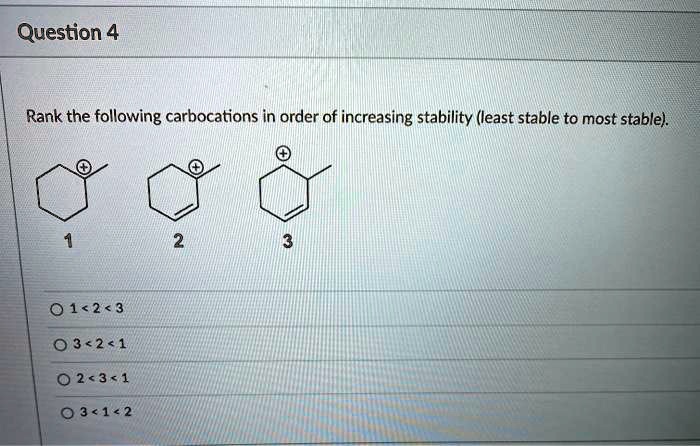
Rank The Following Carbocations In Order Of Increasing Stability
Hey there, you! Grab your coffee, settle in. We’re about to dive into something that might sound a little… intense. Carbocations. Yeah, I know, sounds like something out of a sci-fi movie, right? But trust me, it’s way more interesting than it sounds. Think of them as these little positively charged buddies hanging out in the molecular world, and like most of us, they have their good days and their not-so-good days. Today, we’re ranking them, from the least chill to the most zen. Let’s get this party started, shall we?
So, what exactly is a carbocation? Imagine a carbon atom that’s usually all happy and making four bonds. Well, in a carbocation, it’s lost an electron, making it super electron-deficient and, you guessed it, positively charged. It’s basically the drama queen of the organic chemistry world, always looking for some electron love. And when it comes to stability, oh boy, does it have preferences.
Think of stability like this: how happy is this little guy? Is it jumping around, desperate for a stable relationship (you know, an electron)? Or is it just chilling, content with its lot? That’s what we’re figuring out. And the factors that make one carbocation happier than another? It’s all about how they can spread that positive charge around, like gossip at a high school reunion.
The Least Chill: The Humble Methyl Carbocation
Alright, let's meet our first contestant. Drumroll, please… the methyl carbocation!
\( \text{CH}_3^+ \)
. This little dude is basically the newbie in town. It's just a carbon with three hydrogens, and it's rocking a big ol' positive charge all by itself. No buddies to help it out, no electron-donating neighbors. It’s like being the only one at a party without a date, or maybe a really awkward elevator ride with a stranger. Not exactly ideal, right?Why is it so unstable? Well, that positive charge is just… sitting there. Exposed. Vulnerable. Imagine trying to hold a bunch of balloons by yourself. One wrong move, and they all go flying! The methyl carbocation is pretty much the same. It desperately wants to find some electron density to calm its nerves. It’s the definition of high-strung. It’s like, “Please, somebody, anybody, give me an electron!” You feel for it, don’t you?
This is the baseline, folks. This is where we start. Everything else we talk about will be compared to this little guy’s anxiety. So, when you see the methyl carbocation, just give it a little nod of understanding. It’s going through it.
Slightly Less Anxious: The Primary Carbocation
Next up, we have the primary carbocation. This one’s a little better off. Imagine our positively charged carbon now being attached to one other carbon atom, which is then attached to hydrogens. So, something like
\( \text{RCH}_2^+ \)
, where R is our alkyl group. It's like bringing a friend to that awkward party. Still a bit nerve-wracking, but at least you’re not completely alone!
What’s the big deal with having that one extra carbon? Well, alkyl groups are actually pretty good at donating electron density. It’s like they’re whispering sweet nothings to the positive carbon, saying, “Hey, it’s okay, we’ve got your back.” This is called inductive effect. Think of it as a tiny, helpful push of electrons towards the needy carbon. It’s not a massive relief, but it’s something. It takes some of the edge off that positive charge.
So, the primary carbocation is still a bit on edge, still looking for that electron fix, but it’s got a tiny safety net. It’s like going from a solo act to a duet. The pressure is slightly diffused. It’s definitely more stable than the methyl, which is a win in our book! Small victories, people, small victories.
Getting a Little More Comfortable: The Secondary Carbocation
Now we’re stepping up our game! Meet the secondary carbocation. This one has our positively charged carbon attached to two other carbon atoms. So,
\( \text{R}_2\text{CH}^+ \)
. It’s like bringing two friends to the party. Now things are starting to feel a bit more social, a bit more… stable. The positive charge is getting more company!Why is this better? Remember that inductive effect we talked about? Now we have two alkyl groups pushing electron density towards the positive carbon. That’s double the support! It’s like having a small cheering squad for our anxious carbon. The positive charge is getting spread out over a larger area, which makes it less concentrated and therefore, less intense. It’s like sharing a heavy load with friends – it feels lighter for everyone.
This is where things start to get significantly better. The secondary carbocation is definitely more content than its primary and methyl cousins. It’s still not exactly lounging on a beach, but it’s definitely moved off the edge of the cliff. It’s like, “Okay, this is manageable. I might even have a second drink.”
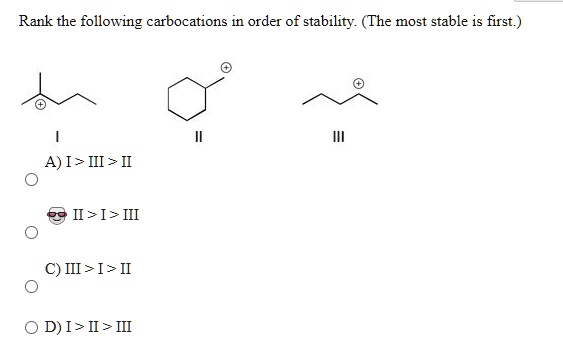
The VIP Club: The Tertiary Carbocation
And now, we arrive at the king of the carbocation hill: the tertiary carbocation! This guy has our positively charged carbon attached to three other carbon atoms.
\( \text{R}_3\text{C}^+ \)
What makes it so darn stable? You guessed it: three alkyl groups are donating electron density. That’s a triple dose of electron-donating goodness! The positive charge is being so effectively dispersed, so diluted, that the carbon atom is practically sighing with relief. It’s like having three people all trying to calm you down when you’re stressed. The intensity of the positive charge is significantly reduced. It’s like, “Ahhh, this is the life.”
But wait, there’s even more to the tertiary carbocation’s popularity contest. It's not just inductive effects. We also have this fancy thing called hyperconjugation. Don't let the name scare you! It’s basically when the electrons in the C-H bonds next to the positive charge can sort of… overlap with the empty p-orbital of the carbocation. Think of it as a little bit of electron sharing happening above and below the plane of the carbocation. It's like extending the electron cloud, making the positive charge even more spread out and stable.
The more hydrogens on the carbons adjacent to the carbocation, the more hyperconjugation can happen. So, a tertiary carbocation, with all those R groups, has a ton of these C-H bonds ready to lend a hand. It's like a full-on support network. This is why tertiary carbocations are the rockstars of the carbocation world. They are the least anxious, the most chill, the most likely to be found sipping on a perfectly mixed cocktail of electrons.
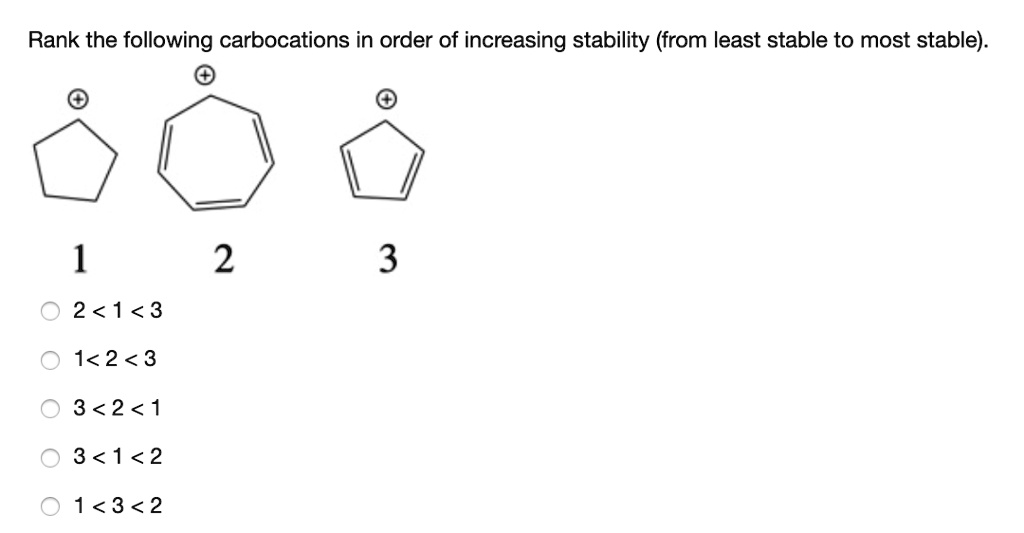
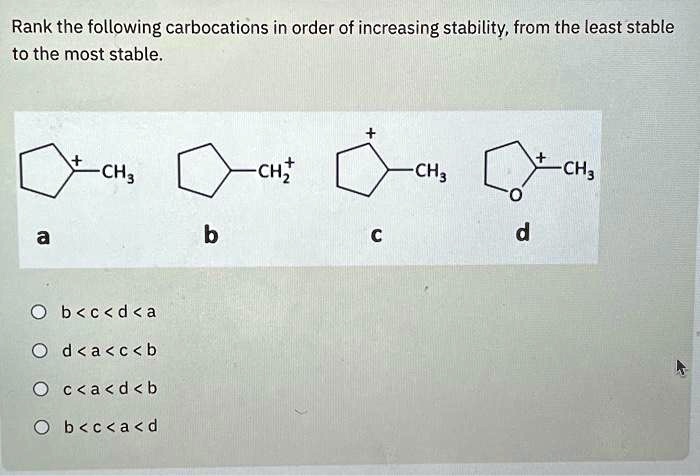
The Unexpected Player: Allylic and Benzylic Carbocations
Okay, okay, I know we’ve been talking about simple alkyl groups. But what about when our carbocation gets a little more… fancy? Enter the allylic and benzylic carbocations. These guys are special. They’re like the wildcard entries in our stability competition.
Let’s talk about the allylic carbocation first. This is where our positive charge is on a carbon atom that's next to a double bond. Like this:
\( \text{R}_2\text{C}^+ - \text{CH} = \text{CH}_2 \)
And then there’s the benzylic carbocation. This is where our positive charge is on a carbon atom that's directly attached to a benzene ring. Like
\( \text{Ph} - \text{CH}_2^+ \)
(where Ph is the benzene ring). Benzene rings are like the ultimate electron-donating superheroes because of their extensive pi electron system. This means that the positive charge can be delocalized all over the benzene ring. We're talking multiple resonance structures here, each one taking some of the positive charge. It's like the positive charge is getting VIP treatment, staying in luxurious, spread-out locations.So, where do these guys fit in? Generally, both allylic and benzylic carbocations are way more stable than simple primary, secondary, and even tertiary carbocations. Why? Because resonance is a much more powerful stabilizing force than inductive effects or even hyperconjugation. It’s like the difference between a gentle breeze and a hurricane of electron donation. The positive charge is just so much more spread out and happy.
Between allylic and benzylic, the benzylic is usually a bit more stable. Why? Because a benzene ring has a more extensive pi system than a single double bond, allowing for even greater delocalization of the positive charge. It’s like a bigger, fancier mansion for the positive charge to relax in.
Putting It All Together: The Grand Ranking!
Alright, the moment of truth! Let’s line them all up from the most anxious, the least stable, to the most chill, the most stable. Get ready for the reveal!
Increasing Order of Stability:
1. Methyl Carbocation \( (\text{CH}_3^+) \) : The loneliest number. No help whatsoever. Maximum drama.
2. Primary Carbocation \( (\text{RCH}_2^+) \) : Gets a little help from one alkyl group via inductive effect. Still a bit stressed.
3. Secondary Carbocation \( (\text{R}_2\text{CH}^+) \) : Two alkyl groups lending a hand. Things are improving! More electron donation, less concentrated positive charge.
4. Tertiary Carbocation \( (\text{R}_3\text{C}^+) \) : Three alkyl groups going all out with inductive effect and hyperconjugation. Pretty darn stable. The crowd favorite for simple alkyl carbocations.
5. Allylic Carbocation \( (\text{C=C-C}^+) \) : Resonance kicks in! The pi electrons of the double bond help delocalize the charge. Big jump in stability.
6. Benzylic Carbocation \( (\text{Ph-C}^+) \) : The king of delocalization! The extensive pi system of the benzene ring allows for massive charge spread. The most stable of the bunch we've discussed.
So, there you have it! The full spectrum of carbocation chill. It’s all about how effectively that positive charge can be spread out and stabilized. Think of it as a social network for a lonely positive charge. The more friends (electron-donating groups and resonance structures) it has, the happier it is.
Next time you see a carbocation, you'll know exactly how it's feeling. Is it a sweaty mess in the corner (methyl)? Or is it the life of the party, surrounded by adoring fans (benzylic)? It’s pretty cool to think about how these tiny molecular interactions dictate so much of what happens in chemical reactions, isn’t it? It’s like a whole tiny world of social dynamics happening right under our noses. Anyway, that’s all for today’s carbocation exposé. Hope your coffee is still warm!
