and the negative bits – and gently pull them apart, letting them swim around freely in the water. It's like a friendly game of "tag" where water is "it," and the ions are the players getting scooped up.
But here's the kicker: some ions are just way more attractive to water than others. It's all about that intermolecular "pizzazz," that "sparkle" that makes water want to hug them tight. Other ions, well, they're more content cuddling up with their own kind. They have a stronger "bond" with their fellow ions than they do with those energetic water molecules.
The Great Water Dive: Our Solubility Rankings!
So, let's get down to brass tacks, or should I say, dissolving tacks? We're ranking these compounds from the least likely to make a splash to the ones that dive in headfirst. Imagine it like a diving competition, where points are awarded for how gracefully you enter the water.
The "Nah, I'm Good" Award: Barium Sulfate (BaSO₄)
First up, the undisputed champion of not dissolving: Barium Sulfate. This compound is practically the king of "social distancing" in water. It's so insoluble, it's like it wears a little sign that says, "Please do not disturb." When you throw barium sulfate into water, it just sits there, looking all smug. The barium ions and sulfate ions are so incredibly happy snuggling with each other, they barely notice the water molecules waving hello.
Think of it like trying to get your cat to take a bath. You can lure it with treats, you can sing sweet lullabies, but at the end of the day, it's going to give you the stink eye and probably try to escape. Barium sulfate is our cat. It loathes the water. Its attraction to its own ions is so strong, the water molecules just can't break it up. It's practically hydrophobic – water-repelling!
In the world of chemistry, we'd say it has a very low solubility product constant (Ksp). Don't let the fancy term scare you; it just means it doesn't like to break apart into ions very much. It's the ultimate wallflower, or perhaps more accurately, the ultimate "stay-at-home" friend. You invite it to the pool party, and it shows up in pajamas with a good book.
The "Maybe Later" Award: Calcium Carbonate (CaCO₃)
Next on our list, we have Calcium Carbonate. This is the stuff that makes up seashells, limestone, and even the chalk we use on blackboards. It's slightly more cooperative than barium sulfate, but not by much. It's like that friend who says they'll come to your party but then shows up an hour late, complains about the music, and leaves after one drink.
Calcium carbonate does dissolve a little bit in water. You can get a tiny, almost imperceptible amount to break apart into calcium ions (Ca²⁺) and carbonate ions (CO₃²⁻). But for the most part, it's like, "Thanks for the invite, water, but I'm perfectly happy hanging out with my carbonate buddies." The bond between the calcium and carbonate ions is pretty strong, so the water molecules have their work cut out for them trying to pry them apart.
If you've ever seen a bathtub ring, or noticed how hard water can be (which is often due to dissolved calcium and magnesium compounds), you're seeing calcium carbonate's reluctance to fully dissolve. It's the reason why scaling builds up in pipes. It's just not that into being dissolved. It prefers to stay solid, forming those lovely (or annoying, depending on your perspective) mineral deposits.

Compared to barium sulfate, it's a bit of a step up in solubility. It's like the friend who'll at least get out of their pajamas, but still won't get on the dance floor. A definite improvement, but not exactly a rave.
The "I'm Trying My Best" Award: Magnesium Sulfate (MgSO₄)
Now we're starting to get somewhere! Enter Magnesium Sulfate, or as you probably know it, Epsom salt. This is the friend who's genuinely trying to be helpful. You put magnesium sulfate in water, and it's like, "Okay, water buddies, let's do this!" The magnesium ions (Mg²⁺) and sulfate ions (SO₄²⁻) are a bit more willing to let go of each other and get mixed up with the water molecules.
Magnesium sulfate is pretty soluble. It's not going to be shy when it comes to joining the water party. You can add a good amount of it to water, and it will happily dissolve, creating that soothing bath experience. Think of it as the friend who actually brings a dish to the potluck and stays for the whole night, even helping with the cleanup.
The attraction between magnesium and sulfate ions is weaker than the attraction in calcium carbonate or barium sulfate. This means that the energetic water molecules have a much easier time breaking those bonds and surrounding the individual ions. It's a nice, healthy interaction. You can imagine the water molecules giving the magnesium and sulfate ions a big, watery hug, and they're like, "Yeah, this feels good!"
So, while it's not the absolute most eager to dissolve, it's definitely a solid contender. It's the reliable guest who always adds to the fun. You can count on magnesium sulfate to make a noticeable difference in your water's composition.
The "Here for a Good Time!" Award: Potassium Iodide (KI)
Getting warmer! Our next contestant is Potassium Iodide. This is like the friend who arrives early, brings the best snacks, and is already initiating a group chat for the next outing. Potassium iodide is very soluble in water.
When you add potassium iodide to water, it's like a stampede of happy ions. The potassium ions (K⁺) and iodide ions (I⁻) are practically leaping into the water, eager to be surrounded and dispersed. The forces holding the potassium and iodide ions together are relatively weak compared to the forces between the water molecules and the ions.
This means that even a small amount of water can accommodate a large amount of potassium iodide. It dissolves quickly and completely, creating a clear solution. It's the friend who's always available for a spontaneous adventure. You suggest a road trip, and they're already packing their bags. No hesitation, just pure enthusiasm.
Potassium iodide is the kind of compound that makes chemistry experiments fun because you don't have to worry about it stubbornly refusing to dissolve. It just goes with the flow. It's the opposite of the lone wolf; it's a team player of the highest order.
The "Life of the Party" Award: Sodium Chloride (NaCl)
And finally, the reigning monarch of everyday solubility: Sodium Chloride! Yes, our humble table salt! This is the friend who is always invited, always has a blast, and is probably organizing the next party. Sodium chloride is incredibly soluble in water.

When you add salt to water, it's an instant party. The sodium ions (Na⁺) and chloride ions (Cl⁻) are so readily attracted to the water molecules that they break apart and disperse with gusto. The water molecules are just itching to get their hands on those ions. They grab them, swirl them around, and make them feel right at home.
You can dissolve a lot of salt in a given amount of water before it starts to get crowded. Think about making a salt solution – you can keep adding salt, and it keeps disappearing into the water, making it tastier (or at least, more like the sea!). It’s that one friend who’s up for anything, the one who gets the dance floor going. They’re the glue of the social group, the one everyone else gravitates towards.
The reason for this epic solubility is the strong electrostatic attraction between the sodium and chloride ions, but also the even stronger attraction between these ions and the polar water molecules. Water is really good at solvating (surrounding and stabilizing) both Na⁺ and Cl⁻ ions. It’s a perfect match made in… well, a beaker!
The Final Order: From Awkward Guest to Dancing Fool!
So, let's line 'em up, from the shyest wallflower to the wildest dancer. In order of increasing solubility in water (meaning, from least to most soluble), our compounds are:
- Barium Sulfate (BaSO₄): The one who sends their regrets before the party even starts.
- Calcium Carbonate (CaCO₃): The one who shows up late, checks their watch, and leaves early.
- Magnesium Sulfate (MgSO₄): The reliable friend who brings a good attitude and maybe a dip.
- Potassium Iodide (KI): The energetic one who’s ready to go from the get-go.
- Sodium Chloride (NaCl): The absolute life of the party, everyone’s favorite!
And there you have it! A little peek into the fascinating world of solubility. It's not just about mixing things in a lab; it's about understanding why some things blend in so easily and others prefer to keep to themselves. Next time you're making a salty soup or admiring a seashell, you'll have a little chuckle knowing the chemistry behind why things do (or don't) dissolve. Happy dissolving, everyone!
