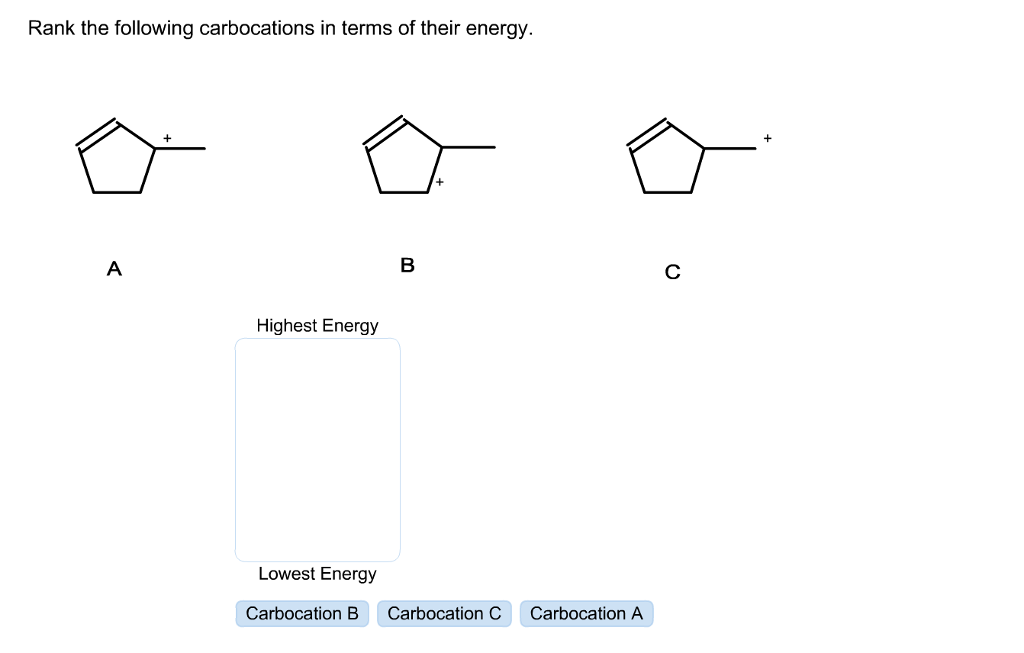
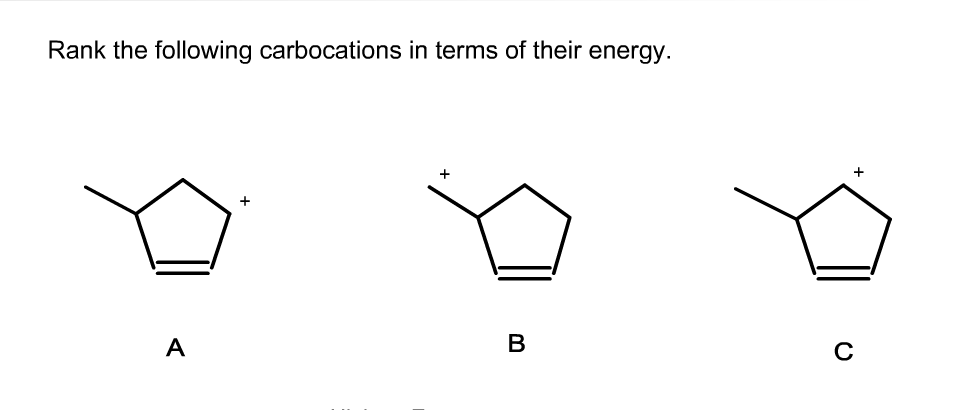
Rank The Following Carbocations In Terms Of Their Energy.
Ever felt like your energy levels just can't keep up? Sometimes it feels like a cosmic joke, right? One minute you're ready to conquer the world, the next you're contemplating the profound mysteries of the ceiling fan. Well, in the wild, wonderful world of chemistry, a similar kind of "energy" drama unfolds, but with molecules instead of humans. Today, we're diving into the surprisingly relatable concept of carbocations and how their energy levels stack up. Think of it as the ultimate molecule mood board – who's feeling the most stable, and who's about to have a full-on breakdown?
Now, before your eyes glaze over with flashbacks to high school chemistry class, let's inject some fun. Carbocations aren't just abstract lab concepts; they're often fleeting intermediates in reactions that lead to the creation of everything from the plastics in your phone to the flavors in your favorite snacks. So, understanding their energy is like getting a sneak peek into the secret life of molecules. It’s like knowing the backstage gossip of a rock concert – fascinating, slightly chaotic, and ultimately important for the show to go on.
At its core, a carbocation is a molecule that’s got a carbon atom carrying a positive charge. Imagine a little dude with a backpack that's way too heavy and he's constantly worried about dropping it. That positive charge means this carbon is feeling a bit… incomplete. It’s looking for some electron love, some stability, some… chill. And just like us, some carbocations are better at finding that chill than others.
The Energy Ladder: Who's Winning the Stability Game?
So, how do we rank these positively charged characters? It all comes down to stability. The more stable a carbocation is, the lower its energy. Think of it like a well-rested yogi versus someone who just pulled an all-nighter cramming for an exam. The yogi is radiating calm (low energy), while the all-nighter is practically vibrating with nervous energy (high energy). The same principle applies here, just with a slightly more scientific vocabulary.
The biggest factors influencing carbocation stability are the groups attached to that positively charged carbon. These surrounding groups can either help stabilize the positive charge or, unfortunately, make it even worse. It's a delicate balancing act, much like trying to juggle a latte, your phone, and your keys while walking down a busy street – one wrong move and everything goes flying!
The Usual Suspects: A Ranking from High to Low Energy
Let's break down the common players in the carbocation energy game. We'll go from the most energetic (least stable) to the most chill (most stable). Imagine a cosmic popularity contest for molecules – the more stable ones are the A-listers, effortlessly blending in and exuding confidence.
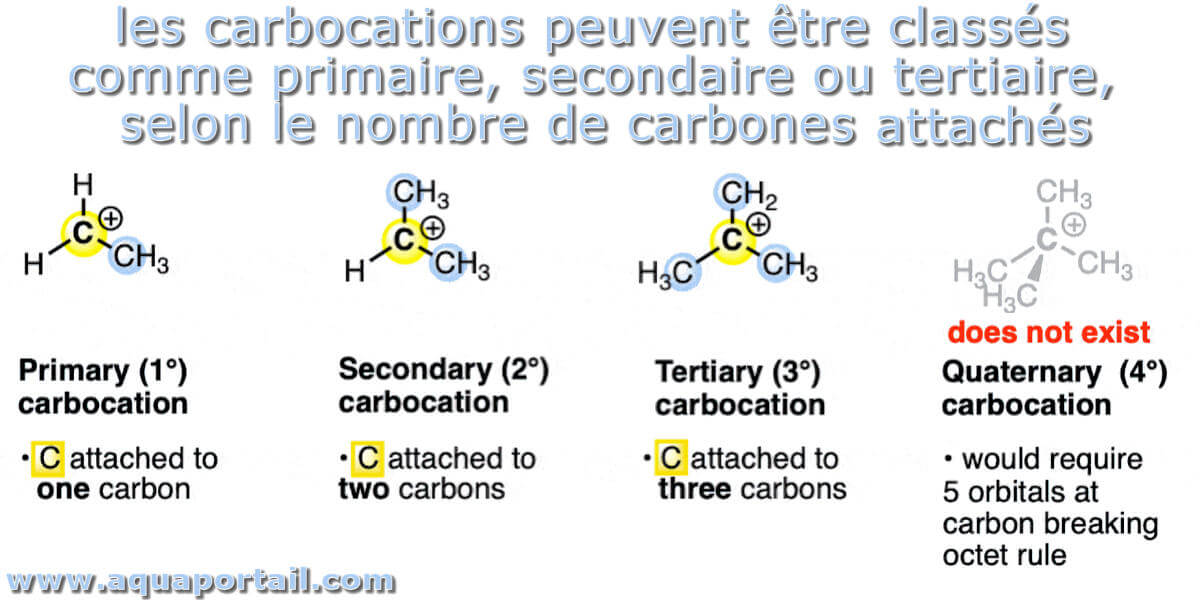
1. The Methyl Carbocation (CH₃⁺): The Unsung Hero of High Energy
At the very top of our energy ladder, feeling the most anxious and unstable, is the methyl carbocation. This little guy has just one carbon atom with a positive charge and three tiny hydrogen atoms hanging on. There's not much going on here to help it out. It's like a solo artist with no backing band – exposed and a bit vulnerable.
Why is it so energetic? Well, the positively charged carbon is pretty much on its own. Hydrogen atoms are not particularly good at sharing their electron love. They can't really delocalize or spread out that positive charge. So, this methyl carbocation is like a phone with 1% battery left – extremely high energy and desperately seeking a charger (or, in this case, electrons).
This is the carbocation you'll see in reactions where there are no other options, or where it's just a fleeting, theoretical entity. It’s the molecule equivalent of that awkward silence after someone tells a bad joke.
2. The Primary Carbocation (RCH₂⁺): A Slight Improvement
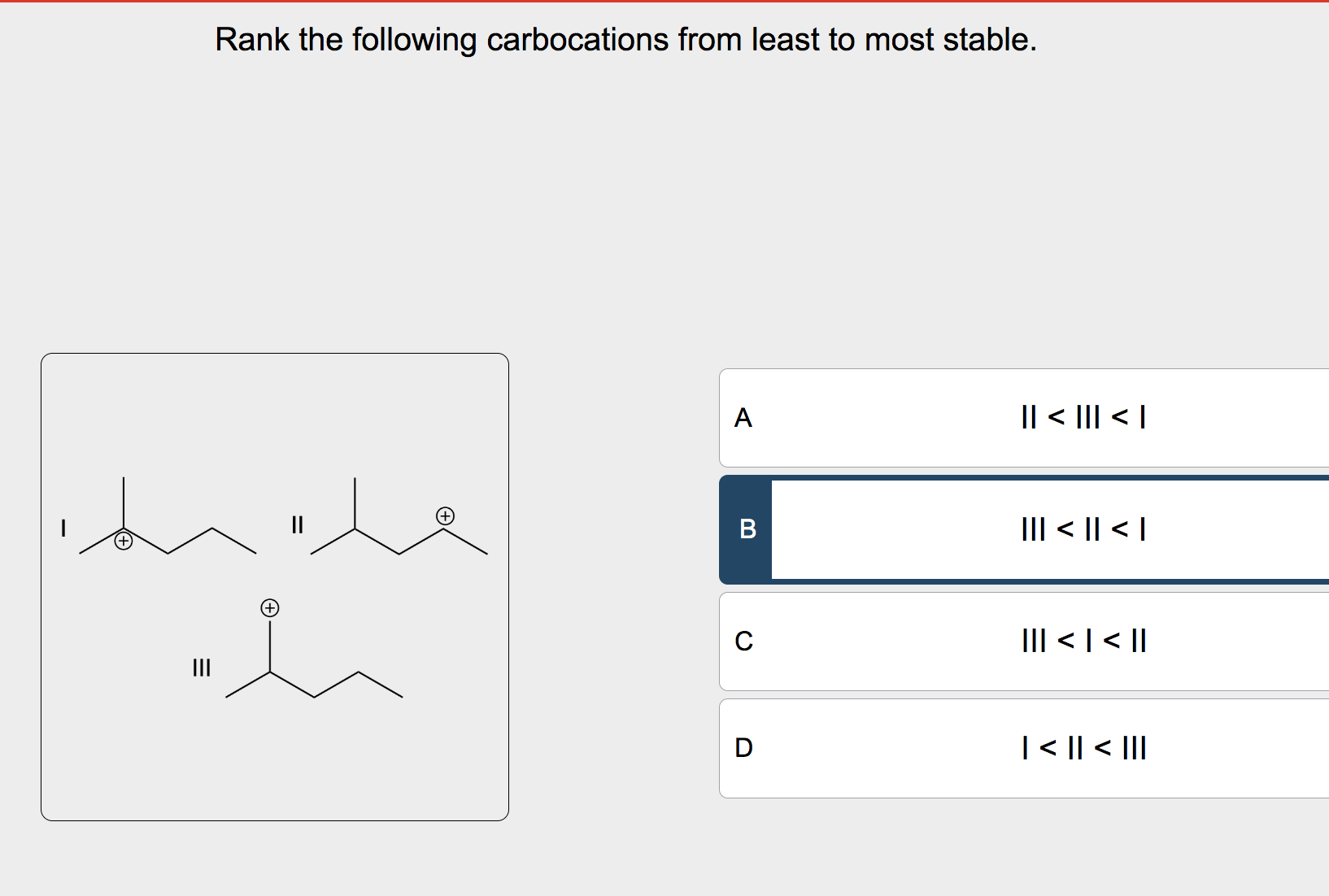
Next up, we have primary carbocations. Here, the positively charged carbon is attached to one other carbon-containing group (we call this an "R" group, which can be anything from a simple methyl group to a more complex chain) and two hydrogen atoms. Think of it as the methyl carbocation getting a plus-one to the party.
The presence of that R group is a small win. Carbon atoms are a bit better at sharing electron density than hydrogen. This is through something called inductive effect. Basically, the R group can push a little bit of electron density towards the positively charged carbon, making it feel a tiny bit more comfortable. It’s like having a friend nod in agreement during a slightly embarrassing moment – it helps, but it doesn't completely fix things.
Primary carbocations are still quite energetic, but they're a step up from the methyl. They're like someone who’s just managed to find a seat on a crowded bus – not ideal, but definitely better than standing.
3. The Secondary Carbocation (R₂CH⁺): Getting Warmer
Now we're entering the realm of "pretty stable." Secondary carbocations have the positively charged carbon attached to two R groups and one hydrogen atom. This is where things start to get significantly more chill.
With two R groups, there's a much more substantial electron-donating effect happening. These two carbon groups are working together, pushing electron density towards the positive center. This is like having two friends there to have your back. The positive charge is more spread out, and the carbocation feels much more at ease. It's like finding a cozy armchair after a long day – pure bliss.
These are commonly encountered in many organic reactions. Think of them as the reliable middle-grounders, not too stressed, not too laid-back, just… getting the job done.
4. The Tertiary Carbocation (R₃C⁺): The Ultimate Chill Master
And finally, we reach the pinnacle of carbocation stability: the tertiary carbocation. Here, the positively charged carbon is connected to three R groups. This is the molecule equivalent of a full spa day, complete with cucumber water and fluffy robes.
With three R groups, the inductive effect is maximized. These three carbon groups are all working in harmony to surround the positive charge with electron density, effectively neutralizing it and making the carbocation incredibly stable. The positive charge is so delocalized and dispersed that it barely feels positive at all. It's like being wrapped in a warm blanket of electron love. These are the carbocations that are most likely to form and persist long enough to participate significantly in reactions.
Think of a tertiary carbocation as that friend who always seems to have it all together, radiating an aura of calm confidence. They’re the ones you go to when you need advice, because they’re just that stable.
The Role of Resonance: When Molecules Get Fancy
But wait, there's more! The energy game gets even more interesting when we introduce the concept of resonance. Resonance happens when a positive charge (or other charge) can be spread out over more than two atoms by delocalizing electrons through adjacent pi systems (double or triple bonds). This is like having a super-powered ability to share your good vibes.
Imagine a carbocation next to a double bond. The electrons in the double bond can shift, allowing the positive charge to be shared between the original carbon and the carbon of the double bond. This is like a group hug for electrons, and it dramatically increases stability. Molecules that can undergo resonance are often much more stable than their non-resonant counterparts, even if they are primary or secondary.
Allylic and Benzylic Carbocations: The Resonant Rockstars
For example, an allylic carbocation (positive charge on a carbon next to a C=C double bond) and a benzylic carbocation (positive charge on a carbon attached to a benzene ring) are significantly more stable than a tertiary carbocation. This is because the positive charge can be delocalized through resonance over multiple carbon atoms. The benzene ring, with its delocalized pi system, is especially good at stabilizing positive charges.
Think of these as the influencers of the carbocation world. They've got that extra something that makes them incredibly popular and sought-after. They're the celebrities of the molecular universe, always in the spotlight (and very stable while doing it).
Putting It All Together: The Grand Energy Symphony
So, if we were to create a definitive ranking of carbocation energy levels, from highest (least stable) to lowest (most stable), it would look something like this:
- Methyl Carbocation (CH₃⁺) – Highest Energy, Least Stable
- Primary Carbocation (RCH₂⁺) – High Energy
- Secondary Carbocation (R₂CH⁺) – Moderate Energy, More Stable
- Tertiary Carbocation (R₃C⁺) – Low Energy, Very Stable
- Allylic and Benzylic Carbocations – Even Lower Energy, Extremely Stable (due to resonance)
This ranking is fundamental to understanding how many chemical reactions proceed. For instance, in reactions where a carbocation is an intermediate, the more stable carbocations are more likely to form. This is why reactions often favor pathways that lead to tertiary or resonance-stabilized carbocations.
It’s like choosing the path of least resistance in life. You’d rather take the express train to your destination than a convoluted bus route, right? Molecules are no different; they'll gravitate towards the most stable (lowest energy) pathways.
Beyond the Lab: What Carbocations Teach Us About Life
Okay, so we've journeyed through the energetic landscape of carbocations. But does this have any bearing on our daily lives, beyond the occasional organic chemistry quiz? Absolutely! The principle of stability is a universal one.
Think about it: we all strive for stability. We build strong relationships, find secure jobs, and create comfortable homes because these things provide us with a sense of stability and lower our "life energy" – our stress, our anxiety, our general feeling of being overwhelmed. Just like a carbocation seeks electron density to feel more stable, we seek supportive networks and fulfilling experiences to feel more grounded.
The more "electron density" we have in our lives – whether it’s strong friendships, a loving family, a passion project, or even just a good book and a cup of tea – the more stable and content we tend to feel. The less support we have, the more we might feel like that highly energetic, unstable methyl carbocation, feeling a bit lost and searching for something to hold onto.
Furthermore, the concept of resonance reminds us that sometimes, the best way to navigate a challenge is to spread the load. Instead of bearing a burden alone, we can share it with others, and together, we can find a more stable and harmonious solution. This is the power of teamwork, community, and open communication.
So, the next time you’re feeling a bit wobbly, a bit high-energy and anxious, remember the humble carbocation. It's a reminder that stability is attainable, and that by understanding what makes us (or them) stable, we can actively work towards creating more of it in our own lives. It’s about finding our electron donors, our resonance structures, our own little pockets of molecular peace. And in this fast-paced world, a little bit of molecular zen can go a long way.
